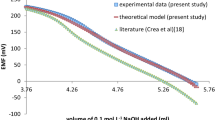
Abstract
Thermodynamic equilibria in urine at 37 °C showed that the inorganic urinary constituents, citrate and oxalate are present as 76 different species. Complexes formed by uric acid and various organic compounds, such as creatinine, proteins, hormones and metabolites, for which the relevant thermodynamic data are not available in the library of the computer program MINTEQA2 Version 3 were not considered. The actual thermodynamic supersaturation of urine with respect to calcium oxalate monohydrate and dihydrate was calculated. The effects of varying composition of urine at the same pH and the violation of the condition of electroneutrality on the calculated supersaturation were shown. The supersaturation of urine should never be computed from incomplete data of urine composition. Incomplete urine composition should always be supplemented with data for the other urinary components. Guidelines for extension of urine composition were suggested. A requisite condition for formation of solid calcium oxalate in urine is the presence of active heterogeneous nuclei. Supersaturation of urine with respect to calcium oxalates per se cannot be regarded as a diagnostic measure of the risk of calcium oxalate stone formation.
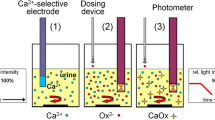
Graphical abstract

COM formation through heterogenous nucleation on hydroxyapatite





Similar content being viewed by others
References
Finlayson B (1982) Pathologic mineralization, nucleation, growth, and retention. In: Nancollas GH (ed) Biological Mineralization and demineralization. Report of the Dahlem workshop on biological mineralization and demineralization. Springer, Berlin, 18–23 Oct 1981, p 271
McPherson RA, Ben-Ezra J (2011) Basic examination of urine. In: McPherson RA, Pincus MR (eds) Henry's clinical diagnosis and management by laboratory methods, Chap 28, 22nd edn. Elsevier Saunders, Philadelphia, PA
Gerber GS, Brendler CB (2011) Evaluation of the urologic patient: history, physical examination, and urinalysis. In: Wein AJ, Kavoussi LR, Novick AC et al (eds) Campbell-Walsh urology, Chap 3, 10th edn. Elsevier Saunders, Philadelphia, PA
Türk C, Knoll T, Petrik A, Sarica K, Skolarikos A, Straub M, Seitz C (2016) Guidelines on urolithiasis. Urolithiasis. European Association of Urology. ISBN 978-90-79754-98-4
Robertson WG (2015) Urolithiasis 43(Suppl 1):S93
WebMD (2015) Calcium (Ca) in urine. http://www.webmd.com/a-to-z-guides/calcium-ca-in-urine#1. Accessed 30 Nov 2017
WebMD (2015) Phosphate in urine. http://www.webmd.com/a-to-z-guides/phosphate-in-urine#1. Accessed 30 Nov 2017
Fischbach FT, Dunning MB (2009) III. Manual of laboratory and diagnostic tests, 8th edn. Lippincott, Williams and Wilkins, Philadelphia
Lundquist P, Märtensson J, Sörbo B, Ohman S (1980) Clin Chem 26:1178
Denis W, Minot AS (1918) J Biol Chem 34:569
Robertson WG, Peacock M, Nordin BEC (1968) Clin Sci 34:579
Lemann J Jr, Pleuss JA, Worcester EM, Hornick L, Schrab D, Hoffmann RG (1996) Kidney Int 49:200
Caudarella R, Vescini F (2009) Arch Ital Urol Androl 81:182
WebMD (2015) Uric acid in urine. http://www.webmd.com/a-to-z-guides/uric-acid-in-urine#1. Accessed 30 Nov 2017
Fogazzi GB, Garigali G (2015) Urinalysis. In: Johnson RJ, Feehally J, Floege J (eds) Comprehensive clinical nephrology, Chap 4, 5th edn. Elsevier Saunders, Philadelphia, PA
Basavaraj DR, Biyani CS, Browning AJ, Cartledge JJ (2007) Eau-ebu Update Ser 5:126
Nielsen AE (1964) Kinetics of precipitation. Pergamon Press Ltd., Oxford
Cussler EL (1997) Diffusion: mass transfer in fluid systems, 2nd edn. Cambridge University Press, New York
Bennema P, Söhnel O (1990) J Cryst Growth 102:547
Söhnel O, Grases F (2017) World J Clinic Urol 6:40
Rodgers AL (2014) BJU Int 114:104
Lewis BN, Randall M (1961) Thermodynamic properties of ideal gases calculated from spectroscopic and other molecular data. In: Pitzer KS, Brewer L (eds) Thermodynamics. McGraw-Hill, New York, p 311
Streit J, Tran-Ho L-C, Königsberger E (1998) Monatsh Chem 129:1225
Allison JD, Brown DS, Novo-Gradac KJ (1991) MINTEQA/PRODEFA2, a geochemical assessment model for environmental system: user’s manual, version 3.0. US Environmental Protection Agency, EPA/6003-91/021
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Söhnel, O., Loučka, T. & Grases, F. Speciation and supersaturation of urine. Monatsh Chem 149, 333–339 (2018). https://doi.org/10.1007/s00706-017-2115-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-017-2115-5