Abstract
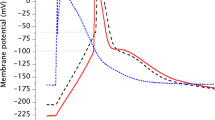
Effect of amino acid l-asparagine on electrical signalling of single Nitellopsis obtusa (Characeaen) cell was investigated using glass-microelectrode technique in current-clamp and voltage-clamp modes. Cell exposure for 30 min to 0.1 mM and 1 mM of asparagine resulted in changes of electrically stimulated action potential (AP) parameters in comparison to standard conditions. Results indicate that asparagine acts in dose-dependent manner: increases AP amplitude by hyperpolarizing AP threshold potential (Eth), prolongs action potential repolarization, increases maximum Cl− efflux amplitude along with the increase of activation and inactivation durations. Presented findings provide new aspects of exogenous amino acids’ effect on plants’ electrical signalling with emphasis on separate single plant cell excitability and AP characteristics.






Similar content being viewed by others
References
Beilby MJ (2007) Action potential in Charophytes. Int Rev Cytol 257:43–82. https://doi.org/10.1016/S0074-7696(07)57002-6
Beilby MJ, Al Khazaaly S (2016) Re-modeling Chara action potential: I. from Thiel model of Ca2+ transient to action potential form. AIMS Biophys 3:431–449. https://doi.org/10.3934/biophy.2016.3.431
Berestovsky GN, Kataev AA (2005) Voltage-gated calcium and Ca2+-activated chloride channels and Ca2+ transients: voltage-clamp studies of perfused and intact cells of Chara. Eur Biophys J 34:973–986. https://doi.org/10.1007/s00249-005-0477-9
Cheng Y, Tian Q, Zhang W-H (2016) Glutamate receptors are involved in mitigating effects of amino acids on seed germination of Arabidopsis thaliana under salt stress. Environ Exp Bot 130:68–78. https://doi.org/10.1016/j.envexpbot.2016.05.004
Chiu JC, Brenner ED, DeSalle R, Nitabach MN, Holmes TC, Coruzzi GM (2002) Phylogenetic and expression analysis of the glutamate-receptor-like gene family in Arabidopsis thaliana. J Biol Chem 276:4338–4343. https://doi.org/10.1093/oxfordjournals.molbev.a004165
Dziubińska H (2003) Ways of signal transmission and physiological role of electrical potentials in plants. Acta Soc Bot Pol 72:309–318. https://doi.org/10.5586/asbp.2003.040
Felle HH, Zimmermann MR (2007) Systemic signalling in barley through action potentials. Planta 226:203–214. https://doi.org/10.1007/s00425-006-0458-y
Forde BG, Roberts MR (2014) Glutamate receptor-like channels in plants: a role as amino acid sensors in plant defence? F1000Prime Rep 6:37. https://doi.org/10.12703/P6-37
Fromm J, Lautner S (2007) Electrical signals and their physiological significance in plants. Plant Cell Environ 30:249–257. https://doi.org/10.1111/j.1365-3040.2006.01614.x
Hedrich R (2012) Ion channels in plants. Physiol Rev 92:1777–1811. https://doi.org/10.1152/physrev.00038.2011
Hepler PK (2005) Calcium: a central regulator of plant growth and development. Plant Cell 17:2142–2155. https://doi.org/10.1105/tpc.105.032508
Homann U, Thiel G (1994) Cl− and K+ channel currents during the action potential in Chara. Simultaneous recording of membrane voltage and patch currents. J Membrane Biol 141,297-309309:297–309. https://doi.org/10.1007/BF00235139
Jones DL, Healey JR, Willett VB, Farrar JF, Hodge A (2005) Dissolved organic nitrogen uptake by plants—an important N uptake pathway? Soil Biol Biochem 37:413–423. https://doi.org/10.1016/j.soilbio.2004.08.008
Katsuhara M, Tazawa M (1992) Calcium-regulated channels and their bearing on physiological activities in Characean cells. Philos Trans R Soc Lond B Biol Sci 338:19–29. https://doi.org/10.1098/rstb.1992.0125
Kisnierienë V, Sakalauskas V (2007) The effect of aluminium on bioelectrical activity of the Nitellopsis obtusa cell membrane after H+-ATPase inhibition. Cent Eur J Biol 2:222. https://doi.org/10.2478/s11535-007-0009-y
Kisnierienė V, Sakalauskas V, Pleskačiauskas A, Yurin V, Rukšėnas O (2009) The combined effect of Cd2+ and ACh on action potentials of Nitellopsis obtusa cells. Cent Eur J Biol 4:343–350. https://doi.org/10.2478/s11535-009-0028-y
Kisnieriene V, Ditchenko TI, Kudryashov AP, Sakalauskas V, Yurin V, Ruksenas O (2012) The effect of acetylcholine on Characeae K+ channels at rest and during action potential generation. Cent Eur J Biol 7:1066–1075. https://doi.org/10.2478/s11535-012-0085-5
Kisnieriene V, Lapeikaite I, Sevriukova O, Ruksenas O (2016) The effects of Ni2+ on electrical signaling of Nitellopsis obtusa cells. J Plant Res 129:551–558. https://doi.org/10.1007/s10265-016-0794-3
Kisnieriene V, Lapeikaite I, Pupkis V (2018) Electrical signalling in Nitellopsis obtusa: potential biomarkers of biologically active compounds. Funct Plant Biol 45:132–142. https://doi.org/10.1071/FP16339
Krol E, Dziubinska H, Stolarz M, Trebacz K (2006) Effects of ion channel inhibitors on cold- and electrically-induced action potentials in Dionaea muscipula. Biol Plant 50 (3): 411-416. https://doi.org/10.1007/s10535-006-0058-5
Krol E, Dziubinska H, Trebacz K, Koselski M, Stolarz M (2007) The influence of glutamic and aminoacetic acids on the excitability of the liverwort Conocephalum conicum. J Plant Physiol 164:773–784. https://doi.org/10.1016/j.jplph.2006.04.015
Lacombe B (2001) The identity of plant glutamate receptors. Science 292:1486b–1487. doi: https://doi.org/10.1126/science.292.5521.1486b
Lam HM, Chiu J, Hsieh MH, Meisel L, Oliveira IC, Shin M, Coruzzi G (1998) Glutamate-receptor genes in plants. Nature 396:125–126. https://doi.org/10.1038/24066
Lunevsky VZ, Zherelova OM, Vostrikov IY, Berestovsky GN (1983) Excitation of Characeae cell membranes as a result of activation of calcium and chloride channels. J Membr Biol 72:43–58. https://doi.org/10.1007/BF01870313
Mousavi SAR, Chauvin A, Pascaud F, Kellenberger S, Farmer EE (2013) Glutamate receptor-like genes mediate leaf-to-leaf wound signalling. Nature 500:422–426. https://doi.org/10.1038/nature12478
Ortiz-Lopez A, Chang H., & Bush D. R. (2000). Amino acid transporters in plants. Biochimica et Biophysica Acta. doi: https://doi.org/10.1016/S0005-2736(00)00144-9
Price MB, Jelesko J, Okumoto S (2012) Glutamate receptor homologs in plants: functions and evolutionary origins. Front Plant Sci 3:235. https://doi.org/10.3389/fpls.2012.00235
Pyatygin SS, Opritov VA, Vodeneev VA (2008) Signaling role of action potential in higher plants. Russ J Plant Physiol 55:285–291. https://doi.org/10.1007/s11183-008-2017-9
Qi Z, Stephens NR, Spalding EP (2006) Calcium entry mediated by GLR3.3, an Arabidopsis glutamate receptor with a broad agonist profile. Plant Physiol 142:963–971. https://doi.org/10.1104/pp.106.088989
Recasens M, Sassetti I, Nourigat A, Sladeczek F, Bockaert J (1987) Characterization of subtypes of excitatory amino acid receptors involved in the stimulation of inositol phosphate synthesis in rat brain synaptoneurosomses. Eur J Pharmacol 141:87–93. https://doi.org/10.1016/0014-2999(87)90413-4
Shepherd VA, Beilby MJ, Al Khazaaly SAS, Shimmen T (2008) Mechano-perception in Chara cells: the influence of salinity and calcium on touch-activated receptor potentials, action potentials and ion transport. Plant Cell Environ 31:1575–1591. https://doi.org/10.1111/j.1365-3040.2008.01866.x
Sivaguru M, Pike S, Gassmann W, Baskin TI (2003) Aluminum rapidly depolymerizes cortical microtubules and depolarizes the plasma membrane: evidence that these responses are mediated by a glutamate receptor. Plant Cell Physiol 44:667–675. https://doi.org/10.1093/pcp/pcg094
Stephens NR, Qi Z, Spalding EP (2008) Glutamate receptor subtypes evidenced by differences in desensitization and dependence on the GLR3.3 and GLR3.4 genes. Plant Physiol 146:529–538. https://doi.org/10.1104/pp.107.108134
Stolarz M, Dziubinska H (2017) Osmotic and salt stresses modulate spontaneous and glutamate-induced action potentials and distinguish between growth and circumnutation in Helianthus annuus seedlings. Front Plant Sci 8:1–13. https://doi.org/10.3389/fpls.2017.01766
Stolarz M, Król E, Dziubinska H (2010) Glutamatergic elements in an excitability and circumnutation mechanism. Plant Signal Behav 5:1108–1111. https://doi.org/10.4161/psb.5.9.12417
Tapken D, Hollmann M (2008) Arabidopsis thaliana glutamate receptor ion channel function demonstrated by ion pore transplantation. J Mol Biol 383:36–48. https://doi.org/10.1016/j.jmb.2008.06.076
Tapken D, Anschütz U, Liu L-H et al (2013) A plant homolog of animal glutamate receptors is an ion channel gated by multiple hydrophobic amino acids. Sci Signal 6:ra47. https://doi.org/10.1126/scisignal.2003762
Thiel G, Homann U, Plieth C (1997) Ion channel activity during the action potential in Chara: new insights with new techniques. J Exp Bot 48:609–622. https://doi.org/10.1093/jxb/48.Special_Issue.609
Tsutsui I, Ohkawa T, Nagai R, Kishimoto U (1987) Role of calcium ion in the excitability and eiectrogenic pump activity of the Chara corallina membrane: I. effects of La 3+, verapamil, EGTA, W-7, and TFP on the action potential. J Membr Biol 108:97–108. https://doi.org/10.1007/BF01871271
Vincill ED, Bieck AM, Spalding EP (2012) Ca(2+) conduction by an amino acid-gated ion channel related to glutamate receptors. Plant Physiol 159:40–46. https://doi.org/10.1104/pp.112.197509
Vincill ED, Clarin AE, Molenda JN, Spalding EP (2013) Interacting glutamate receptor-like proteins in phloem regulate lateral root initiation in Arabidopsis. Plant Cell 25:1304–1313. https://doi.org/10.1105/tpc.113.110668
Wacke M, Thiel G, Hütt M-T (2003) Ca2+ dynamics during membrane excitation of green alga Chara: model simulations and experimental data. J Membr Biol 191:179–192. https://doi.org/10.1007/s00232-002-1054-0
Weiland M, Mancuso S, Baluska F (2016) Signalling via glutamate and GLRs in Arabidopsis thaliana. Funct Plant Biol 43:1–25. https://doi.org/10.1071/FP15109
Williamson RE, Ashley CC (1982) Free Ca2+ and cytoplasmic streaming in the alga Chara. Nature 296:647–651. https://doi.org/10.1038/296647a0
Yoshida R, Mori I, Kamizono N, Shichiri Y, Shimatani T, Miyata F, Honda K, Iwai S (2016) Glutamate functions in stomatal closure in Arabidopsis and fava bean. J Plant Res 129:39–49. https://doi.org/10.1007/s10265-015-0757-0
Zhong B, Sun L, Penny D (2015) The origin of land plants: a phylogenomic perspective. Evol Bioinforma 137. doi: https://doi.org/10.4137/EBO.S29089
Acknowledgements
The authors thank Vidmantas Sakalauskas (Vilnius University) for the technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Lapeikaite, I., Dragunaite, U., Pupkis, V. et al. Asparagine alters action potential parameters in single plant cell. Protoplasma 256, 511–519 (2019). https://doi.org/10.1007/s00709-018-1315-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1315-0