Abstract
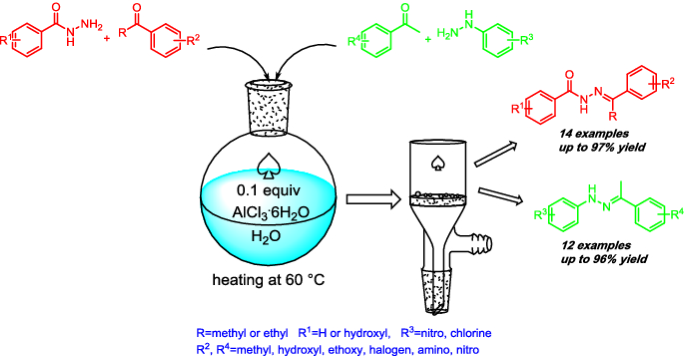
Schiff-bases have important applications in the field of analysis, biomedicine, as well as material sciences. Hydrazones and acylhydrazones are two representative types of Schiff-bases. In this study, a green synthesis of aromatic hydrazones and acylhydrazones via Schiff-base reaction of aryl ketones and aromatic acylhydrazines/hydrazines had been reported. In the synthesis, water was used as solvent and AlCl3·6H2O was used as catalyst. The reaction is simple, highly efficient, and eco-friendly.
Graphic abstract
Similar content being viewed by others
References
Horváth IT, Anastas PT (2007) Chem Rev 107:2167
Schiff H (1864) Justus Liebigs Ann Chem 131:118
Greig LM, Philp D (2001) Chem Soc Rev 30:287
Xin Y, Yuan JY (2012) Polym Chem 3:3045
Mittal SK, Rana S, Kaur N, Banks CE (2018) Analyst 143:2851
Kachwal V, Krishna ISV, Fageria L, Chaudhary J, Roy RK, Chowdhury R, Laskar IR (2018) Analyst 143:3741
Sohma Y, Kent SBH (2009) J Am Chem Soc 131:16313
Miranda LP, Shao H, Williams J, Chen S, Kong T, Garcia R, Chinn Y, Fraud N, Dwyer B, Ye J, Wilken J, Low DE, Cagle EN, Carnevali M, Lee A, Song D, Kung A, Bradburne JA, Paliard X, Kochendoerfer GG (2007) J Am Chem Soc 129:13153
Zheng Y, Ren J, Wu Y, Meng X, Zhao Y, Wu C (2017) Bioconjugate Chem 28:2620
Xue S, Ungur L, Ungur Y, Tang J, Chibotaru LF (2014) Inorg Chem 53:12658
Lv K, Zhang L, Liu M (2014) Langmuir 30:9295
Vinod PS, Archana S, Shweta S (2008) J Appl Polym Sci 110:1336
Wu PL, Peng SY, Magrath J (1995) Synthesis 435–438
Enthaler S, Weidauer M, Irran E, Epping JD, Kretschmer R, Someya CI (2013) J Organomet Chem 745–746:262
Lu A, Huang D, Wang KH, Su Y, Ma J, Xu Y, Hu Y (2016) Synthesis 48:293
Rathelot P, Azas N, El-Kashef H, Delmas F, Di Giorgio C, Timon-David P, Maldonado J, Vanelle P (2002) Eur J Med Chem 37:671
Xie W, Jin Y, Wang PG (1999) ChemTech 29:23
Fringuelli F, Pizzo F, Vaccaro L (2001) Tetrahedron Lett 42:1131
Singh VP, Singh A, Singh SJ (2008) Appl Polym Sci 110:1336
Ghosh T, Mondal B, Ghosh T, Sutradhar M, Mukherjee G, Drew MGB (2007) Inorg Chim Acta 360:1753
Patel MN, Patel CR, Joshi HN (2013) Appl Biochem Biotech 169:1329
Sreeja PB, Kurup MR, Prathapachandra Kurup KA, Jasmin C (2004) Polyhedron 23:575
Soujanya M, Rajitha G (2017) Pharm Chem 9:10
Asiri AM, Bahajaj AA, Ismail IMI, Fatani NA (2006) Dyes Pigments 71:103
Shetty SC, Bhagat VC (2008) Asian J Chem 20:5037
Naim MJ, Alam MJ, Nawaz F, Naidu VGM, Aaghaz S, Sahu M, Siddiqui N, Alam O (2017) Bioorganic Chem 73:24
Baddeley TC, Franca LS, Howie RA, de Lima GM, Skakle JMS, Dias de Souza J, Wardell JL, Wardell SM (2009) Bioorganic Chem 24:213–219
Dennler EB, Frasca AR (1966) Tetrahedron 22:3131
Stevens MFG (1975) J Chem Soc Perkin Trans 2:1555
Jarikote DV, Deshmukh RR, Rajagopal R, Lahoti RJ, Daniel T, Srinivasan KV (2003) Ultrason Sonochem 10:45
Acknowledgements
We thank for financial support given by Middle and Youth Teachers Scientific and Technological Talents Developing Fund (No. ZQ 2018-20), Natural Science Foundation of Shanghai (No. 15ZR1440400), Collaborative Innovation Fund (No. XTCX2016-14), National Youth Fund Project of China (No. 21602135), Shanghai Municipal Education Commission (Plateau Discipline Construction Program).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, Z.X., Li, T., Cheng, L.P. et al. AlCl3·6H2O-catalyzed Schiff-base reaction between aryl ketones and aromatic acylhydrazines/hydrazines in water. Monatsh Chem 150, 1833–1839 (2019). https://doi.org/10.1007/s00706-019-02488-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-019-02488-2