Abstract
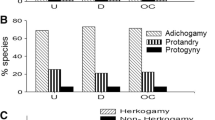
The goal of this study was to assess breeding systems, and inbreeding and outbreeding depression in four vegetation types in the Venezuelan central plains: forest, ecotone, savanna and disturbed areas. Such analysis allowed inferring how reproductive systems are influenced by the structure and complexity of the vegetation. This analysis was conducted considering the life form, successional stage, dispersal syndrome, pollination system specificity, and flowering time. Information on sexual systems, dichogamy and herkogamy, was also included. Experimental pollination tests were carried out on 115 plant species belonging to 41 plant families. Most species were non-agamospermous, and tended to have a mixed breeding system: partially spontaneous self-pollinated, and partially xenogamous. Xenogamous and non-spontaneous self-pollinated species were the second most frequent categories, which are represented by woody species, dispersed by frugivores or abiotically, with polyphilous and monophilous pollination systems, that flower during dry periods in the forest and forest-savanna transition. The high incidence of partial self-incompatibility was associated with herbaceous species, seed dispersal by granivores and epizoochory, and flowering throughout the rainy period in the herbaceous communities. Self-incompatibility was mainly associated with the woody condition and flower production during the dry period in forest communities. The proportion of species with inbreeding depression increased from forest to disturbed areas, and was frequently related with herbs, seed dispersal by granivores and epizoochory, and flowering during the rainy period. Outbreeding strategies and inbreeding depression increased from disturbed areas to forest along with the increase in woody life forms, frugivory and wind dispersal, and flowering during drought period.



Similar content being viewed by others
References
Baker HG (1978) Invasion and replacement in Californian and neotropical grasslands. In: Wilson JR (ed) Plant relations in pastures. CSIRO, East Melbourn, pp 368–383
Barret SCH (2014) Evolution of mating systems: outcrossing versus selfing. In: Losos J (ed) The Princeton guide to evolution. Princeton University Press, Princeton, pp 356–362
Barrios Y, Ramírez N (2008) Depresión por exogamia y biología reproductiva de Nymphaea ampla (Salisb.) DC. (Nymphaeaceae). Acta Bot Venez 31:539–556
Bawa KS, Perry DR, Beach JH (1985) Reproductive biology of tropical lowland rain forest trees. II. Sexual systems and incompatibility mechanisms. Amer J Bot 72:331–345
Bertin RI, Newman CM (1993) Dichogamy in angiosperms. Bot Rev 59:112–152
Bolmgren K, Erikksson O (2010) Seed mass and the evolution of fleshy fruits in angiosperms. Oikos 119:707–718. https://doi.org/10.1111/j.1600-0706.2009.17944.x
Buitrón-Jurado G, Ramírez N (2014) Dispersal spectra, diaspore size and the importance of endozoochory in the equatorial Andean montane forests. Flora 209:299–311
Cardoso JCF, Viana MC, Matias R, Furtado MT, Caetano APS, Consolaro H, Brito VLG (2018) Towards a unified terminology for angiosperm reproductive systems. Acta Bot Brasil 32:329–348. https://doi.org/10.1590/0102-33062018abb0124
Carpenter RJ, Read J, Jaffré T (2003) Reproductive traits of tropical rain-forest trees in New Caledonia. J Trop Ecol 19:351–365. https://doi.org/10.1017/S0266467403003407
Charlesworth D, Charlesworth B (1987) Inbreeding depression and its evolutionary consequences. Annual Rev Ecol Syst 18:237–268
Chazdon RL, Careaga S, Webb C, Vargas O (2003) Community and phylogenetic structure of reproductive traits of woody species in web tropical forests. Ecol Monogr 73:331–348. https://doi.org/10.1890/02-4037
Devlaeminck R, Bossuyt B, Hermy M (2005) Inflow of seeds through the forest edge: evidence from seed bank and vegetation patterns. Pl Ecol 176:1–17
Dorken ME, Husband BC (1999) Self-sterility in the understory herb Clintonia borealis (Liliaceae). Int J Pl Sci 160:577–584
Faegri K, van der Pijl L (1979) The principles of pollination ecology. Pergamon, Oxford
Firetti F (2018) Apomixis in neotropical vegetation. In: Sebata A (ed) Vegetation, vol 7. IntechOpen, London, pp 129–149. https://doi.org/10.5772/intechopen.69411
Fischer M, Matthies D (1997) Mating structure and inbreeding and outbreeding depression in the rare plant Gentianella germanica (Gentianaceae). Amer J Bot 84:1685–1692
Goodwilie C, Kalisz S, Eckert CG (2005) The evolutionary enigma of mixed mating systems in plants: occurrence, theoretical explanations, and empirical evidence. Annual Rev Ecol Evol Syst 36:47–79
Graae BJ (2002) The role of epizoochorous seed dispersal of forest plant species in a fragmented landscape. Seed Sci Res 12:113–121. https://doi.org/10.1079/SSR2002103
Harder LD, Richards SA, Routley MB (2007) Effect of reproductive compensation, gamete discounting and reproductive assurance on mating-system diversity in hermaphrodites. Evolution 62:157–172. https://doi.org/10.1111/j.1558-5646.2007.00272.x
Hilje B, Calvo-Alvarado J, Jiménez-Rodríguez C, Sánchez-Azofeifa A (2015) Tree species composition, breeding systems, and pollination and dispersal syndromes in three forest successional stages in a tropical dry forest in mesoamerica. Trop Conservation Sci 8:76–94
Hokche O, Ramírez N (2008) Sistemas reproductivos de especies de Melastomataceae en la Gran Sabana (Estado Bolívar, Venezuela). Acta Bot Venez 31:387–408
Igic B, Lande R, Kohn JR (2008) Loss of self-incompatibility and its evolutionary consequences. Int J Pl Sci 169:93–104. https://doi.org/10.1086/523362
Jaimes I, Ramírez N (1998) Autoincompatibilidad, autogamia y agamospermia en angiospermas tropicales. Acta Biol Venez 18:59–80
Jara-Guerrero A, De la Cruz M, Méndez M (2011) Seed dispersal spectrum of woody species in south Ecuadorian dry forests: environmental correlates and the effect of considering species abundance. Biotropica 43:722–730
Legendre L, Legendre P (1993) Numerical ecology. Elsevier, Amsterdam
Lemus-Jiménez L, Ramírez N (2005) Sistemas reproductivos de plantas en tres hábitats de la planicie costera de Paraguaná, Venezuela. Revista Biol Trop 53:415–430
Levin DA (2012) Mating system shifts on the trailing edge. Ann Bot (Oxford) 109:613–620
Lloyd DG, Webb CJ (1986) The avoidance of interference between the presentation of pollen and stigmas in angiosperms. I. Dichogamy. New Zealand J Bot 24:135–162
Lloyd DG, Schoen DJ (1992) Self-fertilization and cross-fertilization in plants. 1. Functional dimensions. Int J Pl Sci 153:358–369
Machado IC, Lopes AV, Sazima M (2006) Plant sexual systems and a review of the breeding system studies in the Caatinga, a Brazilian tropical dry forest. Ann Bot (Oxford) 97:277–287
Morales CL, Galetto L (2003) Influence of compatibility system and life form on plant reproductive success. Pl Biol 5:567–573. https://doi.org/10.1055/s-2003-44794
Newstrom L, Robertson A (2005) Progress in understanding pollination systems in New Zealand. New Zealand J Bot 43:1–59. https://doi.org/10.1080/0028825X.2005.9512943
Oliveira PE, Gibbs PE (2000) Reproductive biology of woody plants in a cerrado community of Central Brazil. Flora 195:311–329. https://doi.org/10.1016/S0367-2530(17)30990-8
Opler PA, Baker HG, Frankie GW (1980) Plant reproductive characteristics during secondary succession in neotropical lowland forest ecosystems. Biotropica (Suppl) 12:40–46
Parker MA (1992) Outbreeding depression in selfing annual. Evolution 46:837–841
Raduski AR, Haney EB, Igic B (2012) The expression of selfincompatibility in Angiosperms is bimodal. Evolution 66:1275–1283. https://doi.org/10.1111/j.1558-5646.2011.01505.x
Raffl C, Marcante S, Erschbamer B (2007) The role of spontaneous selfing in pioneer species Saxifraga aizoides. Flora 202:128–132. https://doi.org/10.1016/j.flora.2006.05.002
Raimúndez E, Ramírez N (1998) Estrategia reproductiva de una hierba perenne: Hypoxis decumbens (Hypoxidaceae). Revista Biol Trop 46:555–565
Ramírez N (2002) Reproductive phenology, life-forms, and habitats of the Venezuelan Central Plain. Amer J Bot 89:836–842. https://doi.org/10.3732/ajb.89.5.836
Ramírez N (2003) Diversidad de especies y estructura de la vegetación de una comunidad de sabana en los Altos Llanos Centrales Venezolanos. Acta Biol Venez 23:47–76
Ramírez N (2004a) Ecology of pollination in a tropical Venezuelan savanna. Pl Ecol 173:171–189
Ramírez N (2004b) Pollination specialization and time of pollination on a tropical Venezuelan plain: variations in time and space. Bot J Linn Soc 145:1–16. https://doi.org/10.1111/j.1095-8339.2004.00181.x
Ramírez N (2005) Plant sexual systems, dichogamy, and herkogamy in the Venezuelan Central Plain. Flora 200:30–48. https://doi.org/10.1016/j.flora.2005.01.002
Ramírez N (2007) Biología reproductiva de Amasonia campestris (Aubl.) Moldenke (Verbeneceae) en los Llanos Centrales Venezolanos. Acta Bot Venez 30:385–414. https://doi.org/10.1016/j.baae.2005.01.002
Ramírez N, Briceño H (2021) Pollination types and plant reproductive systems of two areas of Venezuelan cloud forests. Int J Pl Rep Biol 13:82–103. https://doi.org/10.1007/BF00986189
Ramírez N, Brito Y (1990) Reproductive biology of a tropical palm swamp community in the Venezuelan Llanos. Amer J Bot 77:1260–1271
Ramírez N, Hokche O (2019) Outbreeding and inbreeding strategies in herbaceous-shrubby communities in the Venezuelan Gran Sabana Plateau. AoB PLANTS 11:plz032. https://doi.org/10.1093/aobpla/plz032
Ramírez N, Nassar J (2016) Breeding systems in angiosperms: novel inferences from a new analytical approach. Pl Syst Evol 303:119–137. https://doi.org/10.1007/s00606-016-1357-8
Ramírez N, Navarro L (2010) Trends in the reproductive biology of Venezuelan Melochia (Sterculiaceae) species. Pl Syst Evol 289:147–163. https://doi.org/10.1007/s00606-010-0340-z
Ramírez N, Sobrevila C, de Enrech NX, Ruìz-Zapata T (1984) Floral Biology and breeding system of Bauhinia ungulata L. (Leguminosae). a bot-pollinated tree in Venezuelan “Llanos.” Amer J Bot 71:273–280
Richards AJ (1997) Plant breeding systems, 2nd edn. Chapman & Hall, London
Riveros M, Humaña AM, Arroyo MK (1996) Sistemas de reproducción en especies del bosque Valdiviano (40° Latitud Sur). ØYTON 58:167–176
Ruiz-Zapata T, Arroyo MTK (1978) Plant reproductive ecology of a secondary deciduous forest in Venezuela. Biotropica 10:221–230
San José JJ, Fariñas MR (1983) Changes in tree density and species composition in a protected Trachypogon savanna, Venezuela. Ecology 64:447–453
San José JJ, Fariñas MR (1991) Temporal changes in the structure of a Trachypogon savanna protected for 25 years. Acta Oecol 12:237–247
Sarmiento G, Monasterio M (1968) Corte ecológico del estado Guárico. Bol Soc Venez Ci Nat 113:83–160
Schleuning M, Templin M, Vallido G, Becker T, Durka W, Fischer M, Matthies D (2011) Effects of inbreeding, outbreeding, and supplemental pollen reproduction of a hummingbird-pollinated clonal Amazonia herb. Biotropica 43:183–191. https://doi.org/10.1111/j.1744-7429.2010.00663.x
Silberbauer-Gottsberger I, Gottsberger G (1988) A polinização das plantas do cerrado. Revista Brasil Biol 48:651–663
Sobrevila C, Arroyo MTK (1982) Breeding systems in a Montane Tropical cloud forest in Venezuela. Pl Syst Evol 140:19–37
Sokal RR, Rohlf FJ (1998) Biometry. W. H. Freeman and Company, New York
Statsoft Inc (2007) STATISTICA. Data analysis software system. Version 8.0. StatSoft, Tulsa. Available at: www.statsoft.com
Valerio R, Ramírez N (2003) Depresión exogáica y biología reproductiva de Talinum paniculatum (Jacq.) Gaertner (Portulacaceae). Acta Bot Venez 26:111–124
Vuille F (1987) Reproductive biology of the genus Damasonium (Alismataceae). Pl Syst Evol 157:63–71
Walter H, Medina E (1971) Caracterización climática de Venezuela sobre la base de climadiagramas de estaciones particulares. Bol Soc Venez Ci Nat 119:212–240
Webb CJ, Lloyd DG (1986) The avoidance of interference between the presentation of pollen and stigmas in angiosperms II. Herkogamy. New Zealand J Bot 24:163–178
Willson MF, Irvine AK, Walsh NG (1989) Vertebrate dispersal syndromes in some Australian and New Zealand plant communities, with geographic comparisons. Biotropica 21:133–147
Acknowledgements
The author thanks A. Herrera, T. N. C. Vasconcelos and anonymous reviewers for comments on the manuscript. The author thanks O. Hokche, G. Leal, M. López, Y. Brito, and D. Vázquez for field assistance. Special thanks to H. Briceño for field and laboratory support. The author is indebted to all the people who made it possible to complete this research. The commentaries of A. Herrera on the manuscript were much appreciated. To the Sociedad Venezolana de Ciencias Naturales for allowing the use of headquarters at the Estación Biológica de los Llanos, Edo. Guárico, Venezuela. I am grateful to all plant taxonomists for plant identification (see Ramírez 2005).
Funding
This research was partially supported by Proyecto Consejo de Desarrollo Científico y Humanístico, Universidad Central de Venezuela CDCH-03.4098.99 and Proyecto CONICIT S1-96001695.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Additional information
Handling editor: Thais N. C. Vasconcelos.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Appendices
Information on electronic supplementary material
Online Resource 1. Agamospermy indexes and their qualitative categories for 109 plant species from Venezuelan Central Plain.
Online Resource 2. Breeding system indexes and their qualitative categories for 121 distylous and homostylous hermaphrodite and monoecious species from Venezuelan Central Plain.
Online Resource 3. Frequency of breeding system categories according to some functional plant traits and seral stages from Venezuelan Central Plain.
Online Resource 4. Relationship between breeding system index categories and morphological and temporal organization of sexual traits.
Online Resource 5. Inbreeding–outbreeding indexes and their qualitative categories for 121 distylous and homostylous hermaphrodite and monoecious species from Venezuelan Central Plain.
Online Resource 6. Frequency distribution of inbreeding and outbreeding depression categories related to functional traits and plant communities in the Venezuelan Central Plain.
Appendix
Rights and permissions
About this article
Cite this article
Ramírez, N. Breeding systems of four plant communities in the Venezuelan central plains. Plant Syst Evol 308, 17 (2022). https://doi.org/10.1007/s00606-021-01794-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-021-01794-9