Abstract
An immunochromatographic assay (ICA) is presented that can be applied to simultaneous detection of clenbuterol (CLE) and ractopamine (RAC). It is making use of two red and blue silica nanoparticles (SiNPs) that act as labels for encoding the antibodies. This design permits multiplexed analysis in a single test line and does not require an external source for photoexcitation. Anti-CLE was labeled with red SiNPs, and anti-RAC with blue SiNPs. The capture antigens CLE-BSA and RAC-BSA were placed onto the conjugate pad and the test line of the test strip, respectively. Under bare eye examination, no cross-colored lines or nonspecific bioconjugate adsorption were observed, and the visible limit of detections for CLE (red) and RAC (blue) are 3 and 2 ng‧mL−1, respectively. This design allows for multiplexed detection with reduced device dimensions and costs, and with easy integration and manufacturing. Conceivably, the method may be extended to simultaneous determination of numerous other analytes.

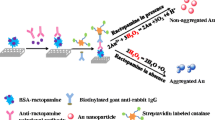
The principle of qualitative detection strategy of multiplex immunochromatographic assay for clenbuterol (CLE) and ractopamine (RAC) is schematically illustrated. Depending on the type and ratio of organic dyes, the color of colored silica nanoparticle can be tuned from red to purple and even to black (lower right corner).







Similar content being viewed by others
References
Shishani E, Chai SC, Jamokha S, Aznar G, Hoffman MK (2003) Determination of ractopamine in animal tissues by liquid chromatography-fluorescence and liquid chromatography/tandem mass spectrometry. Anal Chim Acta 483(1):137–145
Strydom PE, Frylinck L, Montgomery JL, Smith MF (2009) The comparison of three β-agonists for growth performance, carcass characteristics and meat quality of feedlot cattle. Meat Sci 81(3):557–564
Zhai H, Liu Z, Chen Z, Liang Z, Su Z, Wang S (2015) A sensitive electrochemical sensor with sulfonated graphene sheets/oxygen-functionalizedmulti-walled carbon nanotubes modified electrode for the detection of clenbuterol. Sensors Actuators B Chem 210:483–490
Zhang LH, Wang QW, Qi Y, Li L, Wang ST, Wang XH (2019) An ultrasensitive sensor based on polyoxometalate and zirconium dioxide nanocomposites hybrids material for simultaneous detection of toxic clenbuterol and ractopamine. Sensors Actuators B-Chemical 288:347–355
Fu X, Chu Y, Zhao K, Li J, Deng A (2017) Ultrasensitive detection of the beta-adrenergic agonist brombuterol by a SERS-based lateral flow immunochromatographic assay using flower-like gold-silver core-shell nanoparticles. Microchim Acta 184(6):1711–1719
Wang W, Liu L, Song S, Xu L, Kuang H, Zhu J, Xu C (2017) Identification and quantification of eight Listeria monocytogene serotypes from Listeria spp. using a gold nanoparticle-based lateral flow assay. Microchim Acta 184(3):715–724
Wang R, Zhang W, Wang P, Su X (2018) A paper-based competitive lateral flow immunoassay for multi beta-agonist residues by using a single monoclonal antibody labelled with red fluorescent nanoparticles. Microchim Acta 185(3)
Xing C, Liu L, Song S, Feng M, Kuang H, Xu C (2015) Ultrasensitive immunochromatographic assay for the simultaneous detection of five chemicals in drinking water. Biosens Bioelectron 66:445–453
Li J, Macdonald J (2016) Multiplexed lateral flow biosensors: technological advances for radically improving point-of-care diagnoses. Biosens Bioelectron 83:177–192
Peng T, Wang J, Zhao S, Zeng Y, Zheng P, Liang D, Mari GM, Jiang H (2018) Highly luminescent green-emitting Au nanocluster-based multiplex lateral flow immunoassay for ultrasensitive detection of clenbuterol and ractopamine. Anal Chim Acta 1040:143–149
Wang W, Su X, Ouyang H, Wang L, Fu Z (2016) A novel immunochromatographic assay based on a time-resolved chemiluminescence strategy for the multiplexed detection of ractopamine and clenbuterol. Anal Chim Acta 917:79–84
Zhang M-Z, Wang M-Z, Chen Z-L, Fang J-H, Fang M-M, Liu J, Yu X-P(2009) Development of a colloidal gold-based lateral-flow immunoassay for the rapid simultaneous detection of clenbuterol and ractopamine in swine urine. Anal Bioanal Chem 395(8):2591–2599
Washburn EW (1921) The dynamics of capillary flow. Phys Rev 17(3):273–283
Wang C, Hou F, Ma Y (2015) Simultaneous quantitative detection of multiple tumor markers with a rapid and sensitive multicolor quantum dots based immunochromatographic test strip. Biosens Bioelectron 68:156–162
Di Nardo F, Alladio E, Baggiani C, Cavalera S, Giovannoli C, Spano G, Anfossi L (2019)Colour-encoded lateral flow immunoassay for the simultaneous detection of aflatoxin B1 and type-B fumonisins in a single test line. Talanta 192:288–294
Lee S, Mehta S, Erickson D (2016)Two-color lateral flow assay for multiplex detection of causative agents behind acute febrile illnesses. Anal Chem 88(17):8359–8363
Di Nardo F, Baggiani C, Giovannoli C, Spano G, Anfossi L (2017) Multicolor immunochromatographic strip test based on gold nanoparticles for the determination of aflatoxin B1 and fumonisins. Microchim Acta 184(5):1295–1304
Sheng W, Chang Q, Shi Y, Duan W, Zhang Y, Wang S (2018) Visual and fluorometric lateral flow immunoassay combined with a dual-functional test mode for rapid determination of tetracycline antibiotics. Microchim Acta 185(9):404
Yen C-W, de Puig H, Tam JO, Gómez-Márquez J, Bosch I, Hamad-Schifferli K, Gehrke L (2015) Multicolored silver nanoparticles for multiplexed disease diagnostics: distinguishing dengue, yellow fever, and Ebola viruses. Lab Chip 15(7):1638–1641
Panfilova E, Shirokov A, Khlebtsov B, Matora L, Khlebtsov N (2012) Multiplexed dot immunoassay using ag nanocubes, au/ag alloy nanoparticles, and au/ag nanocages. Nano Res 5(2):124–134
Behnke T, Würth C, Hoffmann K, Hübner M, Panne U, Resch-Genger U (2011) Encapsulation of hydrophobic dyes in polystyrene micro- and nanoparticles via swelling procedures. J Fluoresc 21(3):937–944
Sun Q, Zhao G, Dou W (2015) A nonenzymatic optical immunoassay strategy for detection of Salmonella infection based on blue silica nanoparticles. Anal Chim Acta 898:109–115
Sun Q, Zhao G, Dou W (2015) Blue silica nanoparticle-based colorimetric immunoassay for detection of Salmonella pullorum. Anal Methods 7(20):8647–8654
Yu H, Zhao G, Dou W (2015) Simultaneous detection of pathogenic Bacteria using agglutination test based on colored silica nanoparticles. Curr Pharm Biotechnol 16(8):716–723
Winnik FM, Keoshkerian B, Roderick Fuller J, Hofstra PG (1990) New water-dispersible silica-based pigments: synthesis and characterization. Dyes Pigments 14(2):101–112
Giesche H, Matijević E (1991)Well-defined pigments: I. monodispersed silica-acid dyes systems. Dyes Pigments 17(4):323–340
Zhu C, Zhao G, Dou W (2018) A new synthesis method for bright monodispersed core-shell colored silica submicron particles. J Sol-Gel Sci Technol 85(1):76–83
Zhu C, Zhao G, Dou W (2018)Core-shell red silica nanoparticles based immunochromatographic assay for detection of Escherichia coli O157:H7. Anal Chim Acta 1038:97–104
Zhu C, Zhao G, Dou W (2018) Immunochromatographic assay using brightly colored silica nanoparticles as visible label for point-of-care detection of clenbuterol. Sensors Actuators B Chem 266:392–399
Alhogail S, Suaifan GARY, Zourob M (2016) Rapid colorimetric sensing platform for the detection of Listeria monocytogenes foodborne pathogen. Biosens Bioelectron 86:1061–1066
Shelver WL, Smith DJ (2002) Application of a monoclonal antibody-based enzyme-linked immunosorbent assay for the determination of Ractopamine in incurred samples from food animals. J Agric Food Chem 50(10):2742–2747
Liu N, Su P, Gao Z, Zhu M, Yang Z, Pan X, Fang Y, Chao F (2009) Simultaneous detection for three kinds of veterinary drugs: chloramphenicol, clenbuterol and 17-beta-estradiol by high-throughput suspension array technology. Anal Chim Acta 632(1):128–134
Acknowledgments
Support by National Natural Science Foundation of Zhejiang Province (LY17C200003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 799 kb)
Rights and permissions
About this article
Cite this article
Yu, Q., Liu, J., Zhao, G. et al. A silica nanoparticle based 2-color immunochromatographic assay for simultaneous determination of clenbuterol and ractopamine. Microchim Acta 186, 421 (2019). https://doi.org/10.1007/s00604-019-3529-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3529-z