Abstract
Aims
Our aim was to detect the frequency of glucokinase (GCK) gene mutations in a cohort of patients with impaired fasting glucose and to describe the clinical manifestations of identified variants. We also aimed at predicting the effect of the novel missense mutations by computational approach.
Methods
Overall 100 unrelated Italian families with impaired fasting glucose were enrolled and subdivided into two cohorts according to strict and to mild criteria for diagnosis of maturity-onset diabetes of the young (MODY). GCK gene sequencing was performed in all participants.
Results
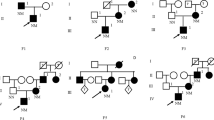
Fifty-three Italian families with 44 different mutations affecting the GCK and co-segregating with the clinical phenotype of GCK/MODY were identified. All mutations were in heterozygous state. In Sample 1, GCK defects were found in 32/36 (88.9%) subjects selected with strict MODY diagnostic criteria, while in Sample 2 GCK defects were found in 21/64 (32.8%) subjects selected with mild MODY diagnostic criteria.
Conclusions
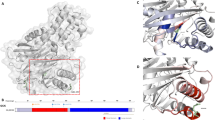
Our study enlarged the wide spectrum of GCK defects by adding 9 novel variants. The application of strict recruitment criteria resulted in 88.9% incidence of GCK/MODY, which confirmed it as the commonest form of MODY in the Italian population. In order to avoid misdiagnosis of GCK/MODY, it could be useful to perform molecular screening even if one or more clinical parameters for the diagnosis of MODY are missing. Computational analysis is useful to understand the effect of GCK defect on protein functionality, especially when the novel identified variant is a missense mutation and/or parents’ DNA is not available.

Similar content being viewed by others
References
Owen K, Hattersley AT (2001) Maturity-onset diabetes of the young: from clinical description to molecular genetic characterization. Best Pract Res Clin Endocrinol Metab 15:309–323
Fajans SS, Bell GI, Polonsky KS (2001) Molecular mechanisms and clinical pathophysiology of maturity-onset diabetes of the young. N Engl J Med 345:971–980
Barrett TG (2007) Differential diagnosis of type 1 diabetes: Which genetic syndromes need to be considered? Pediatr Diabetes 8:15–23
Gao R, Liu Y, Gjesing AP, Hollensted M, Wan X, He S, Pedersen O, Yi X, Wang J, Hansen T (2014) Evaluation of a target region capture sequencing platform using monogenic diabetes as a study-model. BMC Genet 15:13
Froguel P, Vaxillaire M, Sun F, Velho G, Zouali HM, Butel O, Lesage S, Vionnet N, Clément K, Fougerousse F, Tanizawa Y, Weissenbachparallel J, Beckmann JS, Lathrop GM, Passa PH, Permutt MA, Cohen D (1992) Close linkage of glucokinase locus on chromosome 7p to early-onset non-insulin-dependent diabetes mellitus. Nature 356:162–164
Neve B, Fernandez-Zapico ME, Ashkenazi-Katalan V, Dina C, Hamid YH, Joly E, Vaillant E, Benmezroua Y, Durand E, Bakaher N, Delannoy V, Vaxillaire M, Cook T, Dallinga-Thie GM, Jansen H, Charles MA, Clément K, Galan P, Hercberg S, Helbecque N, Charpentier G, Prentki M, Hansen T, Pedersen O, Urrutia R, Melloul D, Froguel P (2005) Role of transcription factor KLF11 and its diabetes-associated gene variants in pancreatic beta cell function. Proc Natl Acad Sci USA 102:4807–4812
Raeder H, Johansson S, Holm PI, Haldorsen IS, Mas E, Sbarra V, Nermoen I, Eide SA, Grevle L, Bjorkhaug L, Sagen JV, Aksnes L, Sovik O, Lombardo D, Molven A, Njolstad PR (2006) Mutations in the CEL VNTR cause a syndrome of diabetes and pancreatic exocrine dysfunction. Nat Genet 38:54–62
Plengvidhya N, Kooptiwut S, Songtawee N, Doi A, Furuta H, Nishi M, Nanjo K, Tantibhedhyangkul W, Boonyasrisawat W, Yenchitsomanus P, Doria A, Banchuin N (2007) PAX4 mutations in Thais with maturity onset diabetes of the young. J Clin Endocr Metab 92:2821–2826
Borowiec M, Liew CW, Thompson R, Boonyasrisawat W, Hu J, Mlynarski WM, El Khattabi I, Kim SH, Marselli L, Rich SS, Krolewski AS, Bonner-Weir S, Sharma A, Sale M, Mychaleckyj JC, Kulkarni RN, Doria A (2009) Mutations at the BLK locus linked to maturity onset diabetes of the young and beta-cell dysfunction. PNAS 25:14460–14465
Colombo C, Porzio O, Liu M, Massa O, Vasta M, Salardi S, Beccaria L, Monciotti C, Toni S, Pedersen O, Hansen T, Federici L, Pesavento R, Cadario F, Federici G, Ghirri P, Arvan P, Iafusco D, Barbetti F (2008) Seven mutations in the human insulin gene linked to permanent neonatal/infancy-onset diabetes mellitus. J Clin Invest 118:2148–2156
Boesgaard TW, Pruhova S, Andersson EA, Cinek O, Obermannova B, Lauenborg J, Damm P, Bergholdt R, Pociot F, Pisinger C, Barbetti F, Lebl J, Pedersen O, Hansen T (2010) Further evidence that mutations in INS can be a rare cause of maturity onset diabetes of the young (MODY). BMC Med Genet 11:42
Bowman P, Flanagan SE, Edghill EL, Damhuis A, Shepherd MH, Paisey R, Hattersley AT, Ellard S (2012) Heterozygous ABCC8 mutations are a cause of MODY. Diabetologia 55:123–127
Bonnefond A, Philippe J, Durand E, Dechaume A, Huyvaert M, Montagne L, Marre M, Balkau B, Fajardy I, Vambergue A, Vatin V, Delplanque J, Le Guilcher D, De Graeve F, Lecoeur C, Sand O, Vaxillaire M, Froguel P (2012) Whole-exome sequencing and high throughput genotyping identified KCNJ11 as the thirteenth MODY gene. PLoS ONE 7:e37423
Lorini R, Klersy C, d’Annunzio G, Massa O, Minuto N, Iafusco D, Bellannè-Chantelot C, Frongia AP, Toni S, Meschi F, Cerutti F, Barbetti F (2009) Maturity-onset diabetes of the young in children with incidental hyperglycemia: a multicenter Italian study of 172 families. Diabet Care 10:1864–1866
Delvecchio M, Mozzillo E, Salzano G, Iafusco D, Frontino G, Patera PI, Rabbone I, Cherubini V, Grasso V, Tinto N, Giglio S, Contreas G, Di Paola R, Salina A, Cauvin V, Tumini S, d’Annunzio G, Iughetti L, Mantovani V, Maltoni G, Toni S, Marigliano M, Barbetti F (2017) Monogenic diabetes accounts for 6.3% of cases referred to 15 Italian pediatric diabetes centers during 2007–2012. J Clin Endocrinol Metab. doi:10.1210/jc.2016-2490
Velho G, Froguel P, Clement K, Pueyo ME, Rakotoambinina B, Zouali H, Passa P, Cohen D, Robert JJ (1992) Primary pancreatic beta-cell secretory defect caused by mutations in the glucokinase in kindreds of maturity onset diabetes of the young. Lancet 340:444–448
Njølstad PR, Søvik O, Cuesta-Muñoz A, Bjørkhaug L, Massa O, Barbetti F, Undlien DE, Shiota C, Magnuson MA, Molven A, Matschinsky FM, Bell GI (2001) Neonatal diabetes mellitus due to complete glucokinase deficiency. N Engl J Med 344:1588–1592
Rubio-Cabezas O, Hattersley AT, Njølstad PR, Mlynarski W, Ellard S, White N, Chi DV, Craig ME, International Society for Pediatric and Adolescent Diabetes, ISPAD Clinical Practice Consensus Guidelines (2014) The diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabet 20:47–64
Stride A, Vaxillaire M, Tuomi T, Barbetti F, Njølstad PR, Hansen T, Costa A, Conget I, Pedersen O, Søvik O, Lorini R, Groop L, Froguel P, Hattersley AT (2000) The genetic abnormality in beta cell determines the response to an oral glucose load. Diabetologia 45:427–435
Tinto N, Zagari A, Capuano M, De Simone A, Capobianco V, Daniele G, Giugliano M, Spadaro R, Franzese A, Sacchetti L (2008) Glucokinase gene mutations: structural and genotype-phenotype analyses in MODY children from South Italy. PLoS ONE 4:1870–1878
Gasperíková D, Tribble ND, Staník J, Hucková M, Misovicová N, van de Bunt M, Valentínová L, Barrow BA, Barák L, Dobránsky R, Bereczková E, Michálek J, Wicks K, Colclough K, Knight JC, Ellard S, Klimes I, Gloyn AL (2009) Identification of a novel β-cell glucokinase (GCK) promoter mutation (−71G > C) that modulates GCK gene expression through loss of allele-specific Sp1 binding causing mild fasting hyperglycemia in humans. Diabetes 58:1929–1935
Sali A, Blundell TL (1993) Comparative protein modeling by satisfaction of spatial restraints. J Mol Biol 234:779–815
Kamata K, Mitsuya M, Nishimura T, Eiki J, Nagata Y (2004) Structural basis for allosteric regulation of the monomeric allosteric enzyme human glucokinase. Structure 12:429–438
Prisco F, Iafusco D, Franzese A, Sulli N, Barbetti F (2000) MODY 2 presenting as neonatal hyperglycaemia: A need to reshape the definition of “neonatal diabetes”? Diabetologia 43:1331–1332
Salina A, Aloi C, Pasquali L, Mascagni A, Cassanello M, Tallone R, Lugani F, Lorini R, d’Annunzio G (2012) Comment on: clinical application of best practice guidelines for genetic diagnosis of MODY2. Diabet Res Clin Pract 95:e29–e30
Zhang J, Li C, Shi T, Chen K, Shen X, Jiang H (2009) Lys169 of human glucokinase is a determinant for glucose phosphorylation: implication for the atomic mechanism of glucokinase catalysis. PLoS ONE 4:6304–6316
Mahalingam B, Cuesta-Munoz A, Davis EA, Matschinsky FM, Harrison RW, Weber IT (1999) Structural model of human glucokinase in complex with glucose and ATP: implications for the mutants that cause hypo- and hyperglycemia. Diabetes 48:1698–1705
Cartegni L, Chew SL, Krainer AR (2002) Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat Rev Genet 3:285–298
Osbak KK, Colclough K, Saint-Martin C, Beer NL, Bellanné-Chantelot C, Ellard S, Gloyn AL (2009) Update on mutations in glucokinase (GCK), which cause maturity-onset diabetes of the young, permanent neonatal diabetes, and hyperinsulinemic hypoglycemia. Hum Mutat 30:1512–1526
Massa O, Meschi F, Cuesta-Munoz A, Caumo A, Cerutti F, Toni S, Cherubini V, Guazzarotti L, Sulli N, Matschinsky FM, Lorini R, Iafusco D, Barbetti F (2001) High prevalence of glucokinase mutations in Italian children with MODY. Influence on glucose tolerance, first-phase insulin response, insulin sensitivity and BMI. Diabetologia 44:898–905
Stanik J, Dusatkova P, Cinek O, Valentinova L, Huckova M, Skopkova M, Dusatkova L, Stanikova D, Pura M, Klimes I, Lebl J, Gasperikova D, Pruhova S (2014) De novo mutations of GCK, HNF1A and HNF4A may be more frequent in MODY than previously assumed. Diabetologia 57:480–484
Acknowledgements
We would like to thank Prof. Gianmarco Ghiggeri, MD. We also thank Anna Capurro for English language revision of the manuscript.
Authors’ contributions
AS and CA conceived and designed the experiments, performed molecular investigation, analyzed data, performed computational analysis, drafted and revised the manuscript, and approved the final manuscript as submitted. NM, RT made clinical diagnosis. FL analyzed data. AM performed β-cell autoantibody detection test. OM, MC revised the manuscript. MM critically reviewed the manuscript and approved the final manuscript as submitted. GdA made clinical diagnosis, conceptualized and designed the study, critically reviewed the manuscript, and approved the final manuscript as submitted. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study has no financial support.
Statement of human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Statement of informed consent
Informed consent was obtained from participant for being included in the study.
Additional information
Managed by Massimo Porta.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aloi, C., Salina, A., Minuto, N. et al. Glucokinase mutations in pediatric patients with impaired fasting glucose. Acta Diabetol 54, 913–923 (2017). https://doi.org/10.1007/s00592-017-1021-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-017-1021-y