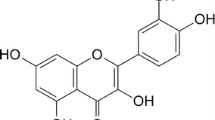
Abstract
This study focuses on the construction of two new inclusion complexes of quercetin with p-sulfonatocalix[4]arene-tetracarboxylic acid and/or p-sulfonatocalix[8]arene-octacarboxylic acid, so that the drug gets soluble in an aqueous media. The structures of the inclusion complexes have been analyzed through FT-IR, NMR and UV–Vis spectroscopy. Evidences indicate that the π–π interaction and hydrogen bonding involve in the inclusion complexes of water-soluble calixarenes (Clx4 and Clx8) with quercetin. The associating constants and stoichiometry of the inclusion complexes constructed between p-sulfonatocalix[n]arene-carboxylic acids (Clx4 and Clx8) and quercetin have been also estimated. In addition, the antioxidant behaviors of the inclusion complexes have been investigated.
Graphical abstract









Similar content being viewed by others
References
Anand David A, Arulmoli R, Parasuraman S (2016) Pharmacogn Rev 10:84
Hollman PCH, Katan MB (1999) Food Chem Toxicol 37:937
Batiha GES, Beshbishy AM, Ikram M, Mulla ZS, El-Hack MEA, Taha AE, Algammal AM, Elewa YHA (2020) Foods 9:374
Kellici TF, Chatziathanasiadou MV, Diamantis D, Chatzikonstantinou AV, Andreadelis L, Christodoulou E, Valsami G, Mavromoustakos T, Tzakos AG (2016) Int J Pharm 511:303
Diamantis DA, Ramesova S, Chatzigiannis CM, Degano I, Gerogianni PS, Karadima KE, Perikleous S, Rekkas D, Gerothanassis IP, Galaris D (2018) Biochim Biophys Acta Gen Subj 1862:1913
Manta K, Papakyriakopoulou P, Chountoulesi M, Diamantis DA, Spaneas D, Vakali V, Naziris N, Chatziathanasiadou MV, Andreadelis I, Moschovou K (2020) Mol Pharm 17:4241
Dabeek WM, Marra MV (2019) Nutrients 11:2288
Srinivas K, King JW, Howard LR, Monrad JK (2010) J Food Eng 100:208
Ilyich T, Lapshina E, Maskevich A, Veiko A, Lavysh A, Palecz B, Stępniak A, Buko V, Zavodnik I (2020) Biophys 65:381
D’Aria F, Serri C, Niccoli M, Mayol L, Quagliariello V, Iaffaioli RV, Biondi M, Giancola C (2017) J Therm Anal Calorim 130:451
Bukhari SB, Memon S, Mahroof-Tahir M, Bhanger M (2009) Spectrochim Acta A 71:1901
Tanigawa S, Fujii M, Hou D-X (2008) Biosci Biotechnol Biochem 72:797
Thangasamy T, Sittadjody S, Lanza-Jacoby S, Wachsberger PR, Limesand KH, Burd R (2007) Nutr Cancer 59:258
Cai X, Fang Z, Dou J, Yu A, Zhai G (2013) Curr Med Chem 20:2572
Troian-Gautier L, Mattiuzzi A, Reinaud O, Lagrost C, Jabin I (2020) Org Biomol Chem 18:3624
Sayin S (2021) Luminescence 36:1716
Sayin S, Yilmaz E, Yilmaz M (2011) Org Biomol Chem 9:4021
Ozyilmaz E, Bayrakci M, Yilmaz M (2016) Bioorg Chem 65:1
Yildiz H, Ozyilmaz E, Bhatti AA, Yilmaz M (2017) Bioprocess Biosyst Eng 40:1189
Ozyilmaz E, Sayin S (2018) Polycycl Aromat Comp 38:272
Ozyilmaz E, Sayin S (2013) Appl Biochem Biotechnol 170:187
Ozyilmaz E, Cetinguney S, Yilmaz M (2019) Int J Biol Macromol 133:1042
Ozyilmaz E, Sayin S (2013) Bioprocess Biosyst Eng 36:1803
Yilmaz M, Karanastasis AA, Chatziathanasiadou MV, Oguz M, Kougioumtzi A, Clemente N, Kellici TF, Zafeiropoulos NE, Avgeropoulos A, Mavromoustakos T (2019) ACS Appl Bio Mater 2:2715
Furer V, Potapova L, Vatsouro I, Kovalev V, Shokova E (2021) In: E3S Web of Conferences. EDP Sciences
Memon S, Yilmaz A, Roundhill MD, Yilmaz M (2004) J Macromol Sci Part A 41:433
Nimse SB, Kim T (2013) Chem Soc Rev 42:366
Oguz M, Bhatti AA, Dogan B, Karakurt S, Durdagi S, Yilmaz M (2020) J Biomol Struct Dyn 38:3801
Pan YC, Hu XY, Guo DS (2021) Angew Chem Int Ed 60:2768
Sayin S, Ozyilmaz E, Oguz M, Yusufoglu R, Yilmaz M (2020) Supramol Chem 32:334
Wang J, Liu D, Guo X, Yan C (2020) J Mol Liq 313:113587
Ahire VK, Malkhede DD (2019) Chem Phys Lett 731:136597
Perret F, Coleman AW (2011) Chem Commun 47:7303
Bhatti AA, Oguz M, Yilmaz M (2018) Appl Surf Sci 434:1217
Ozyilmaz E, Eski F (2020) Bioprocess Biosyst Eng 43:2085
Oguz M, Dogan B, Durdagi S, Bhatti AA, Karakurt S, Yilmaz M (2021) New J Chem 45–18443
Kuntz JID, Gasparro FP, Johnston JMD, Taylor RP (1968) J Am Chem Soc 90:4778
Grant DJW, Mehdizadeh M, Chow AHL, Fairbrother JE (1984) Int J Pharm 18:25
Li H, Chang SL, Chang TR, You Y, Wang XD, Wang LW, Yuan XF, Tan MH, Wang PD, Xu PW (2021) J Mol Liq 334:116070
Laughton MJ, Halliwell B, Evans PJ, Robin J, Hoult S (1989) Biochem Pharmacol 38:2859
Larocca L, Piantelli M, Leone G, Sica S, Teofili L, Panici PB, Scambia G, Mancuso S, Capelli A, Ranelletti F (1990) Br J Haematol 75:489
Orsolic N, Knezevic AH, Sver L, Terzic S, Basic I (2004) J Ethnopharmacol 94:307
Duarte J, Perez-Palencia R, Vargas F, Angeles Ocete M, Perez-Vizcaino F, Zarzuelo A, Tamargo J (2001) Br J Pharmacol 133:117
Cushnie TT, Lamb AJ (2005) Int J Antimicrob Agents 26:343
Gutsche CD, Nam KC (1988) J Am Chem Soc 110:6153
Gutsche CD, Dhawan B, No KH, Muthukrishnan R (1981) J Am Chem Soc 103:3782
Arnaud-Neu F, Collins EM, Deasy M, Ferguson G, Harris SJ, Kaitner B, Lough AJ, McKervey MA, Marques E (1989) J Am Chem Soc 111:8681
Shinkai S, Araki K, Tsubaki T, Arimura T, Manabe O (1987) J Chem Soc Perkin Trans 1:2297
Casnati A, Ting Y, Berti D, Fabbi M, Pochini A, Ungaro R, Sciotto D, Lombardo GG (1993) Tetrahedron 49:9815
Valand NN, Patel MB, Menon SK (2015) RSC Adv 5:8739
Uyar P, Coruh N, İscan M (2014) J Plant Sci 2:339
Acknowledgements
We would like to thank the Scientific and Technological Research Council of Turkey (TUBITAK-Grant Number 119Z131), and the Research Foundation of Selcuk University for financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ozyilmaz, E., Arpaci, P.U., Sayin, S. et al. Inclusion complexes of water-soluble calix[n]arenes with quercetin: preparation, characterization, water solubility, and antioxidant features. Monatsh Chem 153, 201–209 (2022). https://doi.org/10.1007/s00706-021-02885-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-021-02885-6