Abstract
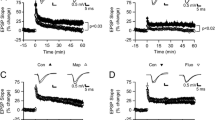
Encoding of episodic memory requires long-term potentiation (LTP) of neurotransmission at excitatory synapses of the hippocampal circuitry. Previous data obtained with the application of exogenous 5-hydroxytryptamine (5-HT) in hippocampal slices indicate that 5-HT blocks LTP, which contrasts with the facilitatory effect of selective serotonin reuptake inhibitors (SSRIs) on learning and memory observed in vivo. Here, we investigated the effects of endogenous 5-HT, released from terminals by the monoamine releaser 3,4-methylenedioxymethamphetamine (MDMA), on LTP of field EPSPs induced by theta-burst stimulation and recorded at CA3/CA1 synapses of rat hippocampal slices. LTP was greater in the presence of MDMA (10 µM; 45.76 ± 15.75 %; n = 28) than in controls (31.26 ± 11.03; n = 21; p < 0.01). This facilitatory effect on LTP persisted when the entry of MDMA in noradrenergic terminals was prevented by the selective noradrenaline reuptake inhibitor nisoxetine (44.90 ± 14.07 %; n = 27 vs. 34.49 ± 12.94 %; n = 20 in controls; p < 0.05). In both conditions, the facilitation of LTP was abolished by the SSRI citalopram that prevented the entry of MDMA in 5-HT terminals and the subsequent 5-HT release. These data show that, unlike exogenous 5-HT application, release of endogenous 5-HT does not impair cellular mechanisms responsible for induction of LTP, indicating that 5-HT is not detrimental to learning and memory. Moreover, facilitation of LTP by endogenous 5-HT may underlie the in vivo positive effects of augmented 5-HT tone on cognitive performance.



Similar content being viewed by others
References
Abad S, Fole A, Del Olmo N, Pubill D, Pallàs M, Junyent F, Camarasa J, Camins A, Escubedo E (2014) MDMA enhances hippocampal-dependent learning and memory under restrictive conditions, and modifies hippocampal spine density. Psychopharmacology (Berl). Psychopharmacology 231(5):863–874
Anderson IM, Juhasz G, Thomas E, Downey D, McKie S, Deakin JF, Elliott R (2011) The effect of acute citalopram on face emotion processing in remitted depression: a pharmacoMRI study. Eur Neuropsychopharmacol 21(1):140–148
Andrade R, Beck SG (2010) Cellular effects of serotonin in the CNS. In: Muller CP, Jacobs B (eds) Handbook of the behavioral neurobiology of serotonin. Academic Press, London, pp 219–231
Barnes NM, Sharp T (1999) A review of central 5-HT receptors and their function. Neuropharmacology 38(8):1083–1152
Bliss TVP, Goddard GV, Riives M (1983) Reduction of long-term potentiation in the dentate gyrus of the rat following selective depletion of monoamines. J Physiol (London) 334:475–491
Bliss TVP, Collingridge GL, Morris R (2007) Synaptic plasticity in the hippocampus. In: Andersen P, Morris R, Amaral D, Bliss T, O’Keefe J (eds) The hippocampus book. Oxford University Press Inc, New York, pp 343–474
Corradetti R, Ballerini L, Pugliese AM, Pepeu G (1992) Serotonin blocks the long-term potentiation induced by primed burst stimulation in the CA1 region of rat hippocampal slices. Neuroscience 46:511–518
Del-Ben CM, Ferreira CA, Alves-Neto WC, Graeff FG (2008) Serotonergic modulation of face-emotion recognition. Braz J Med Biol Res 41:263–269
Elliott R, Zahn R, Deakin JF, Anderson IM (2011) Affective cognition and its disruption in mood disorders. Neuropsychopharmacology 36(1):153–182
Fitzgerald JL, Reid JJ (1993) Interactions of methylenedioxymethamphetamine with monoamine transmitter release mechanisms in rat brain slices. Naunyn Schmiedebergs Arch Pharmacol 347:313–323
Garrett AS, Maddock RJ (2006) Separating subjective emotion from the perception of emotion-inducing stimuli: an fMRI study. Neuroimage 33:263–274
Harmer CJ, Bhagwagar Z, Cowen PJ, Goodwin GM (2002) Acute administration of citalopram facilitates memory consolidation in healthy volunteers. Psychopharmacology 163:106–110
Harmer CJ, Bhagwagar Z, Perrett DI, Völlm BA, Cowen PJ, Goodwin GM (2003) Acute SSRI administration affects the processing of social cues in healthy volunteers. Neuropsychopharmacology 28(1):148–152
Hayward G, Goodwin GM, Cowen PJ, Harmer CJ (2005) Low-dose tryptophan depletion in recovered depressed patients induces changes in cognitive processing without depressive symptoms. Biol Psychiatry 57(5):517–524
Hensler JG (2006) Serotonergic modulation of the limbic system. Neurosci Biobehav Rev 30(2):203–214
Hyttel J, Larsen JJ (1985) Neurochemical profile of Lu 19-005, a potent inhibitor of uptake of dopamine, noradrenaline, and serotonin. J Neurochem 44:1615–1622
Izquierdo I, Medina JH (1995) Correlation between the pharmacology of long-term potentiation and the pharmacology of memory. Neurobiol Learn Mem 63(1):19–32
Jahnsen H (1980) The action of 5-hydroxytryptamine on neuronal membranes and synaptic transmission in area CA1 of the hippocampus in vitro. Brain Res 197:83–94
Jensen O, Lisman JE (2005) Hippocampal sequence-encoding driven by a cortical multi-item working memory buffer. Trends Neurosci 28(2):67–72
Johnston D, Wu SMS (1995) Extracellular field recordings. In: Johnston D, Wu SMS (eds) Foundations of cellular neurophysiology. MIT Press, Cambridge, pp 423–439
Klancnik JM, Phillips AG (1991) Modulation of synaptic plasticity in the dentate gyrus of the rat by electrical stimulation of the median raphe nucleus. Brain Res 557(1–2):236–240
Kuypers KP, Ramaekers JG (2007) Acute dose of MDMA (75 mg) impairs spatial memory for location but leaves contextual processing of visuospatial information unaffected. Psychopharmacology 189(4):557–563
Larson J, Lynch G (1986) Induction of synaptic potentiation in hippocampus by patterned stimulation involves two events. Science 232:985–988
Lesch KP, Waider J (2012) Serotonin in the modulation of neural plasticity and networks: implications for neurodevelopmental disorders. Neuron 76(1):175–191
McKie S, Del-Ben C, Elliott R, Williams S, del Vai N, Anderson I, Deakin JF (2005) Neuronal effects of acute citalopram detected by pharmacoMRI. Psychopharmacology 180(4):680–686
Mlinar B, Corradetti R (2003) Endogenous 5-HT, released by MDMA through serotonin transporter- and secretory vesicle-dependent mechanisms, reduces hippocampal excitatory synaptic transmission by preferential activation of 5-HT1B receptors located on CA1 pyramidal neurons. Eur J Neurosci 18:1559–1571
Mlinar B, Mascalchi S, Morini R, Giachi F, Corradetti R (2008) MDMA induces EPSP-Spike potentiation in rat ventral hippocampus in vitro via serotonin and noradrenaline release and coactivation of 5-HT4 and beta1 receptors. Neuropsychopharmacology 33:1464–1475
Morini R, Mlinar B, Baccini G, Corradetti R (2011) Enhanced hippocampal long-term potentiation following repeated MDMA treatment in Dark-Agouti rats. Eur Neuropsychopharmacol 21:80–91
Moyano S, Frechilla D, Del Río J (2004) NMDA receptor subunit and CaMKII changes in rat hippocampus induced by acute MDMA treatment: a mechanism for learning impairment. Psychopharmacology 173(3–4):337–345
Otto T, Eichenbaum H, Wiener SI, Wible CG (1991) Learning-related patterns of CA1 spike trains parallel stimulation parameters optimal for inducing hippocampal long-term potentiation. Hippocampus 1:181–192
Pugliese AM, Ballerini L, Passani MB, Corradetti R (1994) EPSP-Spike potentiation during primed burst-induced long-term potentiation in the CA1 region of rat hippocampal slices. Neuroscience 62:1021–1032
Pugliese AM, Passani MB, Corradetti R (1998) Effect of the selective 5-HT1A receptor antagonist WAY 100635 on the inhibition of e.p.s.ps produced by 5-HT in the CA1 region of rat hippocampal slices. Br J Pharmacol 124(1):93–100
Richter-Levin G, Segal M (1991) The effects of serotonin depletion and raphe grafts on hippocampal electrophysiology and behavior. J Neurosci 11(6):1585–1596
Rodsiri R, Spicer C, Green AR, Marsden CA, Fone KC (2011) Acute concomitant effects of MDMA binge dosing on extracellular 5-HT, locomotion and body temperature and the long-term effect on novel object discrimination in rats. Psychopharmacology 213(2–3):365–376
Ropert N (1988) Inhibitory action of serotonin in CA1 hippocampal neurons in vitro. Neuroscience 26:69–81
Rozas C, Loyola S, Ugarte G, Zeise ML, Reyes-Parada M, Pancetti F, Rojas P, Morales B (2012) Acutely applied MDMA enhances long-term potentiation in rat hippocampus involving D1/D5 and 5-HT2 receptors through a polysynaptic mechanism. Eur Neuropsychopharmacol 22(8):584–595
Rudnick G, Wall SC (1992) The molecular mechanism of ‘ecstasy’ [3,4-methylenedioxy-methamphetamine (MDMA)]: serotonin transporters are targets for MDMA-induced serotonin release. Proc Natl Acad Sci USA 89:1817–1821
Schaefer HS, Putnam KM, Benca RM, Davidson RJ (2006) Event-related functional magnetic resonance imaging measures of neural activity to positive social stimuli in pre- and post-treatment depression. Biol Psychiatry 60:974–986
Segal M (1980) The action of serotonin in the hippocampal slice preparation. J Physiol (London) 303:423–439
Sitte HH, Scholze P, Schloss P, Pifl C, Singer EA (2000) Characterization of carrier-mediated efflux in human embryonic kidney 293 cells stably expressing the rat serotonin transporter: a superfusion study. J Neurochem 74:1317–1324
Staubli U, Otaky N (1994) Serotonin controls the magnitude of LTP induced by theta bursts via an action on NMDA- receptor-mediated responses. Brain Res 643:10–16
Stough C, King R, Papafotiou K, Swann P, Ogden E, Wesnes K, Downey LA (2012) The acute effects of 3,4-methylenedioxymethamphetamine and d-methamphetamine on human cognitive functioning. Psychopharmacology 220(4):799–807
Tse WS, Bond AJ (2002) Serotonergic intervention affects both social dominance and affiliative behaviour. Psychopharmacology 161(3):324–330
van der Veen FM, Evers EA, van Deursen JA, Deutz NE, Backesm WH, Schmitt JA (2006) Acute tryptophan depletion reduces activation in the right hippocampus during encoding in an episodic memory task. Neuroimage 31(3):1188–1196
Villani F, Johnston D (1993) Serotonin inhibits induction of long-term potentiation at commissural synapses in hippocampus. Brain Res 606:304–308
Wang SH, Morris RG (2010) Hippocampal-neocortical interactions in memory formation, consolidation, and reconsolidation. Annu Rev Psychol 61(49–79):C1–C4
Whitlock JR, Heynen AJ, Shuler MG, Bear MF (2006) Learning induces long-term potentiation in the hippocampus. Science 313:1093–1097
Winterer J, Stempel AV, Dugladze T, Földy C, Maziashvili N, Zivkovic AR, Priller J, Soltesz I, Gloveli T, Schmitz D (2011) Cell-type-specific modulation of feedback inhibition by serotonin in the hippocampus. J Neurosci 31(23):8464–8475
Young SN, Leyton M (2002) The role of serotonin in human mood and social interaction Insight from altered tryptophan levels. Pharmacol Biochem Behav 71(4):857–865
Acknowledgments
Funding for this study was provided by European Commission Grants (Ecstasy damage: QLG3-CT2002-00809 and Newmood: LSHM-CT-2004-503474).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mlinar, B., Stocca, G. & Corradetti, R. Endogenous serotonin facilitates hippocampal long-term potentiation at CA3/CA1 synapses. J Neural Transm 122, 177–185 (2015). https://doi.org/10.1007/s00702-014-1246-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-014-1246-7