Abstract
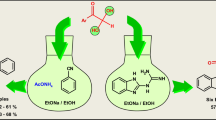
A novel series of (5-aryl-4-oxoimidazolidin-2-ylidene)cyanamides was successfully synthesized via the reaction of arylglyoxal hydrates with cyanoguanidine through benzilic rearrangement mechanism. The reaction was also optimized by changing various reaction factors for endeavor to increase the yield of the target product and reduce the by-product 1-cyano-3-(2-oxo-2-arylethylidene)guanidine formation.
Graphic abstract


Similar content being viewed by others
References
Javahershenas R, Khalafy J, Prager RH (2019) J Chem Rev 1:233
Filimonov VD, Yusubov MS, Chi K-V (1998) Russ Chem Rev 67:725
Riley HA, Gray AR (1935) Org Synth 15:67
Khalafy J, Ezzati M, Madadi P, Marjani AP, Asl HY (2017) S Afr J Chem 70:132
Young RM, Davies-Coleman MT (2011) Tetrahedron Lett 52:4036
Tiecco M, Testaferri L, Tingoli M, Bartoli D (1990) J Org Chem 55:4523
Yin G, Zhou B, Meng X, Wu A, Pan Y (2006) Org Lett 8:2245
Cao Z, Shi D, Qu Y, Tao C, Liu W, Yao G (2013) Molecules 18:15717
Floyd MB, Du MT, Fabio PF, Jacob LA, Johnson BD (1985) J Org Chem 50:5022
Karpov SV, Grigorev AA, Kayukov YS, Karpova IV, Nasakin OE, Tafeenko VA (2016) J Org Chem 81:6402
Lokhande PD, Waghmare SR, Gaikwad H, Hankare PP (2013) Indian J Chem 52B:300
Lokhande PD, Waghmare SR, Gaikwad H, Meshram SA, Hankare PP (2012) Int J Curr Res 4:199
Eftekhari-Sis B, Zirak M, Akbari A (2013) Chem Rev 113:2958
Mishra R, Panday AK, Choudhury LH, Pal J, Subramanian R, Verma A (2017) Eur J Org Chem 2017:2789
Zhang G-N, Zhang M, Du X, Zhu M, Wang J, Shi D, Wang Y (2019) Heterocycles 98:535
Gong J, Peshkov AA, Yu J, Amandykova S, Gimnkhan A, Huang J, Kashtanov S, Pereshivko OP, Peshkov VA (2020) RSC Adv 10:10113
Tan X-F, Zhang F-G, Ma J-A (2020) Beilstein J Org Chem 16:638
Chang X, Yang X, Chen Z, Zhong W (2019) Synlett 30:1431
Heidari M, Anary-Abbasinejad M, Ghazanfari D, Akhgar MR (2018) Lett Org Chem 15:773
Alizadeh-Bami F, Mehrabi H, Ranjbar-Karimi R (2019) J Sulfur Chem 40:469
Shu W-M, Ma J-R, Zheng K-L, Sun H-Y, Wang M, Yang Y, Wu A-X (2014) Tetrahedron 70:9321
Bandyopadhyay D, Smith LC, Garcia DR, Yadav RN, Banik BK (2014) Org Med Chem Lett 4:9
Kolos NN, Chechina NV, Zamigailo LL, Vashchenko EV (2013) Chem Heterocycl Compd 49:872
Madhavachary R, Zarganes-Tzitzikas T, Patil P, Kurpiewska K, Kalinowska-Tłuścik J, Dömling A (2018) ACS Comb Sci 20:192
Bayat M, Nasri S, Notash B (2017) Tetrahedron 73:1522
Khalafy J, Rimaz M, Farajzadeh S, Ezzati M (2013) S Afr J Chem 66:179
Rimaz M, Khalafy J (2010) ARKIVOC 2010:110
Anary-Abbasinejad M, Jaafari M, Talebizadeh M (2019) Org Chem Res 5:139
Jiang B, Ye Q, Fan W, Wang S-L, Tu S-J, Li G (2014) Chem Commun 50:6108
Jiang B, Fan W, Sun M-Y, Ye Q, Wang S-L, Tu S-J, Li G (2014) J Org Chem 79:5258
Kolos NN, Kibkalo BV, Zamigaylo LL, Omelchenko IV, Shishkin OV (2015) Russ Chem Bull 64:864
Khalafy J, Etivand N, Marjani AP, Khalillou N (2019) J Heterocycl Chem 56:1857
Tu XJ, Hao WJ, Ye Q, Wang S-S, Jiang B, Li G, Tu S-J (2014) J Org Chem 79:11110
Khalafy J, Rimaz M, Panahi L, Rabiei H (2011) Bull Korean Chem Soc 32:2428
Modugu NR, Pittala PK (2017) Tetrahedron Lett 58:3859
Javahershenas R, Khalafy J (2018) Asian J Green Chem 2:318
Nandwana NK, Patel Om PS, Srivathsa MR, Kumar A (2019) ACS Omega 4:10140
Khodabakhshi S, Karami B, Eskandari K, Farahi M (2014) Tetrahedron Lett 55:3753
Bardasov IN, Kayukova OV, Kayukov YaS, Ershov OV, Nasakin OE, Tafeenko VA (2009) Russ J Org Chem 45:1325
Nouri A, Marjani AP, Khalafy J (2019) J Heterocycl Chem 56:2912
Poursattar Marjani A, Ebrahimi Saatluo B, Nouri F (2018) Iran J Chem Chem Eng 37:149
Rezaei M, Bayat M, Notash B (2020) J Heterocycl Chem 57:880
Rayudu SV, Karmakar D, Kumar P (2019) Tetrahedron Lett 60:151025
Jiang B, Li Y, Tu M-S, Wang S-L, Tu S-J, Li G (2012) J Org Chem 77:7497
Moustafa AH, Amer AA (2018) Tetrahedron 74:324
Prakash N, Elamaran M, Ingarsal N (2015) Chem Sci Trans 4:94
Moustafa AH, Shestakov AS, Potapov AYu, Romanov PS, Stolpovskaya NV, Shikhaliev KhS, Krugovov DA (2012) Vestnik VSU, Series: Chem Biol Pharm 2:55
Amer AA, Moustafa AH (2017) Mol Divers 21:875
Hussein BRM, Moustafa AH (2019) Synth Commun 49:2401
Moustafa AH, Amer AA (2017) Monatsh Chem 148:2129
Fesenkoa AA, Shutalev AD (2020) Tetrahedron 76:131340
Shutalev AD, Fesenko AA (2011) Tetrahedron 67:6883
Takimoto M, Ebisuno T, Shiba R (1983) Bull Chem Soc Jpn 56:3319
Solovyev PA, Shutalev AD (2009) Chem Heterocycl Compds 45:809
Lambert DM, Masereel B, Gallez B, Geurts M, Scriba GKE (1996) J Pharm Sci 85:1077
Call L (1970) Monatsh Chem 101:344
Birtwell S (1953) J Chem Soc 1953:1725
Grytsai O, Druzhenko T, Demange L, Ronco C, Benhida R (2018) Tetrahedron Lett 59:164
Soliman AM, Mohamed ShK, El-Remaily MAAA, Abdel-Ghany H (2014) J Heterocycl Chem 51:1322
Dolzhenko AV, Foo MC, Tan BJ, Dolzhenko AV, Chiu GNC, Chui WK (2009) Heterocycles 78:1761
Kosasayama A, Konno T, Higashi k, Ishikawa F (1979) Chem Pharm Bull 27:841
Kreutzberger A, Sellheim M (1985) J Heterocycl Chem 22:721
Moustafa AH, Ahmed WW, Khodairy A (2017) J Heterocycl Chem 54:3490
Albayati MR, Mohamed MFA, Moustafa AH (2020) Synth Commun 50:1217
Shestakov AS, Moustafa AH, Bushmarinov IS, Goloveshkin AS, Shapovalov AV, Shikhaliev KhS, Prezent MA, Sidorenko OE (2017) J Heterocycl Chem 54:551
Paul S, Gupta M, Gupta R, Loupy A (2002) Synthesis 1:75
Joshi KC, Pathak VN, Goyal MK (1981) J Heterocycl Chem 18:1651
Putnam TJ, Merrit HH (1937) Science 85:525
Krall RL, Penry JK, White BG, Kupferberg HJ, Swinyard EA (1978) Epilepsia 19:409
Bac P, Maurois P, Dupont C, Pages N, Stables JP, Gressens P, Evrard P, Vamecq J (1998) J Neurosci 18:4363
Bardovskyi R, Grytsai O, Ronco C, Benhid R (2020) New J Chem 44:8171
Cheung S-T, Fonda ML (1979) Biochem Biophys Res Commun 90:940
Dunnavant WR, James FL (1956) J Am Chem Soc 78:2740
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moustafa, A.H., Hussein, B.R.M. Reaction of arylglyoxal hydrate derivatives with cyanoguanidine under benzilic rearrangement effect. Monatsh Chem 152, 1285–1290 (2021). https://doi.org/10.1007/s00706-021-02838-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-021-02838-z