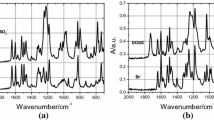
Abstract
A new type of six-armed π-conjugated structure was built up from benzene as the central core and electron-rich triazine rings as the peripheral group. The hexasubstituted product was liquid and did not exhibit any liquid crystal properties while the equimolar mixtures in 1:1 ratio of the hexasubstituted compound and 4-(dodecyloxy)benzoic acid resulted in an organic salt exhibiting a columnar mesophase characterized with a dendritic growing texture. Examination of the relationship between structure and properties and the presence of the number of carbon atoms in the alkyl chain suggested that, the higher increase led to the phase transition at low temperature to form a columnar mesophase as an ionic liquid crystal composed of cations and anions. These are often stabilized by resonance with strongly delocalized charges. The liquid crystalline properties of the organic salt were investigated by differential scanning calorimetry (DSC) and polarizing optical microscopy (POM).
Graphical abstract







Similar content being viewed by others
References
Giacomelli G, Porcheddu A, Luca LD (2004) Curr Org Chem 15:1497
Shahbaz M, Urano S, LeBreton PR, Rossman MA, Hosmane RS, Leonard NJ (1984) J Am Chem Soc 106:2805
Li J, Tao L, Wang Y, Yao Y, Guo Q (2021) Front Chem 18:482
Kroke E, Schwarz M, Horath-Bordon E, Kroll P, Noll B, Norman AD (2002) New J Chem 26:508
Phillips RM (2016) Cancer Chemother Pharmacol 77:441
Ide M, Kato T, Nakata M, Saito K, Yoshida T, Awaya T, Heike T (2015) Brain Dev 37:825
Alazawi SK, Al-Jumaili MHA (2022) J Chem Res 7:1
Yan W, Zhao Y, He J (2018) Mol Med Rep 18:75
Abu-Aisheh NM, Mustafa SM, Mubarak SM, El-Abadelah MM, Voelter W (2012) Lett Org Chem 9:65
Hynes J Jr, Dyckman AJ, Lin S, Wrobleski ST, Wu H, Gillooly KM, Kanner SB, Lonial H, Loo D, McIntyre KW, Pitt S (2008) J Med Chem 10:4
Cascioferro S, Parrino B, Spanò V, Carbone A, Montalbano A, Barraja P, Diana P, Cirrincione G (2017) Eur J Med Chem 15:523
Singla P, Luxami V, Paul K (2015) Eur J Med Chem 102:39
Lim J, Kong SY, Yun YJ (2018) J Nanomater 25:2018
Wu C, Zhang H, Hu M, Shan G, Gao J, Liu J, Zhou X, Yang J (2020) Adv Electron Mater 6:2000253
Zhang H, Zang XF, Hong YP, Chen ZE (2021) Synth Met 280:116882
Li B, Lei Q, Qin T, Zhang X, Zhao D, Wang F, Li W, Zhang Z, Fan L (2021) CrystEngComm 23:8260
Zhang M, Ren S, Guo Q, Shen B (2021) Microporous Mesoporous Mater 326:111395
An ZF, Chen RF, Yin J, Xie GH, Shi HF, Tsuboi T, Huang W (2011) Chem Eur J 17:10871
Akkurt N, Al-Jumaili MH, Eran BB, Ocak H, Torun L (2019) Turk J Chem 43:1436
Akkurt N, Al-Jumaili MH, Ocak H, Cakar F, Torun L (2020) Turk J Chem 44:726
Bakr EA, Al-Jumaili MH (2020) Mol Cryst Liq Cryst 710:40
Sun C, Hudson ZM, Helander MG, Lu ZH, Wang S (2011) Organometallics 30:5552
Banerjee R, Brown DR, Weerapana E (2013) Synlett 24:1599
Steffensen MB, Hollink E, Kuschel F, Bauer M, Simanek EE (2006) Polym Chem 44:3411
Janairo JI, Sakaguchi T, Mine K, Kamada R, Sakaguchi K (2018) Protein Pept Lett 25:4
Al-Jumaili MH, Akkurt N, Torun L (2021) Monatsh Chem 152:551
Kouvetakis J, Grotjahn D, Becker P, Moore S, Dupon R (1994) Chem Mater 6:636
Couderc G, Hulliger J (2010) Chem Soc Rev 39:1545
Whitesides GM, Simanek EE, Mathias JP, Seto CT, Chin D, Mammen M, Gordon DM (1995) Acc Chem Res 28:37
Azhar U, Bashir MS, Babar M, Arif M, Hassan A, Riaz A, Mujahid R, Sagir M, Suri SU, Show PL, Chang JS (2022) Chemosphere 302:134792
Al-Jumaili M, Hamed A, Akkurt N, Torun L (2020) Indones J Chem 20:705
Sonoda M, Inaba A, Itahashi K, Tobe Y (2001) Org Lett 26:2419
Acknowledgements
Authors thank to Professor Belkis Bilgin Eran for valuable discussions on POM studies. The authors acknowledge the funding from the following agents. The Scientific and Technological research Council of Turkey (TUBITAK) with the project no. 114Z722 and The Office of Scientific Research of Yildiz Technical University, project no. FCD-2021-3892.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Al-Jumaili, M.H.A., Ocak, H. & Torun, L. Hydrogen-bonded ionic liquid crystals based on multi-armed structure: synthesis and characterization. Monatsh Chem 153, 939–947 (2022). https://doi.org/10.1007/s00706-022-02969-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-022-02969-x