Abstract
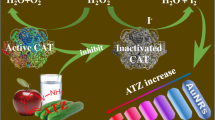
The authors describe a colorimetric method for the determination of copper ions (Cu2+) based on anti-etching of gold nanorods (AuNRs) induced by cuprous ions (Cu+). The formation of Cu+ from the reaction between Cu2+ and sodium ascorbate causes the deactivation of the horseradish peroxidase (HRP), which is used as the catalyst for the reaction of 3,3′,5,5’-Tetramethylbenzidine (TMB) and hydrogen peroxide (H2O2), resulting in a decrease in the amount of TMB2+ product that can etch AuNRs, which is accompanied by the color changes from yellow to light orange. Thus, determination of Cu2+ is achieved on the basis of AuNR absorbance at 450 nm. A determination limit as low as 0.28 nM at the 3Sblank level, and the detection of 0.4 nM Cu2+ in solution can be achieved with naked eyes, and a dynamic range of 0 to 1.6 nM toward Cu2+ was obtained.

Schematic representation of a colorimetric method for determination of Cu2+ based on the anti-etching of gold nanorods induced by Cu+, which is from the reaction of Cu2+ with sodium ascorbate.





Similar content being viewed by others
References
Viguier RF, Hulme AN (2006) A sensitized europium complex generated by micromolar concentrations of copper (I): toward the detection of copper (I) in biology. J Am Chem Soc 128:11370–11371
Song Y, Qu K, Xu C, Ren J, Qu X (2010) Visual and quantitative detection of copper ions using magnetic silicananoparticles clicked on multiwalled carbon nanotubes. Chem Commun 46:6572–6574
Cotruvo JA Jr, Aron AT, Ramos-Torres MK, Chang C (2015) Synthetic fluorescent probes for studying copper in biological systems. Chem Soc Rev 44:4400–4414
Boal AK, Rosenzweig AC (2009) Structural biology of copper trafficking. Chem Rev 109:4760–4779
Bull PC, Thomas GR, Rommens JM, Forbes JR, Cox DW (1993) The Wilson disease gene is a putative copper transporting P-type ATPase similar to the Menkes gene. Nat Genet 5:327–337
Domaille DW, Que EL, Chang CJ (2008) Synthetic fluorescent sensors for studying the cell biology of metals. Nat Chem Biol 4:168–175
Georgopoulos PG, Roy A, Yonone-Lioy MJ, Opiekun RE, Lioy PJ (2001) Environmental copper: its dynamics and human exposure issues. J. Toxicol. Environ. Health. Part B 4:341–394
Stern BR (2010) Essentiality and toxicity in copper health risk assessment: overview, update and regulatory considerations. J Toxicol Environ Health, Part A 73:114–127
Wu JF, Boyle EA (1997) Low blank preconcentration technique for the determination of lead, copper, and cadmium in small-volume seawater samples by isotope dilution ICPMS. Anal Chem 69:2464–2470
Chan MS, Huang SD (2000) Direct determination of cadmium and copper in seawater using a transversely heated graphite furnace atomic absorption spectrometer with zeeman effect background. Talanta 51:373–380
Liu Y, Ding D, Zhen Y, Guo R (2017) Amino acid-mediated ‘turn-off/turn-on’ nanozyme activity of gold nanoclusters for sensitive and selective detection of copper ions and histidine. Biosens Bioelectron 92:140–146
Li X, Fan K, Kang W, Yang R, Qu B, Lu L (2019) γ-Aminobutyric acid-modified graphene oxide as a highly selective and low-toxic fluorescent nanoprobe for relay recognition of copper(II) and cysteine. Microchim Acta 186:461
Gong YJ, Zhang XB, Zhang CC, Luo AL, Fu T, Tan W, Shen GL, Yu RQ (2012) Through bond energy transfer: a convenient and universal strategy toward efficient ratiometric fluorescent probe for bioimaging applications. Anal Chem 84:10777–10784
Yang P, Zhao Y, Lu Y, Xu QZ, Xu XW, Dong L, Yu SH (2011) Phenol formaldehyde resin nanoparticles loaded with CdTe quantum dots: a fluorescence resonance energy transfer probe for optical visual detection of copper(II) ions. ACS Nano 5:2147–2154
Wang Y, Wang L, Zhang C, Liu F (2019) A lateral flow assay for copper(II) utilizing catalytic and stem-loop based signal amplification. Microchim Acta 186:82
Zhao X, Wu KL, Lyu HY, Zhang XX, Liu ZX, Fan GC, Zhang X, Zhu XX, Liu QY (2019) Porphyrin functionalized co(OH)2/GO nanocomposites as an excellent peroxidase mimic for colorimetric biosensing. Analyst 144:5284–5291
Liu H, Ding YN, Yang BC, Liu ZX, Zhang X, Liu QY (2018) Iron doped CuSn(OH)6 microspheres as a peroxidase-mimicking artificial enzyme for H2O2 colorimetric detection. ACS Sus Chem Eng 6:14383–14393
Jin CQ, Lian JJ, Gao Y, Guo KK, Wu KL, Gao LN, Zhang XX, Zhang X, Liu QY (2019) Si doped CoO nanorods as peroxidase mimics for colorimetric sensing of reduced glutathione. ACS Sus. Chem. Eng. 7:13989–13998
Ding YN, Liu H, Gao LN, Fu M, Luo XL, Zhang X, Zhang XX, Liu QY, Zeng RC (2019) Fe-doped Ag2S with excellent peroxidase-like activity for colorimetric determination of H2O2. J alloy compd 785:1189–1197
Zhou Y, Wang S, Zhang K, Jiang X (2008) Visual detection of copper(II) by azide- and alkyne-functionalized gold nanoparticles using click chemistry. Angew Chem Int Ed 120:7564–7566
Guo L, Xu S, Ma X, Qiu B, Lin Z, Chen G (2016) Dual-color plasmonic enzyme-linked immunosorbent assay based on enzyme-mediated etching of au nanoparticles. Sci Rep 6:32755
Chen Z, Lin Y, Ma X, Guo L, Qiu B, Chen G, Lin Z (2017) Multicolor biosensor for fish freshness assessment with the naked eye. Sensors Actuators B Chem 252:201–208
Ma X, Lin Y, Guo L, Qiu B, Chen G, Yang HH, Lin Z (2017) A universal multicolor immunosensor for semiquantitative visual detection of biomarkers with the naked eyes. Biosens Bioelectron 87:122–128
Chen ZH, Chen CQ, Huang HW, Luo F, Guo LH, Zhang L, Lin ZY, Chen GN (2018) Target-induced horseradish peroxidase deactivation for multicolor colorimetric assay of hydrogen sulfide in rat brain microdialysis. Anal Chem 90:6222–6228
Xu S, Ouyang W, Xie P, Lin Y, Qiu B, Lin Z, Chen G, Guo L (2017) Highly uniform gold nanobipyramids for ultrasensitive colorimetric detection of influenza virus. Anal Chem 89:1617–1623
Xianyu YL, Zhu K, Chen WW, Wang XF, Zhao HM, Sun JS, Wang Z, Jiang XY (2013) Enzymatic assay for cu(II) with horseradish peroxidase and its application in colorimetric logic gate. Anal Chem 85:7029–7032
Mu JS, Li J, Zhao X, Yang EC, Zhao XJ (2018) Novel urchin-like Co9S8 nanomaterials with efficient intrinsic peroxidase-like activity for colorimetric sensing of copper (II) ion. Sensors Actuators B Chem 258:32–41
Chang CC, Wang GQ, Takarada T, Maeda M (2017) Iodine-mediated etching of triangular gold nanoplates for colorimetric sensing of copper ion and aptasensing of chloramphenicol. ACS Appl Mater Interfaces 9:34518–34525
Gao Q, Ji L, Wang QN, Yin K, Li JH, Chen LX (2017) Colorimetric sensor for highly sensitive and selective detection of copper ion. Anal Methods 9:5094–5100
Li XX, Qing ZH, Li YN, Zou Z, Yang S, Yang RH (2019) Natural peptide probe screened for high-performance fluorescent sensing of copper ion: especially sensitivity, rapidity, and environment-friendliness. ACS Omega 4:793–800
Liu C, Ning DH, Zhang C, Liu ZJ, Zhang RL, Zhao J, Zhao TT, Liu BH, Zhang ZP (2017) Dual-colored carbon dot ratiometric fluorescent test paper based on a specific spectral energy transfer for semiquantitative assay of copper ions. ACS Appl Mater Interfaces 9:18897–18903
Acknowledgements
We gratefully acknowledge financial support from the National Natural Science Foundation of China (21505026, 21275057, 51978290, and 51478196) and Project of Education Science Planning in Guangdong Province (2017GXJK186), Science and Technology Planning Project of Guangdong Province 2017A020212003, Key-Area Research, Development Program of Guangdong Provice (2019B110209002), Cooperation Project of Gaoming Water Supply Company of Foshan Water Industry Group (No. 20190901).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A colorimetric method for Cu2+ determination based on the etching of AuNRs by TMB and H2O2 reaction product
Electronic supplementary material
ESM 1
(DOC 1.02 mb)
Rights and permissions
About this article
Cite this article
Xu, S., Chen, X., Chen, X. et al. Visual assay for determination of copper ions based on anti-etching of gold nanorods induced by cuprous ions. Microchim Acta 187, 157 (2020). https://doi.org/10.1007/s00604-020-4149-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-4149-3