Abstract
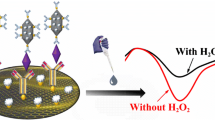
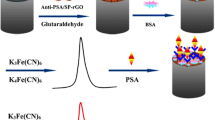
An ultrasensitive luminol electrochemiluminescence (ECL) immunosensor was constructed for the detection of prostate specific antigen (PSA) using glucose oxidase–decorated hemin-graphene oxide-gold nanoflowers ternary nanocomposites as probes. Graphene oxide was first modified with hemin and then with gold nanoflowers through an in situ growth method, which has significantly boosted the catalytic activity of this graphene oxide–based peroxidase mimetics. The biocatalytical activity of this ECL immunosensor was thoroughly investigated to achieve selective recognition of the analyte molecules (PSA) by specific binding between antigens and antibodies. The limit of detection was calculated to be 0.32 pg mL−1 with a signal-to-noise ratio of 3. A broad linear range from 7.5 × 10−4 to 2.5 ng mL−1 was obtained. Such step-by-step assembled biosensor showed controlled nanostructure and exhibited promising application in analysis of human serum samples with a recovery range of 90.6–111.8% and a RSD range of 3.9–5.5%.
Graphical abstract







Similar content being viewed by others
References
Wu L, Qu X (2015) Cancer biomarker detection: recent achievements and challenges. Chem Soc Rev 44:2963–2997. https://doi.org/10.1039/c4cs00370e
Huang X, Liu Y, Yung B et al (2017) Nanotechnology-enhanced no-wash biosensors for in vitro diagnostics of cancer. ACS Nano 11:5238–5292. https://doi.org/10.1021/acsnano.7b02618
Wu X, Hao C, Kumar J et al (2018) Environmentally responsive plasmonic nanoassemblies for biosensing. Chem Soc Rev 47:4677–4696. https://doi.org/10.1039/c7cs00894e
Husain RA, Barman SR, Chatterjee S et al (2020) Enhanced biosensing strategies using electrogenerated chemiluminescence: recent progress and future prospects. J Mater Chem B 8:3192–3212. https://doi.org/10.1039/c9tb02578b
Kal-Koshvandi AT (2020) Recent advances in optical biosensors for the detection of cancer biomarker alpha-fetoprotein (AFP). Trac-Trend Anal Chem 128:115920. https://doi.org/10.1016/j.trac.2020.115920
Ma F, Li Y, Tang B, Zhang CY (2016) Fluorescent biosensors based on single-molecule counting. Acc Chem Res 49:1722–1730. https://doi.org/10.1021/acs.accounts.6b00237
Zhou H, Liu J, Xu JJ et al (2018) Optical nano-biosensing interface via nucleic acid amplification strategy: construction and application. Chem Soc Rev 47:1996–2019. https://doi.org/10.1039/c7cs00573c
Tang L, Li J (2017) Plasmon-based colorimetric nanosensors for ultrasensitive molecular diagnostics. ACS Sens 2:857–875. https://doi.org/10.1021/acssensors.7b00282
Du FX, Chen YQ, Meng CD et al (2021) Recent advances in electrochemiluminescence immunoassay based on multiple-signal strategy. Curr Opin Electrochem 28:100725. https://doi.org/10.1016/j.coelec.2021.100725
Wang Y, Zhao GH, Chi H et al (2021) Self-luminescent lanthanide metal-organic frameworks as signal probes in electrochemiluminescence immunoassay. J Am Chem Soc 143:504–512. https://doi.org/10.1021/jacs.0c12449
Wang B, Shi S, Yang X et al (2020) Separation-free electrogenerated chemiluminescence immunoassay incorporating target assistant proximity hybridization and dynamically competitive hybridization of a DNA signal probe. Anal Chem 92:884–891. https://doi.org/10.1021/acs.analchem.9b03662
Hong D, Jo E-J, Kim K et al (2020) Ru(bpy)32+-loaded mesoporous silica nanoparticles as electrochemiluminescent probes of a lateral flow immunosensor for highly sensitive and quantitative detection of troponin I. Small 16:2004535. https://doi.org/10.1002/smll.202004535
Khoshfetrat SM, Hashemi P, Afkhami A et al (2021) Cascade electrochemiluminescence-based integrated graphitic carbon nitride-encapsulated metal-organic framework nanozyme for prostate-specific antigen biosensing. Sens Actuators, B Chem 348:130658. https://doi.org/10.1016/j.snb.2021.130658
Jia HY, Yu SQ, Yang L et al (2021) Near-infrared electrochemiluminescence of dual-stabilizer-capped Au nanoclusters for immunoassays. Acs Appl Nano Mater 4:2657–2663. https://doi.org/10.1021/acsanm.0c03284
Yu L, Zhang Q, Kang Q et al (2020) Near-infrared electrochemiluminescence immunoassay with biocompatible Au nanoclusters as tags. Anal Chem 92:7581–7587. https://doi.org/10.1021/acs.analchem.0c00125
Hong GL, Su CP, Huang ZN et al (2021) Electrochemiluminescence immunoassay platform with immunoglobulin G-encapsulated gold nanoclusters as a “two-in-one” probe. Anal Chem 93:13022–13028. https://doi.org/10.1021/acs.analchem.1c02850
Kannan P, Chen J, Su FM et al (2019) Faraday-cage-type electrochemiluminescence immunoassay: a rise of advanced biosensing strategy. Anal Chem 91:14792–14802. https://doi.org/10.1021/acs.analchem.9b04503
Song Y, Zhang W, He SJ et al (2019) Perylene diimide and luminol as potential-resolved electrochemiluminescence nanoprobes for dual targets immunoassay at low potential. ACS Appl Mater Interfaces 11:33676–33683. https://doi.org/10.1021/acsami.9b11416
Cao J-T, Fu X-L, Zhao L-Z, et al (2020) Highly efficient resonance energy transfer in g-C3N4-Ag nanostructure: proof-of-concept toward sensitive split-type electrochemiluminescence immunoassay. Sens Actuators B-Chem 311:127926. https://doi.org/10.1016/j.snb.2020.127926
Wang WX, Liu YH, Shi TH et al (2020) Biosynthesized quantum dot for facile and ultrasensitive electrochemical and electrochemiluminescence immunoassay. Anal Chem 92:1598–1604. https://doi.org/10.1021/acs.analchem.9b04919
Liu Q, Liu XP, Wei YP et al (2017) Electrochemiluminescence immunoassay for the carcinoembryonic antigen using CdSe: Eu nanocrystals. Microchim Acta 184:1353–1360. https://doi.org/10.1007/s00604-017-2114-6
Qin DM, Jiang XH, Mo GC, et al (2020) Electrochemiluminescence immunoassay of human chorionic gonadotropin using silver carbon quantum dots and functionalized polymer nanospheres. Microchim Acta 187:482. https://doi.org/10.1007/s00604-020-04450-0
Wang XF, Gao HF, Qi HL et al (2018) Proximity hybridization-regulated immunoassay for cell surface protein and protein-overexpressing cancer cells via electrochemiluminescence. Anal Chem 90:3013–3018. https://doi.org/10.1021/acs.analchem.7b04359
Ge JJ, Zhao Y, Li CL, Jie GF (2019) Versatile electrochemiluminescence and electrochemical “on-off” assays of methyltransferases and aflatoxin B1 based on a novel multifunctional DNA nanotube. Anal Chem 91:3546–3554. https://doi.org/10.1021/acs.analchem.8b05362
Wang C, Li ZH, Ju HX (2021) Copper-doped terbium luminescent metal organic framework as an emitter and a co-reaction promoter for amplified electrochemiluminescence immunoassay. Anal Chem 93:14878–14884. https://doi.org/10.1021/acs.analchem.1c03988
Chen Y, Zhou SW, Li LL, Zhu JJ (2017) Nanomaterials-based sensitive electrochemiluminescence biosensing. Nano Today 12:98–115. https://doi.org/10.1016/j.nantod.2016.12.013
Yildiz G, Bolton-Warberg M, Awaja F (2021) Graphene and graphene oxide for bio-sensing: general properties and the effects of graphene ripples. Acta Biomater 131:62–79. https://doi.org/10.1016/j.actbio.2021.06.047
Song YJ, Qu KG, Zhao C et al (2010) Graphene oxide: intrinsic peroxidase catalytic activity and its application to glucose detection. Adv Mater 22:2206–2210. https://doi.org/10.1002/adma.200903783
Zhao Y, Zhang YQ and Jie GF (2021) An "on-off' electrochemiluminescence biosensor based on DNA nanotweezer probe coupled with tripod capture DNA for high sensitive detection of Pb2+. Sens Actuators B-Chem 326:128985. https://doi.org/10.1016/j.snb.2020.128985
Pothipor C, Jakmunee J, Bamrungsap S, Ounnunkad K (2021) An electrochemical biosensor for simultaneous detection of breast cancer clinically related microRNAs based on a gold nanoparticles/graphene quantum dots/graphene oxide film. Analyst 146:4000–4009. https://doi.org/10.1039/d1an00436k
Cao JT, Yang JJ, Zhao LT et al (2018) Graphene oxide@gold nanorods-based multiple-assisted electrochemiluminescence signal amplification strategy for sensitive detection of prostate specific antigen. Biosens Bioelectron 99:92–98. https://doi.org/10.1016/j.bios.2017.07.050
Liu WY, Yang HM, Ge SG et al (2015) Application of bimetallic PtPd alloy decorated graphene in peroxydisulfate electrochemiluminescence aptasensor based on Ag dendrites decorated indium tin oxide device. Sens Actuators B-Chem 209:32–39. https://doi.org/10.1016/j.snb.2014.11.079
Dong YL, Zhang HG, Rahman ZU et al (2012) Graphene oxide-Fe3O4 magnetic nanocomposites with peroxidase-like activity for colorimetric detection of glucose. Nanoscale 4:3969–3976. https://doi.org/10.1039/c2nr12109c
Liu J, Cui MR, Niu L et al (2016) Enhanced peroxidase-like properties of graphene-hemin-composite decorated with Au nanoflowers as electrochemical aptamer biosensor for the detection of K562 leukemia cancer cells. Chem-a Euro J 22:18001–18008. https://doi.org/10.1002/chem.201604354
Liu J, Zhai FF, Zhou H, et al (2019) Nanogold flower-inspired nanoarchitectonics enables enhanced light-to-heat conversion ability for rapid and targeted chemo-photothermal therapy of a tumor. Adv Healthcare Mater 8. https://doi.org/10.1002/adhm.201801300
Zhou H, Ding KX, Yu Q, et al (2021) Enhanced electrochemiluminescence ratiometric cytosensing based on surface plasmon resonance of Au nanoparticles and nanosucculent films. Biosens Bioelectro 189. https://doi.org/10.1016/j.bios.2021.113367
Acknowledgements
We thank all the patients and colleagues who contributed to the study.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 21675074) and the Shandong Natural Science Foundation of Shandong (ZR2017MH042), Shandong Traditional Chinese Medicine Science and Technology Development Plan Project (2017–202).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, G., Guan, X., Li, B. et al. Hemin-graphene oxide-gold nanoflower-assisted enhanced electrochemiluminescence immunosensor for determination of prostate-specific antigen. Microchim Acta 189, 297 (2022). https://doi.org/10.1007/s00604-022-05387-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-022-05387-2