Abstract
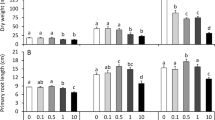
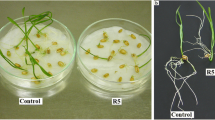
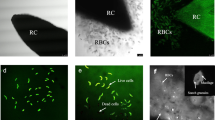
Allelopathic compounds released by invasive alien plants can suppress the growth of plants in their vicinity. The aim of this study was to investigate changes in tissue and cell structure in roots of radish seedlings treated with 10% aqueous extracts of rhizomes from the invasive knotweeds Fallopia japonica and F. ×bohemica. After 7 days of growth without and with aqueous extracts from these rhizomes, the anatomical and ultrastructural changes in the radish seedling roots were analyzed with light and transmission electron microscopy, and hydrogen peroxide was localized with diaminobenzidine, to define oxidative stress. The roots of radish seedlings treated with the knotweed extracts were shorter and thicker, due to the shorter and wider shapes of their cortex cells, which were organized in more columns than the control roots. There were signs of cell damage and oxidative stress in the root cap cells, and to a lesser extent in the meristematic zone. As well as the irregularly shaped nuclei and plasma membrane detached from the cell wall, the most prominent ultrastructural effects in the root cap cells of these aqueous rhizome extracts were the ring-shaped form of the mitochondria and large endoplasmic reticulum bodies. Excessive vacuolization was seen for the cells of the root apical meristem.








Similar content being viewed by others
References
Abrahim D, Braguini WL, Kelmer-Bracht AM, Ishii-Iwamoto EL (2000) Effects of four monoterpenes on germination, primary root growth, and mitochondrial respiration in maize. J Chem Ecol 26(3):611–624
Aliotta G, Cafiero G, Fiorentino A, Strumia S (1993) Inhibition of radish germination and root growth by coumarin and phenylpropanoids. J Chem Ecol 19(2):175–183
Babula P, Vaverkova V, Poborilova Z, Ballova L, Masarik M, Provaznik I (2014) Phytotoxic action of naphthoquinone juglone demonstrated on lettuce seedlings roots. Plant Physiol Biochem 84:78–86
Bailey JP, Bímová K, Mandák B (2009) Asexual spread versus sexual reproduction and evolution in Japanese knotweed s.l. sets the stage for the Battle of the Clones. Biol Invasions 11:1189–1203
Bell PR, Mühlethaler K (1964) The degeneration and reappearance of mitochondria in the egg cells of a plant. J Cell Biol 20:235–248
Benitez-Alfonso Y, Faulkner C, Ritzenthaler C, Maule AJ (2010) Plasmodesmata: gateways to local and systemic virus infection. Mol Plant Microbe Intaract 23(11):1403–1412
Bímová K, Mandák B, Pyšek P (2003) Experimental study of vegetative regeneration of four invasive Reynoutria taxa (Polygonaceae). Plant Ecol 166:1–11
Bogatek R, Gniazdowska A (2007) ROS and phytohormones in plant-plant allelopathic interaction. Plant Signal Behav 2(4):3317–3318
Bonnett HT, Newcomb EB (1965) Polyribosomes and cisternal accumulations in root cells of radish. J Cell Biol 27:423–432
Burgos NR, Talbert RE, Kim KS, Kuk YI (2004) Growth inhibition and root ultrastructure of cucumber seedlings exposed to allelochemicals from rye (Secale cereale). J Chem Ecol 30(3):671–689
Callaway RM, Aschehoug ET (2000) Invasive plants versus their new and old neighbors: a mechanism for exotic invasion. Science 290:521–523
Chen H, Tuck T, Ji X, Zhou X, Kelly G, Cuerrier A, Zhang J (2013) Quality assessment of Japanese knotweed (Fallopia japonica) grown on Prince Edward Island as a source of resveratrol. J Agric Food Chem 61:6383–6392
Cruz-Ortega R, Anaya AL, Hernández-Bautista BE, Laguna-Hernández G (1998) Effects of allelochemical stress produced by Sicyos deppei on seedling root ultrastructure of Phaseolus vulgaris and Cucurbita ficifolia. J Chem Ecol 24(12):2039–2057
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci. https://doi.org/10.3389/fenvs.2014.00053
De Feo V, De Martino L, Quaranta E, Pizza C (2003) Isolation of phytotoxic compounds from tree-of-heaven (Ailanthus altissima Swingle). J Agric Food Chem 51:1177–1180
Dolenc Koce J, Šoln K (2018) Phytotoxic effects of Fallopia japonica and F. ×bohemica leaves. Phyton 57(1/2):47–57
Fan P, Hay AE, Marston A, Lou H, Hostettmann K (2009) Chemical variability of the invasive neophytes Polgonum cuspidatum Sieb and Zucc. and Polygonum sachalinensis F. Schmidt ex Maxim. Biochem Syst Ecol 37:24–34
Fan P, Hostettmann K, Lou H (2010) Allelochemicals of the invasive neophyte Polygonum cuspidatum Sieb. & Zucc. (Polygonaceae). Chemoecology 20:223–227
Frantík T, Kovářová M, Koblihová H, Bartůňková K, Nývltová Z, Vosátka M (2013) Production of medically valuable stilbenes and emodin in knotweed. Ind Crop Prod 50:237–243
Gechev TS, Van Breusegem F, Stone JM, Denev I, Laloi C (2006) Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. Bioessays 28(11):1091–1101
Gniazdowska A, Bogatek R (2005) Allelopathic interactions between plants. Multi site action of allelochemicals. Acta Physiol Plant 27(38):395–407
Gonzáles LF, Rojas MC (1999) Role of wall peroxidases in oat growth inhibition by DIMBOA. Phytochemisty 50:931–937
Gotté M, Ghosh R, Bernard S, Nguema-Ona E, Vicré-Gibouin M, Hara-Nishimura I, Driouich A (2015) Methyl jasmonate affects morphology, number and activity od endoplasmic reticulum bodies in Raphanus sativus root cells. Plant Cell Physiol 56(1):61–72
Hara-Nishimura I, Matsushima R (2003) A wound-inducible organelle derived from endoplasmic reticulum: a plant strategy against environmental stresses? Curr Opin Plant Biol 6:583–588
Haugland E, Brandsaeter LO (1996) Experiments on bioassay sensitivity in the study of allelopathy. J Chem Ecol 22(10):1845–1859
Huysmans M, Lema S, Coll NS, Nowack MK (2017) Dying two deaths – programmed cell death regulation in development and disease. Curr Opin Plant Biol 35:37–44
Jambunathan N (2010) Determination and detection of reactive oxygen species (ROS), lipid peroxidation and electrolyte leakage in plants. In: Sunkar R (ed) Plant stress tolerance, methods in molecular biology. Humana Press, Oklahoma, p 295
Kumpf RP, Nowack MK (2015) The root cap: a short story of life and death. J Exp Bot 66(19):5651–5662
Lee SH, Singh AP, Chung GC, Kim YS, Kong B (2002) Chilling root temperature causes rapid ultrastructural changes in cortical cells of cucumber (Cucumis sativus L.) root tips. J Exp Bot 53(378):2225–2237
Li P, Ding L, Zhang L, He J, Huan Z (2019) Weisiensin B inhibits primary and lateral root development by interfering with polar auxin transport in Arabidopsis thaliana. Plant Physiol Biochem 139:738–745
Lowe S, Browne M, Boudjelas S, De Porter M (2000) 100 of the world’s worst invasive alien species. A selection from the global invasive species database. The Invasive Species Specialist Group (ISSG) a specialist group of the Species Survival Commission (SSC) of the World Conservation Union (IUCN), New Zealand, pp 12
Lupini A, Araniti F, Sunseri F, Abenavoli MR (2014) Coumarin interacts with auxin polar transport to modify root system architecture in Arabidopsis thaliana. Plant Growth Regul 74(1):23–31
Matsushima R, Hayashi Y, Kondo M, Shimada T, Nishimura M, Hara-Nishimura I (2002) An endoplasmic reticulum-derived structure that is induced under stress conditions in Arabidopsis. Plant Physiol 130:1807–1814
Miyazono Y, Hirashima S, Ishihara N, Kusukawa J, Nakamura K, Ohta K (2018) Uncoupled mitochondria quickly shorten along their long axis to form indented spheroids, instead of rings, in a fission-independent manner. Sci Rep. https://doi.org/10.1038/s41598-017-18582-6
Moravcová L, Pyšek P, Jarošík V, Zákravský P (2011) Potential phytotoxic and shading effects of invasive Fallopia (Polygonaceae) taxa on the germination of dominant native species. NeoBiota 9:31–47
Murrell C, Gerber E, Krebs C, Parepa M, Schaffner U, Bossdorf O (2011) Invasive knotweed affects native plants through allelopathy. Am J Bot 98(1):38–43
Nakano RT, Yamada K, Bednarek P, Nishimura M, Hara-Nishimura I (2014) ER bodies in plants of the Brassicales order: biogenesis and association with innate immunity. Front Plant Sci 5(73):1–17
Pacifici E, Di Mambro R, Dello Ioio R, Costantino P, Sabatini S (2018) Acidic acid elongation drives cell differentiation in the Arabidopsis root. EMBO J 37:e99134
Piqueras A, Olmos E, Hellín E (1994) Cytological changes related with salt tolerance in embryogenic callus of Citrus limon. Plant Cell Tiss Org 39:13–18
Ponomareva AA, Polygalova OO (2012) Changes in mitochondrial shape in wheat root cells exposed to mitochondrial poisons. Russ J Plant Physiol 59(3):428–433
Potters G, Pasternak TP, Guisez Y, Palme KJ, Jansen MAK (2007) Stress-induced morphogenic responses: growing out of trouble? Trends Plant Sci 12(3):98–105
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Siemens TJ, Blossey B (2007) An evaluation of mechanisms preventing growth and survival of two native species in invasive Bohemian knotweed (Fallopia ×bohemica, Polygonaceae). Am J Bot 94(5):776–783
Šoln K, Likar M, Dolenc Koce J (2021) Effects of rhizome extracts from invasive knotweed species Fallopia japonica and F. ×bohemica on radish seed germination and root growth of seedlings. Allelopath J 52(1):103–118
Soltys D, Rudzinska-Langwald A, Kurek W, Gniazdowska A, Sliwinska E, Bogatek R (2011) Cyanamide mode of action during inhibition of onion (Allium cepa L.) root growth involves disturbances in cell division and cytoskeleton formation. Planta 254:609–621
Strgulc Krajšek S, Dolenc Koce J (2015) Sexual reproduction of knotweed (Fallopia sect. Reynoutria) in Slovenia. Preslia 87(1):17–30
Takahashi L, Aparacida Sert M, Kelmer-Bracht AM, Bracht A, Ishii-Iwamoto EL (1998) Effects of rutin and quercetin on mitochondrial metabolism and on ATP level in germinating tissues of Glycine max. Plant Physiol Biochem 36(7):495–501
Tucker Serniak L (2016) Comparison of the allelopathic effects and uptake of Fallopia japonica phytochemicaly by Raphanus sativus. Weed Res 56:97–101
van Doorn WG, Beers EP, Dangl JL, Franklin-Tong VE, Gallois P, Hara-Nishimura I, Jones AM, Kawai-Yamada M, Lam E, Mundy J, Mur LJA, Petersen M, Smertenko A, Taliansky M, Van Breusegem F, Wolpert T, Woltering E, Zhivotovsky B, Bozhkov PV (2011) Morphological classification of plant cell deaths. Cell Death Differ 18:1241–1246
Yamada K, Goto-Yamada S, Nakazaki A, Kunieda T, Kuwata K, Nagano AJ, Nishimura M, Hara-Nishimura I (2020) Endoplasmic reticulum-derived bodies enable a single-cell chemical defense in Brassicaceae plants. Commun Biol. https://doi.org/10.1038/s42003-019-0739-1
Acknowledgements
The authors are thankful to Dr. Aleš Kladnik for the suggestions of image analysis, Dr. Maks Merela and his co-workers for rhizome grinding, and Dr. Chris Berrie for thorough reading of the manuscript and English editing. We are also deeply grateful to the editor and the anonymous reviewers for valuable comments and suggestions.
Availability of data and material
The authors declare availability of data and material in personal laboratory diaries.
Code availability
Not applicable.
Funding
This study was financially supported by the Slovenian Research Agency (grant no. P1-0212).
Author information
Authors and Affiliations
Contributions
All authors contributed critically to the manuscript and approved the final manuscript. KŠ and NŽ performed the experiment. JDK contributed to data and image analysis.
Corresponding author
Ethics declarations
Ethics approval
No ethical approval was needed.
Consent to participate
The authors declare consent to participate.
Consent for publication
The authors declare consent for publication in Journal of Chemical Ecology.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Handling Editor: Peter Nick
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
Radish seedling stained with diaminobenzidine after 5 days for control seedling (a) and seedling exposed to the aqueous extracts of Fallopia japonica (b) and F. × bohemica (c) rhizomes. Bar: 1 cm. (PNG 450 kb)
ESM 2
Radish seedling stained with diaminobenzidine after 7 days for control seedling (a) and seedling exposed to the aqueous extracts of Fallopia japonica (b) and F. × bohemica (c) rhizomes. Bar: 1 cm. (PNG 8321 kb)
Rights and permissions
About this article
Cite this article
Šoln, K., Žnidaršič, N. & Dolenc Koce, J. Root growth inhibition and ultrastructural changes in radish root tips after treatment with aqueous extracts of Fallopia japonica and F. ×bohemica rhizomes. Protoplasma 259, 343–355 (2022). https://doi.org/10.1007/s00709-021-01668-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-021-01668-4