Abstract
Aims
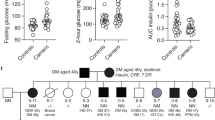
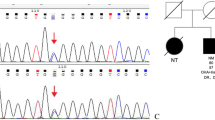
Paired box 4 (PAX4) mutations cause maturity-onset diabetes of the young, type 9 (MODY9). The molecular defect and alteration of PAX4 function associated with the mutation PAX4 IVS7-1G>A in a family with MODY9 and severe diabetic complications were studied.
Methods
We investigated the functional consequences of PAX4 IVS7-1G>A on mRNA splicing using minigene assays. Wild-type and mutant PAX4 were expressed in mouse pancreatic β- and α-cell lines, and protein levels and translocation of PAX4 into the nucleus were determined. We also examined transcriptional repression of PAX4 target-gene promoters and β-cell viability under diabetic-like (high-glucose) conditions.
Results
PAX4 IVS7-1G>A disrupts an acceptor splice site, causing an adjacent cryptic splice site within exon 8 to be used, resulting in a three-nucleotide deletion and glutamine deletion at position 250 (p.Q250del). Wild-type and PAX4 Q250del proteins were expressed at similar levels and could translocate normally into the nucleus in βTC3 and αTC1.9 cells. However, the repressor functions of PAX4 Q250del on human insulin and glucagon promoters in INS-1 832/13 and αTC1.9 cells were significantly decreased, compared with that of wild-type PAX4. Moreover, the rate of apoptosis was increased in INS-1 cells over-expressing PAX4 Q250del when cultured in high-glucose conditions.
Conclusions
PAX4 IVS7-1G>A caused aberrant mRNA splicing and PAX4 Q250 deletion. The mutation impaired PAX4 repressor functions on target-gene promoters and increased susceptibility to apoptosis upon high glucose exposure. Thus, PAX4 IVS7-1G>A contributes to the pathogenesis of diabetes in this MODY9 family through β-cell dysfunction.




Similar content being viewed by others
References
Fajans SS, Bell GI, Polonsky KS (2001) Molecular mechanisms and clinical pathophysiology of maturity-onset diabetes of the young. N Engl J Med 345(13):971–980
Yamagata K, Furuta H, Oda N, Kaisaki PJ, Menzel S, Cox NJ et al (1996) Mutations in the hepatocyte nuclear factor-4alpha gene in maturity-onset diabetes of the young (MODY1). Nature 384(6608):458–460
Froguel P (1996) Glucokinase and MODY: from the gene to the disease. Diabet Med 13(9 Suppl 6):S96–S97
Yamagata K, Oda N, Kaisaki PJ, Menzel S, Furuta H, Vaxillaire M et al (1996) Mutations in the hepatocyte nuclear factor-1 alpha gene in maturity-onset diabetes of the young (MODY3). Nature 384(6608):455–458
Stoffers DA, Ferrer J, Clarke WL, Habener JF (1997) Early-onset type-II diabetes mellitus (MODY4) linked to IPF1. Nat Genet 17(2):138–139
Horikawa Y, Iwasaki N, Hara M, Furuta H, Hinokio Y, Cockburn BN et al (1997) Mutation in hepatocyte nuclear factor-1 beta gene (TCF2) associated with MODY. Nat Genet 17(4):384–385
Malecki MT, Jhala US, Antonellis A, Fields L, Doria A, Orban T et al (1999) Mutations in NEUROD1 are associated with the development of type 2 diabetes mellitus. Nat Genet 23(3):323–328
Bell GI, Froguel P, Nishi S, Pilkis SJ, Stoffel M, Takeda J et al (1993) Mutations of the human glucokinase gene and diabetes mellitus. Trends Endocrinol Metab 4(3):86–90
Plengvidhya N, Boonyasrisawat W, Chongjaroen N, Jungtrakoon P, Sriussadaporn S, Vannaseang S et al (2009) Mutations of maturity-onset diabetes of the young (MODY) genes in Thais with early-onset type 2 diabetes mellitus. Clin Endocrinol 70(6):847–853
Lee HJ, Ahn CW, Kim SJ, Song YD, Lim SK, Kim KR et al (2001) Mutation in hepatocyte nuclear factor-1alpha is not a common cause of MODY and early-onset type 2 diabetes in Korea. Acta Diabetol 38(3):123–127
Chapla A, Mruthyunjaya MD, Asha HS, Varghese D, Varshney M, Vasan SK et al (2015) Maturity-onset diabetes of the young in India—a distinctive mutation pattern identified through targeted next-generation sequencing. Clin Endocrinol 82(4):533–542
Plengvidhya N, Kooptiwut S, Songtawee N, Doi A, Furuta H, Nishi M et al (2007) PAX4 mutations in Thais with maturity onset diabetes of the young. J Clin Endocrinol Metab 92(7):2821–2826
Kooptiwut S, Plengvidhya N, Chukijrungroat T, Sujjitjoon J, Semprasert N, Furuta H et al (2012) Defective PAX4 R192H transcriptional repressor activities associated with maturity onset diabetes of the young and early onset-age of type 2 diabetes. J Diabetes Complicat 26(4):343–347
Biason-Lauber A, Boehm B, Lang-Muritano M, Gauthier BR, Brun T, Wollheim CB et al (2005) Association of childhood type 1 diabetes mellitus with a variant of PAX4: possible link to beta cell regenerative capacity. Diabetologia 48(5):900–905
Shimajiri Y, Sanke T, Furuta H, Hanabusa T, Nakagawa T, Fujitani Y et al (2001) A missense mutation of Pax4 gene (R121W) is associated with type 2 diabetes in Japanese. Diabetes 50(12):2864–2869
Shimajiri Y, Shimabukuro M, Tomoyose T, Yogi H, Komiya I, Takasu N (2003) PAX4 mutation (R121W) as a prodiabetic variant in Okinawans. Biochem Biophys Res Commun 302(2):342–344
Mauvais-Jarvis F, Smith SB, Le May C, Leal SM, Gautier JF, Molokhia M, Riveline JP, Rajan AS, Kevorkian JP, Zhang S, Vexiau P, German MS, Vaisse C (2004) PAX4 gene variations predispose to ketosis-prone diabetes. Hum Mol Gen 13(24):3151–3159
Sosa-Pineda B (2004) The gene Pax4 is an essential regulator of pancreatic beta-cell development. Mol Cells 18(3):289–294
Sosa-Pineda B, Chowdhury K, Torres M, Oliver G, Gruss P (1997) The Pax4 gene is essential for differentiation of insulin-producing beta cells in the mammalian pancreas. Nature 386(6623):399–402
Brun T, Franklin I, St-Onge L, Biason-Lauber A, Schoenle EJ, Wollheim CB et al (2004) The diabetes-linked transcription factor PAX4 promotes beta-cell proliferation and survival in rat and human islets. J Cell Biol 167(6):1123–1135
Smith SB, Ee HC, Conners JR, German MS (1999) Paired-homeodomain transcription factor PAX4 acts as a transcriptional repressor in early pancreatic development. Mol Cell Biol 19(12):8272–8280
He KH, Lorenzo PI, Brun T, Jimenez Moreno CM, Aeberhard D, Ortega JV et al (2011) In vivo conditional Pax4 overexpression in mature islet beta-cells prevents stress-induced hyperglycemia in mice. Diabetes 60(6):1705–1715
Jo W, Endo M, Ishizu K, Nakamura A, Tajima T (2011) A novel PAX4 mutation in a Japanese patient with maturity-onset diabetes of the young. Tohoku J Exp Med 223(2):113–118
Gaildrat P, Killian A, Martins A, Tournier I, Frebourg T, Tosi M (2010) Use of splicing reporter minigene assay to evaluate the effect on splicing of unclassified genetic variants. Methods Mol Biol 653:249–257
Cooper TA (2005) Use of minigene systems to dissect alternative splicing elements. Methods 37(4):331–340
Petersen HV, Jorgensen MC, Andersen FG, Jensen J, Tove F, Jorgensen R et al (2000) Pax4 represses pancreatic glucagon gene expression. Mol Cell Biol Res Commun 3(4):249–254
Gamez-Pozo A, Palacios I, Kontic M, Menendez I, Camino I, Garcia-Miguel P, Abelairas J, Pestana A, Alonso J (2007) Pathogenic validation of unique germline intronic variants of RB1 in retinoblastoma patients using minigenes. Hum Mutat 28(12):1245
Weisschuh N, Wissinger B, Gramer E (2012) A splice site mutation in the PAX6 gene which induces exon skipping causes autosomal dominant inherited aniridia. Mol Vis 18:751–757
Nagamura Y, Yamazaki M, Shimazu S, Sano K, Tsukada T, Sakurai A (2012) A novel splice site mutation of the MEN1 gene identified in a patient with primary hyperparathyroidism. Endocr J 59(6):523–530
Brun T, He KH, Lupi R, Boehm B, Wojtusciszyn A, Sauter N et al (2008) The diabetes-linked transcription factor Pax4 is expressed in human pancreatic islets and is activated by mitogens and GLP-1. Hum Mol Genet 17(4):478–489
Weir GC, Sharma A, Zangen DH, Bonner-Weir S (1997) Transcription factor abnormalities as a cause of beta cell dysfunction in diabetes: a hypothesis. Acta Diabetol 34(3):177–184
Rath MF, Bailey MJ, Kim JS, Coon SL, Klein DC, Moller M (2009) Developmental and daily expression of the Pax4 and Pax6 homeobox genes in the rat retina: localization of Pax4 in photoreceptor cells. J Neurochem 108(1):285–294
Reichman S, Kalathur RK, Lambard S, Ait-Ali N, Yang Y, Lardenois A et al (2010) The homeobox gene CHX10/VSX2 regulates RdCVF promoter activity in the inner retina. Hum Mol Genet 19(2):250–261
Jacobson PA, Schladt D, Israni A, Oetting WS, Lin YC, Leduc R et al (2012) Genetic and clinical determinants of early, acute calcineurin inhibitor-related nephrotoxicity: results from a kidney transplant consortium. Transplantation 93(6):624–631
Acknowledgments
JS was supported by the joint Royal Golden Jubilee (RGJ) PhD Program of Thailand Research Fund (TRF) and Mahidol University (Grant No. PHD/0096/2550). This work was supported by a Siriraj Research Development Grant, a Faculty of Medicine Siriraj Hospital, Mahidol University Grant (to SK and NP). NP was supported by a TRF Research Career Development Grant and the Office of the Higher Education Commission and Mahidol University under the National Research Universities Initiative. PY was supported by a Siriraj Charoemprakiat Grant and a TRF Senior Research Scholar Award. JS conducted experiments, analyzed data, and prepared the manuscript. SK, NC, and WT conducted experiments and analyzed data. NP corresponded with patients, collected clinical data, discussed the experimental results, and edited the manuscript. PY planned the project and experiments, discussed the results, and edited the manuscript.
Conflict of interest
The authors declare no conflicts of interest.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Siriraj Institutional Review Board, Bangkok, Thailand, and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human and animal rights
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants for being included in the study.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Managed by Massimo Federici.
Rights and permissions
About this article
Cite this article
Sujjitjoon, J., Kooptiwut, S., Chongjaroen, N. et al. Aberrant mRNA splicing of paired box 4 (PAX4) IVS7-1G>A mutation causing maturity-onset diabetes of the young, type 9. Acta Diabetol 53, 205–216 (2016). https://doi.org/10.1007/s00592-015-0760-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-015-0760-x