Abstract
Objective
Ferritin autophagy is characterized by intracellular ferroptosis and selective ferritin degradation. However, the role of ferritin in the development of intervertebral disc degeneration (IDD) has not been elucidated. The study aimed to investigate the role of serum iron metabolism markers, especially serum ferritin (SF), in IDD.
Methods
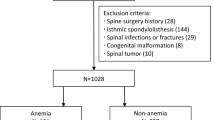
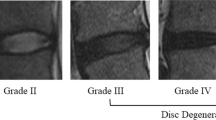
217 patients who came to the spine surgery department of our hospital for low back pain were recruited, and blood samples were collected for routine examination after admission. The cumulative grade was also calculated by summing up the Pfirrmann grade of all lumbar discs.
Results
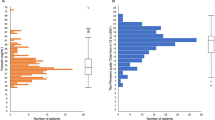
Correlation analysis showed that cumulative grade was correlated with SF (r = − 0.185, p = 0.006), not with serum iron (SI), transferrin saturation (TS), unsaturated iron-binding capacity (UIBC) and total iron-binding capacity (TIBC) (all p > 0.05). In addition, SF levels in the low severity IDD were significantly higher than high severity IDD in cumulative grade (p = 0.003) and single disc grade. No statistically significant difference was found in the other four indicators. A statistically significant difference was observed between the high (cumulative grade > 17) and low score (cumulative grade ≤ 17) groups in terms of age. According to the ROC curve, the cut-off value of SF levels was 170.5. Patients with SF < 170.5 ng/mL had severe disc degeneration. The sensitivity and specificity were 0.635 and 0.602, respectively.
Conclusion
This study preliminarily showed that SF was negatively correlated with the degree of IDD and can be used to predict IDD severity.




Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the present study are available from the corresponding author upon reasonable request.
References
Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, Carter A, Casey DC, Charlson FJ, Chen AZ, Coggeshall M (2016) Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388(10053):1545–1602. https://doi.org/10.1016/S0140-6736(16)31678-6
Mohanty S, Dahia CL (2019) Defects in intervertebral disc and spine during development, degeneration, and pain: new research directions for disc regeneration and therapy. Wiley Interdiscip Rev Dev Biol 8(4):e343. https://doi.org/10.1002/wdev.343
Sakai D, Schol J (2017) Cell therapy for intervertebral disc repair: clinical perspective. J Orthop Translat 9:8–18. https://doi.org/10.1016/j.jot.2017.02.002
Mohd Isa IL, Abbah SA, Kilcoyne M, Sakai D, Dockery P, Finn DP et al (2018) Implantation of hyaluronic acid hydrogel prevents the pain phenotype in a rat model of intervertebral disc injury. Sci Adv 4(4):eaaq0597. https://doi.org/10.1126/sciadv.aaq0597
Xu G, Liu C, Jiang J, Liang T, Yu C, Qin Z et al (2020) A novel mechanism of intervertebral disc degeneration: imbalance between autophagy and apoptosis. Epigenomics 12(13):1095–1108. https://doi.org/10.2217/epi-2020-0079
Molinos M, Almeida CR, Caldeira J, Cunha C, Gonçalves RM, Barbosa MA (2015) Inflammation in intervertebral disc degeneration and regeneration. J R Soc Interface 12(104):20141191. https://doi.org/10.1098/rsif.2014.1191
Le Maitre CL, Freemont AJ, Hoyland JA (2005) The role of interleukin-1 in the pathogenesis of human intervertebral disc degeneration. Arthritis Res Ther 7(4):R732–R745. https://doi.org/10.1186/ar1732
Chen J, Mei Z, Huang B, Zhang X, Liu J, Shan Z et al (2019) IL-6/YAP1/β-catenin signaling is involved in intervertebral disc degeneration. J Cell Physiol 234(5):5964–5971. https://doi.org/10.1002/jcp.27065
Le Maitre CL, Pockert A, Buttle DJ, Freemont AJ, Hoyland JA (2007) Matrix synthesis and degradation in human intervertebral disc degeneration. Biochem Soc Trans 35(Pt 4):652–655. https://doi.org/10.1042/BST0350652
Le Maitre CL, Freemont AJ, Hoyland JA (2004) Localization of degradative enzymes and their inhibitors in the degenerate human intervertebral disc. J Pathol 204(1):47–54. https://doi.org/10.1002/path.1608
Dong W, Liu J, Lv Y, Wang F, Liu T, Sun S et al (2019) miR-640 aggravates intervertebral disc degeneration via NF-κB and WNT signalling pathway. Cell Prolif 52(5):e12664. https://doi.org/10.1111/cpr.12664
Fang F, Jiang D (2016) IL-1β/HMGB1 signalling promotes the inflammatory cytokines release via TLR signalling in human intervertebral disc cells. Biosci Rep. https://doi.org/10.1042/BSR20160118
Wang J, Hu J, Chen X, Huang C, Lin J, Shao Z et al (2019) BRD4 inhibition regulates MAPK, NF-κB signals, and autophagy to suppress MMP-13 expression in diabetic intervertebral disc degeneration. Faseb J 33(10):11555–11566. https://doi.org/10.1096/fj.201900703R
Bin S, Xin L, Lin Z, Jinhua Z, Rui G, Xiang Z (2021) Targeting miR-10a-5p/IL-6R axis for reducing IL-6-induced cartilage cell ferroptosis. Exp Mol Pathol 118:104570. https://doi.org/10.1016/j.yexmp.2020.104570
MacKenzie EL, Iwasaki K, Tsuji Y (2008) Intracellular iron transport and storage: from molecular mechanisms to health implications. Antioxid Redox Signal 10(6):997–1030. https://doi.org/10.1089/ars.2007.1893
Weiss G (2009) Iron metabolism in the anemia of chronic disease. Biochim Biophys Acta 1790(7):682–693. https://doi.org/10.1016/j.bbagen.2008.08.006
Zhang C, Wang B, Zhao X, Li X, Lou Z, Chen X et al (2018) Iron deficiency accelerates intervertebral disc degeneration through affecting the stability of DNA polymerase epsilon complex. Am J Transl Res 10(11):3430–3442
Moroishi T, Nishiyama M, Takeda Y, Iwai K, Nakayama KI (2011) The FBXL5-IRP2 axis is integral to control of iron metabolism in vivo. Cell Metab 14(3):339–351. https://doi.org/10.1016/j.cmet.2011.07.011
Lu S, Song Y, Luo R, Li S, Li G, Wang K et al (2021) Ferroportin-dependent iron homeostasis protects against oxidative stress-induced nucleus pulposus cell ferroptosis and ameliorates intervertebral disc degeneration in vivo. Oxid Med Cell Longev 2021:6670497. https://doi.org/10.1155/2021/6670497
Zhang Y, Han S, Kong M, Tu Q, Zhang L, Ma X (2021) Single-cell RNA-seq analysis identifies unique chondrocyte subsets and reveals involvement of ferroptosis in human intervertebral disc degeneration. Osteoarthr Cartil 29(9):1324–1334. https://doi.org/10.1016/j.joca.2021.06.010
Arosio P, Ingrassia R, Cavadini P (2009) Ferritins: a family of molecules for iron storage, antioxidation and more. Biochim Biophys Acta 1790(7):589–599. https://doi.org/10.1016/j.bbagen.2008.09.004
Yang RZ, Xu WN, Zheng HL, Zheng XF, Li B, Jiang LS et al (2021) Involvement of oxidative stress-induced annulus fibrosus cell and nucleus pulposus cell ferroptosis in intervertebral disc degeneration pathogenesis. J Cell Physiol 236(4):2725–2739. https://doi.org/10.1002/jcp.30039
Verbiest H (1979) The significance and principles of computerized axial tomography in idiopathic developmental stenosis of the bony lumbar vertebral canal. Spine (Phila Pa 1976) 4(4):369–378. https://doi.org/10.1097/00007632-197907000-00005
Muraki S, Yamamoto S, Ishibashi H et al (2004) Impact of degenerative spinal diseases on bone mineral density of the lumbar spine in elderly women[J]. Osteoporos Int 15(9):724–728. https://doi.org/10.1007/s00198-004-1600-y
Zou D, Li W, Deng C et al (2019) The use of CT Hounsfield unit values to identify the undiagnosed spinal osteoporosis in patients with lumbar degenerative diseases[J]. Eur Spine J 28(8):1758–1766. https://doi.org/10.1007/s00586-018-5776-9
Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine (Phila Pa 1976) 26(17):1873–1878. https://doi.org/10.1097/00007632-200109010-00011
Zhang X, Huang Z, Xie Z, Chen Y, Zheng Z, Wei X et al (2020) Homocysteine induces oxidative stress and ferroptosis of nucleus pulposus via enhancing methylation of GPX4. Free Radic Biol Med 160:552–565. https://doi.org/10.1016/j.freeradbiomed.2020.08.029
Theil EC (2013) Ferritin: the protein nanocage and iron biomineral in health and in disease. Inorg Chem 52(21):12223–12233. https://doi.org/10.1021/ic400484n
Torti FM, Torti SV (2002) Regulation of ferritin genes and protein. Blood 99(10):3505–3516. https://doi.org/10.1182/blood.v99.10.3505
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ et al (2017) Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171(2):273–285. https://doi.org/10.1016/j.cell.2017.09.021
Arosio P, Levi S (2010) Cytosolic and mitochondrial ferritins in the regulation of cellular iron homeostasis and oxidative damage. Biochim Biophys Acta 1800(8):783–792. https://doi.org/10.1016/j.bbagen.2010.02.005
Feng C, Liu H, Yang M, Zhang Y, Huang B, Zhou Y (2016) Disc cell senescence in intervertebral disc degeneration: causes and molecular pathways. Cell Cycle 15(13):1674–1684. https://doi.org/10.1080/15384101.2016.1152433
Yurube T, Buchser WJ, Moon HJ, Hartman RA, Takayama K, Kawakami Y et al (2019) Serum and nutrient deprivation increase autophagic flux in intervertebral disc annulus fibrosus cells: an in vitro experimental study. Eur Spine J 28(5):993–1004. https://doi.org/10.1007/s00586-019-05910-9
Samartzis D, Karppinen J, Mok F, Fong DY, Luk KD, Cheung KM (2011) A population-based study of juvenile disc degeneration and its association with overweight and obesity, low back pain, and diminished functional status. J Bone Joint Surg Am 93(7):662–670. https://doi.org/10.2106/JBJS.I.01568
Ruiz-Fernández C, Francisco V, Pino J, Mera A, González-Gay MA, Gómez R et al (2019) Molecular relationships among obesity, inflammation and intervertebral disc degeneration: Are adipokines the common link? Int J Mol Sci. https://doi.org/10.3390/ijms20082030
Pan Q, Luo Y, Xia Q et al (2021) Ferroptosis and liver fibrosis[J]. Int J Med Sci 18(15):3361–3366. https://doi.org/10.7150/ijms.62903
Buzzetti E, Petta S, Manuguerra R et al (2019) Evaluating the association of serum ferritin and hepatic iron with disease severity in non-alcoholic fatty liver disease[J]. Liver Int 39(7):1325–1334. https://doi.org/10.1111/liv.14096
Chen L, Zhu Z, Peng X et al (2014) Hepatic magnetic resonance imaging with T2* mapping of ovariectomized rats: correlation between iron overload and postmenopausal osteoporosis[J]. Eur Radiol 24(7):1715–1724. https://doi.org/10.1007/s00330-014-3178-x
Funding
We would like to thank all the participants in the studies. This study was supported by Shanghai East Hospital Xuri Young Excellent Talents Program 2019xrrcjh04 and Key Laboratory of Inorganic Coating Materials, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study, or acquisition and analysis of data; drafting the article or revising it critically for important intellectual content; and the final approval of the version to be submitted. All authors are committed to being responsible for their work and consent to the publication of this paper.
Corresponding authors
Ethics declarations
Conflict of interest
The author declared no conflict of interest.
Consent to participant
All the authors listed have approved the manuscript that is enclosed. Informed consent was obtained from all participants.
Ethics approval
This is an observational study. The Ethics Committee of Shanghai East Hospital has confirmed that no ethical approval is required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, Y., Li, C., Shen, B. et al. Is intervertebral disc degeneration associated with reduction in serum ferritin?. Eur Spine J 31, 2950–2959 (2022). https://doi.org/10.1007/s00586-022-07361-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-022-07361-1