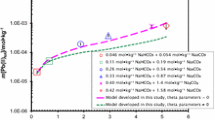
Abstract
The solubility behavior of hexavalent neptunium (Np) was systematically investigated as function of [CaCl2] = 0.25–4.5 mol dm−3 [0.252–5.26 mol (kg H2O)−1] and pHm 8–12 (pHm = − log{m(H+)/mol (kg H2O)−1}) under oxidizing conditions adjusted by hypochlorite. As solubility limiting Np(VI) solid phase, hitherto unknown, non-stoichiometric calcium neptunates, Ca x NpO3+x (s,hyd), were identified by applying a wide range of analytical techniques including quantitative chemical analysis, powder XRD, Np L3-edge XANES, and SEM–EDX. The Ca:Np ratio in the equilibrium solid phase increased systematically with the pHm values in the batch solubility samples, and ranged between 0.60:1 and 1.66:1. For pHm > 10.5, the solubility of the calcium neptunates is around log{[Np]/mol (kg H2O)−1} ≈ − 6.1 ± 0.4 and does not show a strong dependence on [CaCl2]. For pHm < 10.5, log [Np] ranges from − 6.6 in 0.25 mol dm−3 CaCl2 to − 3.6 in 4.5 mol dm−3 CaCl2, and increases systematically with [CaCl2]. Based on the solubility data, the principle solid–liquid equilibrium reactions were qualitatively evaluated. The results for Np(VI) obtained in the present work were found to be different from the behavior of U(VI) in alkaline CaCl2 solutions, where the well-defined crystalline CaU2O7·3H2O(cr) phase controls the U(VI) solubility over a wide range of pHm and [CaCl2] conditions.
Graphical abstract











Similar content being viewed by others
References
Guillaumont R, Fanghänel Th, Fuger J, Grenthe I, Neck V, Palmer DA, Rand MH (2003) Chemical thermodynamics vol. 5, update on the chemical thermodynamics of uranium, neptunium, plutonium, americium and technetium. Elsevier, North-Holland
Morss LR, Edelstein NM, Fuger J (2008) The chemistry of the actinide and transactinide elements. Springer, Dordrecht
Neck V, Altmaier M, Fanghänel T (2007) C R Chim (France) 10:959
Gaona X, Fellhauer D, Altmaier M (2013) Pure Appl Chem 85:2027
Cassol A, Magon L, Tomat G, Portanova R (1972) Inorg Chem 11:515
Kato Y, Kimura T, Yoshida Z, Nitani N (1996) Radiochim Acta 74:21
Gaona X, Tits J, Dardenne K, Liu X, Denecke MA, Wieland E, Altmaier M (2012) Radiochim Acta 100:759
Bagnall KW, Laidler JB (1964) J Chem Soc 2693. https://doi.org/10.1039/jr9640002693
Saito T, Wang J, Kitazawa T, Takahashi M, Takeda M, Nakada M, Nakamoto T, Masaki NM, Yamashita T, Saeki M (1999) Radioanal Nucl Chem 239:319
Keller C, Koch L, Walter KH (1965) J Inorg Nucl Chem 27:1205
Williams CW, Blaudeau J-P, Sullivan JC, Antonio MR, Bursten B, Soderholm L (2001) J Am Chem Soc 123:4346
Bolvin H, Wahlgren U, Moll H, Reich T, Geipel G, Fanghänel T, Grenthe I (2001) J Phys Chem A 105:11441
Gaona X, Wieland E, Tits J, Scheinost A, Dähn R (2013) Appl Geochem 28:109
Clark DL, Conradson SD, Donohoe RJ, Gordon PL, Keogh DW, Palmer PD, Scott BL, Tait CD (2013) Inorg Chem 52:3547
Neck V, Altmaier M, Rabung T, Lützenkirchen J, Fanghänel T (2009) Pure Appl Chem 81:1555
Altmaier M, Neck V, Fanghänel T (2008) Radiochim Acta 96:541
Fellhauer D, Neck V, Altmaier M, Lützenkirchen J, Fanghänel T (2010) Radiochim Acta 98:541
Yalcintas E, Gaona X, Altmaier M, Dardenne K, Polly R, Geckeis H (2016) Dalton Trans 45:8916
Fellhauer D, Rothe J, Altmaier M, Neck V, Runke J, Wiss T, Fanghänel T (2016) Radiochim Acta 104:355
Fellhauer D, Altmaier M, Gaona X, Lützenkirchen J, Fanghänel T (2016) Radiochim Acta 104:381
Altmaier M, Neck V, Müller R, Fanghänel T (2005) Abstract No. A1-3, 10th international conference on chemistry and migration behaviour of actinides and fission products in the geosphere. Avignon, France
Vochten R, van Haverbeke L (1990) Mineral Petrol 43:65
Sandino MCA, Grambow B (1994) Radiochim Acta 66/67:37
Rai D, Felmy AR, Hess NJ, LeGore VL, McCready DE (2002) Radiochim Acta 90:495
Diaz Arocas P, Grambow B (1998) Geochim Cosmochim Acta 62:245
Allen PG, Shuh DK, Bucher JJ, Edelstein NM, Palmer CEA, Silva RJ, Nguyen SN, Marquez LN, Hudson EA (1996) Radiochim Acta 75:47
Altmaier M, Yalcintas E, Gaona X, Neck V, Müller R, Schlieker M, Fanghänel T (2017) J Chem Therm 114:2
Pashalidis I, Kim JI, Lierse C, Sullivan JC (1993) Radiochim Acta 60:99
Hagan PG, Cleveland JM (1966) J Inorg Nucl Chem 28:2905
Runde W, Neu MP, Conradson SD, Clark DL, Palmer PD, Reilly SD, Scott BL, Tait CD (1996) Mat Res Soc Symp Proc 465:693
Hartmann T, Paviet-Hartmann P, Wetteland C, Lu N (2003) Rad Phys Chem 66:335
Nguyen-Trung C (2002) Ph.D. thesis, University of Nancy
Clark DL, Conradson SD, Neu MP, Palmer PD, Runde W, Tait CD (1997) J Am Chem Soc 119:5259
Lippmann F (1977) Neues Jahrb Mineral. Abh 130:243
Gamsjäger H, Königsberger E, Preis W (2000) Aquat Geochem 6:119
Reilly SD, Neu MP (2006) Inorg Chem 45:1839
Rothe J, Butorin S, Dardenne K, Denecke MA, Kienzler B, Löble M, Metz V, Seibert A, Steppert M, Vitova T, Walther C, Geckeis H (2012) Rev Sci Instrum 83:1
Acknowledgements
Technical support by V. Petrov (Moscow State University), S. Moisei-Rabung and P. Leske (KIT—Institute for Nuclear Waste Disposal) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fellhauer, D., Gaona, X., Rothe, J. et al. Neptunium(VI) solubility in alkaline CaCl2 solutions: evidence for the formation of calcium neptunates Ca x NpO3+x (s,hyd). Monatsh Chem 149, 237–252 (2018). https://doi.org/10.1007/s00706-017-2116-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-017-2116-4