Abstract
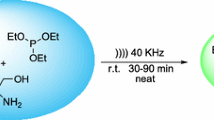
A simple and efficient procedure for the synthesis of substituted imidazo[1,2-a]pyridines under ultrasound irradiation has been developed. The reactions were carried out using ionic liquids as catalyst. The reaction procedure demonstrated a broad substrate scope for both acetophenones and 2-aminopyridines, and provided convenient access to a wide variety of imidazo[1,2-a]pyridines. The present method offers several advantages compared to traditional heating methods such as higher yields, shorter reaction times, milder reaction conditions, and easier work-up procedure.
Graphical abstract

Similar content being viewed by others
References
Gudmundsson KS, Williams JD, Drach JC, Townsend LB (2003) J Med Chem 46:1449
Gueiffier A, Mavel S, Lhassani M, Elhakmaoui A, Snoeck R, Andrei G, Chavignon O, Teulade JC, Witvrouw M, Balzarini J, De Clercq E, Chapat JP (1998) J Med Chem 41:5108
Lhassani M, Chavignon O, Chezal JM, Teulade JC, Chapat JP, Snoeck R, Andrei G, Balzarini J, De Clercq E, Gueiffier A (1999) Eur J Med Chem 34:271
Chandra MD, Reddy DR, Nageswara RS, Adimurthy S (2013) Adv Synth Catal 355:2217
Starrett JE, Montzka TA, Crosswell AR, Cavanagh RL (1989) J Med Chem 32:2204
Hieke M, Rödl CB, Wisniewska JM, la Buscató E, Stark H, Schubert-Zsilavecz M, Steinhilber D, Hofmann B, Proschak E (2012) Bioorg Med Chem Lett 22:1969
Hodgkiss RJ, Middleton RW, Parrick J, Rami HK, Wardman P, Wilson GD (1992) J Med Chem 35:1920
Donohoe TJ, Kabeshov MA, Rathi AH, Smith IED (2012) Org Biomol Chem 10:1093
Zeng J, Tan YJ, Leow ML, Liu X-W (2012) Org Lett 14:4386
Cao H, Liu X, Liao J, Huang J, Qiu H, Chen Q, Chen Y (2014) J Org Chem 79:11209
Bagdi AK, Rahman M, Santra S, Majee A, Hajra A (2013) Adv Synth Catal 355:1741
Meng X, Wang Y, Yu C, Zhao P (2014) RSC Adv 4:27301
Monir K, Bagdi AK, Ghosh M, Hajra A (2014) Org Lett 16:4630
Wang H, Wang Y, Liang D, Liu L, Zhang J, Zhu Q (2011) Angew Chem Int Ed 50:5678
Chioua M, Soriano E, Infantes L, Jimeno ML, Marco-Contelles J, Samadi A (2013) Eur J Org Chem 2013:35
Patil SV, Gaikwad ND, Bobade VD (2016) Arab J Chem 9:S1649
Karade NN, Kondre JM, Gampawar SV, Shinde SV (2009) Synth Commun 39:2279
Stasyuk AJ, Banasiewicz M, Cyrański MK, Gryko DT (2012) J Org Chem 77:5552
Kielesinski L, Tasior M, Gryko DT (2015) Org Chem Front 2:21
Ranu BC, Jana R (2006) Eur J Org Chem 16:3767
Wu X, Han X, Zhou L, Li A (2012) Indian J Chem 51A:791
Chakraborti AK, Roy SR, Kumar D, Chopra P (2008) Green Chem 10:1111
Wasserscheid P, Eichmann M (2001) Catal Today 66:309
Toma Š, Šebesta R (2012) Ionic liquids as (co-) solvents and co-catalysts for organocatalytic reactions. In: Ionic liquids in biotransformations and organocatalysis. Wiley, Hoboken, p 331
Choi J, Benedetti TM, Jalili R, Walker A, Wallace GG, Officer DL (2016) Chem Eur J 22:14158
Palimkar SS, More VS, Srinivasan KV (2008) Ultrason Sonochem 15:853
Jin TS, Liu LB, Zhao Y, Li TS (2005) J Chem Res 3:162
Mosslemin MH, Nateghi MR (2010) Ultrason Sonochem 17:162
Nikpassand M, Mamaghani M, Shirini F, Tabatabaeian K (2010) Ultrason Sonochem 17:301
Zou Y, Wu H, Hu Y, Liu H, Zhao X, Ji H, Shi D (2011) Ultrason Sonochem 18:708
Santra S, Bagdi AK, Majee A, Hajra A (2013) Adv Synth Catal 355:1065
Yu J, Jin Y, Zhang H, Yang X, Fu H (2013) Chem Eur J 19:16804
Zhu DJ, Chen JX, Liu MC, Ding JC, Wu HY (2009) J Braz Chem Soc 20:482
Adib M, Mohamadi A, Sheikhi E, Ansari S, Bijanzadeh HR (2010) Synlett 11:1606
Xie YY, Chen ZC, Zheng QG (2002) Synthesis 11:1505
Ueno M, Togo H (2004) Synthesis 16:2673
Samanta S, Jana S, Mondal S, Monir K, Chandra SK, Hajra A (2016) Org Biomol Chem 14:5073
Kour D, Khajuria R, Kapoor KK (2016) Tetrahedron Lett 57:4464
Mutai T, Tomoda H, Ohkawa T, Yabe Y, Araki K (2008) Angew Chem Int Ed 47:9522
Sundberg RJ, Dahlhausen DJ, Manikumar G, Mavunkel B, Biswas A, Srinivasan V, King F, Waid P (1988) J Heterocycl Chem 2:129
Wang FJ, Xu H, Xin M, Zhang Z (2016) Mol Divers 20:659
Habibi YA, Pourbasheer E, Danandeh JM (2008) Bull Korean Chem Soc 29:833
Acknowledgements
The first author would like to thank the Science Achievement Scholarship of Thailand for financial support. In addition, we wish to thank the Department of Chemistry, Faculty of Science, Chiang Mai University, Thailand, for facilities supporting this research.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paengphua, P., Chancharunee, S. Facile synthesis of imidazo[1,2-a]pyridines promoted by room-temperature ionic liquids under ultrasound irradiation. Monatsh Chem 149, 1835–1840 (2018). https://doi.org/10.1007/s00706-018-2238-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-018-2238-3