Abstract
Purpose
This review provides an overview of the patent literature on posteriorly placed intrapedicular bone anchors. Conventional pedicle screws are the gold standard to create a fixation in the vertebra for spinal fusion surgery but may lack fixation strength, especially in osteoporotic bone. The ageing population demands new bone anchors that have an increased fixation strength, that can be placed safely, and, if necessary, can be removed without damaging the surrounding tissue.
Methods
The patent search was conducted using a classification search in the Espacenet patent database. Only patents with a Cooperative Patent Classification of A61B17/70 or A61B17/7001 concerning spinal positioners and stabilizers were eligible for inclusion. The search query resulted in the identification of 731 patents. Based on preset inclusion criteria, a total of 56 unique patents on different anchoring methods were included, reviewed and categorized in this study.
Results
Five unique fixation methods were identified; (1) anchors that use threading, (2) anchors that utilize a curved path through the vertebra, (3) anchors that (partly) expand, (4) anchors that use cement and (5) anchors that are designed to initiate bone ingrowth. Of the anchor designs included in this study, eight had a corresponding commercial product, six of which were evaluated in clinical trials.
Conclusion
This review provides insights into worldwide patented intrapedicular bone anchors that aim to increase the fixation strength compared to the conventional pedicle screw. The identified anchoring methods and their working principles can be used for clinical decision-making and as a source of inspiration when designing novel bone anchors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Background of spinal fusion surgery
Spinal fusion surgery is performed to stabilize the spine in cases of spine degeneration, deformity, fractures, intervertebral disc disease, or after tumour removal [1]. Data from 2018 show that the most frequently performed spinal fusion procedure is interbody fusion with more than 350.000 procedures performed annually in the USA alone [2]. The number of performed spinal fusion surgeries is expected to grow. This growing trend is thought to be the result of an ageing population with increasing degenerative disorders and the ongoing advances in medical technology such as improved imaging technologies and anaesthesia, which allow more patients to be considered for spinal fusion surgery [3,4,5,6]. In line with this, the number of performed fusion surgeries in patients aged 65 and older in the USA increased by 239% between 1998 and 2008 [7]. Factors such as new fixation devices, new bone grafting materials and the increased availability of minimally invasive surgery, are thought to play a role in this rapid increase.
During spinal fusion surgery, two or more adjacent vertebrae are interlocked or fixed to each other to prevent all motion between them. Fixation induces subsequent bony overgrowth and fusion, which will ensure the mechanical stability in the long term. In posterior approaches for spinal fusion surgery, the vertebrae are fixed to each other using screws and rods. Pedicle screws are placed bilaterally through the pedicles of each vertebra included in the construct. The screws are connected to each other by using rods (Fig. 1a). The effectiveness of the spinal fixation, before bony fusion has occurred, depends on the anchoring strength of the pedicle screw [8].
The vertebrae consist of a cortical shell that is very dense and encloses the porous cancellous bone. Due to its porosity, the cancellous bone is more elastic and has a lower compressive strength than the cortical bone. Placing the screw through the small tubular shaped pedicle increases the contact between the screw and the cortical bone resulting in greatly improved fixation strength.
History of the pedicle screw
The discovery of X-rays, by Roentgen in 1885 gave a better understanding of the anatomy and biomechanics of the spine and allowed for the development of surgical spine interventions. The first procedure in which a spine was partly fused in order to regain stability was described by Hibbs in 1912 [9]. This spinal fusion was achieved by partly fracturing the spinal processes in order to generate contact between the bones of adjacent vertebrae. This approach required long-term immobilisation of the patient as even minor motion at the bony interface could prevent fusion. To achieve immediate fixation, the use of screws through adjacent facet joints were proposed by King in 1944 [10]. As an alternative solution, Boucher (1959) presented the use of screws through the pedicles [11], while Harrington (1962) presented the use of rods connected with hooks around the bone (lamina) to achieve scoliosis correction [12]. In 1976 Roy Camille described the use of pedicle screws in combination with plates allowing fixation to multiple pedicle screws, which distributes the force acting on the screw [13]. Currently, the pedicle screw in the gold standard for fixation of the spine and can be used to form a construct with plates, rods and wires.
Challenges when using pedicle screws
The current pedicle screws are placed via a posterior approach through the pedicle into the vertebral body. In open surgery a large incision is made along the midline of the spine over the spinous processes. The muscles are then detached from the bone and pushed aside with retractors to expose the posterior aspects of the vertebrae: the spinous processes, lamina, facet joints and transverse processes (Fig. 1b). Minimally invasive procedures aim to reduce the soft tissue damage by performing the surgery through small incisions in line with the entry points for the pedicle screws, at the drawback of the surgeon having less direct visual feedback to position the screws safely. Incorrectly placed screws may damage vascular and nervous tissue or result in poor fixation strength of the construct [14, 15]. To achieve safe placement of pedicle screws, image guidance, either via 2D fluoroscopy or 3D navigation, has become an essential part of spinal fusion surgery [16]. However, fluoroscopy has the drawbacks of exposing the patient and staff in the operating room to radiation while only providing 2D-imaging in one plane at a time. In contrast, intraoperative computed tomography (CT)-based 3D navigation uses intraoperative references to track and match the patient position as to provide highly accurate positional feedback based on the patient’s own 3D imaging information [16].
During fixation, especially when deformities are corrected, large axial and lateral forces are exerted on the pedicle screw. The resistance to axial pull-out is mainly determined by the holding strength of the screw in the cortical bone of the pedicle [17]. Larger screws may increase the fixation strength but are also more challenging to place without breaching the cortical bone [18]. The lateral forces are mainly absorbed by the cancellous bone [8, 19]. In order to achieve successful fusion, the screw must prevent motion between the adjacent vertebrae even when large forces are applied.
Inadequate screw fixation can cause complications such as screw loosening or breaking of the screw. In a study by Wu et al. [20], 4.7% of 658 placed screws had loosened and 0.46% had broken within three and a half years after placement. A lower bone density is correlated with decreased fixation strength of a screw, which may result in screw loosening and pull-out [21]. This is especially a problem in elderly as they often suffer from osteoporosis [8]. With an ageing population and an increasing need for spinal fusion surgery, improvements in the current concept of pedicle screw fixation are in demand.
Goal
Pedicle screws have several advantages over other means of fixation in the spine, but the design of the screw has virtually not changed since its introduction. Insufficient fixation strength remains an issue, especially in the osteoporotic bone of elderly patients. The goal of this review is to provide a systematic overview of patented intrapedicular bone anchors. The included bone anchor designs are compared based on the design, placement, fixation and pull-out strength as well as the removal strategy. This review could provide insights into the future direction of spine fixation and could be a source of inspiration for the design of new anchors and aid clinical decision-making.
Materials and methods
Patent search method
The patent search was conducted using the Espacenet patent database, which contains a large number of patents and supports extensive search queries. A classification search was conducted in which only patents with a Cooperative Patent Classification (CPC) of A61B17/70 or A61B17/7001 were included. Class A61B17/70 includes spinal positioners and stabilizers. Subcategories were not included as the patents in these subcategories focus on spinal positioners such as plates or the tools used to place these spinal positioners, which falls outside the scope of this review. As an exception, subclass A61B17/7001 was included, as this subclass includes screws or hooks combined with longitudinal elements which do not contact the vertebrae. This is the class in which the regular pedicle screw is listed. Subclasses were again not considered, as these focus on other devices used during spinal fusion surgery, such as the connection rods used between the screws and the longitudinal elements. The search query was enriched by a title search using key words, ensuring that only patents focused on anchoring were included. This resulted in the following final search query: (cpc = "A61B17/70" OR cpc = "A61B17/7001") AND (ti any "fix*" OR ti any "anchor*" OR ti any "screw*" OR ti any "fast*"). As a last constraint, only patents written in the English language were included.
Eligibility criteria
The scope of this study is to provide an overview of bone anchors that are placed through the pedicle, intending to replace the use of conventional pedicle screws. The identified bone anchors should allow for the connection by rods to achieve bone fusion between multiple vertebrae. Patents focusing only on the screw head or tools used during the placement of pedicle screws were not included in this review.
General results
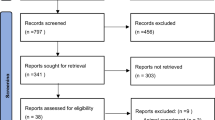
The listed search query resulted in the identification of 731 patents (February 2022). These patents were included or excluded based on the set eligibility criteria. The titles, abstracts and drawings of the patents were screened to determine if the eligiility criteria were met. When there was uncertainty as to whether a patent should be included, the description was read. Patents with the same inventors and describing the same anchoring method were indicated as duplicates although these patents might not be duplicates in the legal sense. In these cases, only the most recent patent was included in this review. This resulted in 56 unique patents that were included in this review.
Results
Overview
The patents were categorised based on the anchoring method used to ensure a good fixation between the screw and the surrounding bone. Five unique fixation methods were identified: (1) anchors that use threading, (2) anchors that utilize a curved path through the vertebra, (3) anchors that (partly) expand, (4) anchors that use cement and (5) anchors that are designed to initiate bone ingrowth. The five fixation methods are schematically represented in Fig. 2.
Schematic representation of the five identified methods to increase the fixation of bone anchors: (1) anchors that use threading, (2) anchors that utilize a curved path through the vertebra, (3) anchors that (partly) expand, (4) anchors that use cement and (5) anchors that are designed to initiate bone ingrowth.
Threaded anchors
Threaded anchors were found in fifteen patents [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36], which all describe methods to ensure fixation of the bone anchor by using one or multiple threaded sections. The fixation strength of a threaded anchor depends on the shape, material and surface properties of the threaded section in contact with the surrounding bone. Screws designed for fixation in cortical bone are often characterised by threads with a small pitch, in order to increase the number of threads in contact with the thin layer of cortical bone. Screws that are designed for fixation within cancellous bone are characterised by a larger pitch and larger thread depth to increase the contact area with the cancellous bone.
The anchors described are intended for placement via a posterior approach. After placement, the distal section of the anchor is located in the vertebral body surrounded by cancellous bone, while the proximal section of the anchor is located in the pedicle and will have contact with the cortical bone. The bone anchor described by Crook et al. [22] uses a triple lead screw that allows for a quick insertion while increasing the fixation in the cortical bone due to the smaller pitch. Yoon and Lee [23] described an anchor with a quadruple threads in the proximal section of which two threads continue in the distal section while the other two threads are carved out of the distal section. This results in a distal section with a pitch that is twice as large as the proximal section as well as a larger thread depth (Fig. 3a).
Threaded anchors in which the threads differs over the length of the anchor to optimise it for fixation within the surrounding type of bone were found in eight other patents [24,25,26,27,28,29,30,31]. The threads can be optimized by using a multi-threaded proximal section or by decreasing the minor diameter of the screw while keeping the major diameter constant to make the thread depth deeper in the distal section. The anchor described by Cole [32] does the opposite and consists of a single-threaded proximal section with a constant cross section and a tapered distal tip, which is double threaded.
Jung et al. [33] described an anchor that has a shaft in which three sections can be identified (Fig. 3b). The proximal portion is intended for fixation within the cortical bone layer and has a constant diameter that is threaded with a small pitch. The middle section has the same diameter as the proximal portion will be located in the cancellous bone and is equipped with threads with a larger pitch. The third, and most distal section, is shaped like a corkscrew, and the cancellous bone is retained inside this screw section. Alon [34] described an anchor with interrupted threaded sections that only compresses the cancellous bone without chipping the bone and thus weakening the fixation (Fig. 3c).
Anchors relying on thread (blue) in order to create a fixation with the surrounding bone. a Anchor with a smaller pitched thread at the proximal end of the anchor. Figure adapted from Yoon and Lee [23]. b Anchor with a changing pith and thread depth along the length of the anchor. Figure adapted from Jung et al. [33]. c Anchor with an interrupted threaded section to increase the fixation in the cancellous bone. Figure adapted from Alon [34]. d Anchor with a threaded section as well as a flexible section. Figure adapted from Casutt [35]. e Anchor with a flexible connection between the screw thread and the screw. Figure adapted from Biedermann et al. [36]
The anchor described by Casutt [35] employs a different way of improving fixation strength. The described anchor has a flexible zone between the more rigid upper and lower shaft regions allowing the anchor to bend along with the bone (Fig. 3d). High strains on the surrounding bone and high stresses in the shaft of the anchor are thus avoided. The placement and the design of the flexible zone can be matched with the vertebra characteristics. Biedermann et al. [36] described an anchor with incorporated flexibility as well, but in this case the screw threads and the core are connected by a ridge that allows small motions between the two (Fig. 3e).
Curved anchors
This group includes anchors that improve the fixation strength of the anchors by placing them along a curved path to employ a shape lock principle. Seven of the included patents [37,38,39,40,41,42,43] belong in this category. The included anchors may be pre curved, or include one or more flexible sections that allow for bending of the anchor.
Ben-Arye et al. [37] and Matityahu et al. [38] both described anchors that are pre curved. The curve is thought to increase the fixation as the anchor cannot be pulled out with an axial pull force. To further improve the fixation, the distal tips of the anchors that meet within the vertebral body can be connected by tightening a cable that runs through both anchors as described by Ben-Arye et al. [37] (Fig. 4a). When the anchor must be removed, for instance to treat an infection, the cable can be untightened and, subsequently, the individual anchors can be pulled out.
Anchors implanted in a curved path (green) to create an improved fixation. a Pre curved bone anchor consisting of two parts that are connected at the distal tip by a flexible wire rope that runs through the anchor. Figure adapted from Ben-Arye et al. [37]. b Bone anchors with a joint that allows bending of the anchor. Figure adapted from Gonzalez-Blohm et al. [39]. c Partly threaded anchor with a flexible section. Figure adapted from Errico et al. [40]
Gonzalez-Blohm et al. [39], Glerum et al. [43] and Meek et al. [41] described anchors that can be introduced into a vertebra via the pedicle after which the anchor bends superior and can enter the adjacent vertebra via the vertebral endplate. Gonzalez-Blohm et al. [39] and Glerum et al. [43] described a bendable section that is created by interconnected (compliant) segments (Fig. 4b). Meek et al. [41] describes an anchor that has a flexible section made out of a tube that has a helical shape with interlocking teeth similar to the flexible section in the anchor described by Errico et al. [40] (Fig. 4c). The interlocking teeth allow for flexibility while being able to transfer longitudinal rotation. Saidha and White [42] described a flexible anchor that has a bendable corkscrew structure, which allows placing the anchor in a curved pathway.
Expandable anchors
Expandable anchors were found in sixteen patents [44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59]. All of them describe bone anchors with one or more structures that can be expanded after placement in order to increase the fixation strength of the anchor. A proposed use of expansion is a secondary structure that acts similar to a wall plug [44,45,46,47]. In these anchors, the plug is first inserted into a premade cavity, after which a screw is placed leading to radial expansion of the plug. The plug shapes to the cavity increasing the pull-out strength and preventing micro-motion of the anchor.
Other patents also describe anchors that use radial expansion in order to improve the fixation, but without the use of a secondary structure [48,49,50]. Gooch [48] described an anchor that expands partly in the pedicle, Hawkins et al. [49] and Maestretti et al. [50] described anchors in which the expansion will take place inside the vertebral body, resulting in the anchor expanding inside the cancellous bone. Hawkins et al. [49] described a bone anchor consisting of two curved blades each having a barbed side at the inside of the curve meant for bone engagement (Fig. 5a). Both blades are introduced into the premade cavity where the bone engaging sides of the blades oppose each other and are in contact with the cavity’s surface. The two blades can be connected at the proximal side resulting in an expansion of the distal tips. This ensures the fixation of the anchor within the surrounding bone.
Anchors that have an expanding structure (yellow) to achieve a fixation with the surrounding bone. a Anchor consisting out of two pre curved parts that engage in the bone. Figure adapted from Hawkins et al. [49]. b Anchor with to laterally expanding blades. Figure adapted from Feng et al. [53]. c Anchor with multiple laterally expanding blades Mcdonald and Thornes [56]. d Anchor with root-like shaped struts. Figure adapted from Gregory and Ghobial [58]. e Anchor with a part that expands in a predrilled cavity by filling the expanding section with bone cement. Figure adapted from Froehlich [59]
The anchors described by Biedermann et al. [51], and Wang et al. [52] expand a fixating structure, such as a thread or a series of barbs, into the bone cavity wall. The expansion can be achieved by introducing a pin that results in expansion of the structure or by using smart materials that result in the desired expansion by heating the anchor once it is placed.
Six patents have an expandable section at the distal end of the anchor to enhance the fixation within the vertebral body [53,54,55,56,57,58,59]. Feng et al. [53] proposed to obtain the expansion by advancing two flexible strips through designated curved channels that run through the anchor. The strips extend laterally into the bone as they are advanced through the channels (Fig. 5b). Kim [54] proposed to use similar lateral expanding structures to increase the fixation strength. Nijenbanning [55], Mcdonald and Thornes [56] and Chan [57] described anchors that have a similar shape in their expanded form, resembling the expansion of an umbrella (Fig. 5c). Consequently, the strips do not cut through the bone but merely push it aside. The anchor described in Georgy and Ghobial [58] comprises of a number of pre-shaped struts that form a root-like structure in their expanded state (Fig. 5d). The anchor can be placed through a hollow screw after which the root-like structure can expand. The fixation can be further enhanced by using bone cement or other filler material. The anchor described by Froehlich [59] is able to expand in a predrilled cavity with the use of filler material (Fig. 5e). The anchor comprises a section with a flexible wall. Once the filler material is introduced in the anchor, the flexible wall will expand to assume the shape of the premade cavity. After hardening of the filler, the anchor is fixed within the bone.
Cement augmented anchors
Five anchors that use cement to increase the fixation strength of the anchor were included [31, 60,61,62,63]. In all the identified anchors in this group, a hollow section is present, to allow a fluid to run through it [31, 60,61,62,63]. These anchors can be used in combination with bone cement, for example PolyMethyl MethAcrylate (PMMA). The bone cement is pushed through the central channel and exits through one of the side channels and is pressed into the pores of the cancellous bone (Fig. 6a). As the cement hardens, a rigid construct between the screw and the porous bone is achieved. Thus, the screw is thought to have a higher pull-out force. Kohm and Ferdinand [63] propose to use such a hollow screw and place it in premade linked cavities, which are filled with cement in order to increase the fixation strength (Fig. 6b).
Anchors that use cement (orange), or aim to initiate bone ingrowth (red). [59]. a A threaded anchor with a channel through which the bone cement can be fed. Figure adapted from Zhou [62]. b Anchor fixed in the bone by utilizing two interlinked cavities filled with cement. Figure adapted from Kohm and Ferdinand [63]. c The ridges on the anchor surface cause elastic deformation of the surrounding bone during introduction, resulting in bone growth. Figure adapted from Huwais [64]. d The use of a plug that breaks upon introduction of the screw. The plug chips encourage bone ingrowth. Figure adapted from Kohketsu and Ojima [66]. e A porous structure on the threaded bone anchor that allows for bone ingrowth. Figure adapted from Liu and Yang [70]. f A threaded bone anchor with holes that allow for bone ingrowth. Figure adapted from Kyle and Patel [71]
Bone ingrowth anchors
The group bone ingrowth supporting anchors includes fifteen patents [60, 64,65,66,67,68,69,70,71,72,73,74,75,76,77]. Bone integration in the anchors can be achieved by a smart design of the shape of the anchor, as described by Huwais [64] and Juszczyk [65]. The two (partly) threaded anchors described in these patents have a noncircular cross section. Huwais [64] described an anchor with ridges that stress the bone upon implementation (Fig. 6c). The applied stresses on the bone should be between the yield point and the ultimate tensile strength, and aims to induce bone growth. Juszczyk [65] described an anchor with an elliptical cross section that has a similar working principle.
Kohketsu and Ojima and Andersson et al. described the use of artificial bone in order to obtain a firm fixation of a bone anchor [66, 67]. A plug made of artificial bone such as hardened calcium phosphate can be used. Introduction of a screw into the plug breaks it, however, the chips of the plug material ensure the fixation of the screw as they are absorbed by the bone to promote bone growth (Fig. 6d) [66].
A number of anchors increase their fixation strength by enhancing the outer surface of the anchor with a bone ingrowth-promoting layer. One option is to roughen the outer surface of the bone anchor as to create a bone-implant interface [68]. Additionally, a porous structure that allows bone ingrowth can be used to increase the fixation strength of a bone anchor. In the device described by Arnin [68], for instance, the proximal section of the bone anchor is coated with a porous hydroxyl apatite layer to promote bone ingrowth [69]. Another option is to use a porous threaded section (Fig. 6e) [70]. Li et al. [60] proposed a porous distal section that does not only allow for bone ingrowth, but also lowers the local stress to the distal tip due to the increased flexibility of this section. This anchor can also be enhanced by a channel for the application of bone cement. Finally, another option is to increase the pore diameter to allow and stimulate bone ingrowth (Fig. 6f) [71,72,73,74]. These holes can also be used for drug delivery and the application of fillers. In addition, non-threaded anchor sections can be enhanced with holes and materials that promote bone growth [75, 76]. Mehl and Mesiwala [77], for example, proposed an anchor that during insertion, accumulates bone chips in the holes and ridges that run along the anchor in order to achieve fast bone ingrowth.
From patent to commercialisation
To get more insight in the commercialisation of the designs presented in the included patents, a list of commercial product corresponding to the designs included in this study was made. These products were identified through a review of the products of the companies to which the patents were assigned and compering the resemblance between the patents and the products. This was done for each paten filed by a company. Approximately 74% of the patents included in this study are filed by a company, 10% by an academic institution and 16% by independent inventors. From this, it can be deduced that this field of research is primarily industry-driven. A list of commercial products corresponding to the bone anchors included in this study is presented in Table 1.
Most of the identified products focus on bone ingrowth, but are not (yet) used as bone anchors for application in spinal fusion surgery. Some are in the trial phase for use as a medical product, while others are clinically used but not as a substitute for pedicle screws but for hip fractures or sacroiliac (SI)-joint fusion.
Discussion
Comparative analysis
Of the patents included in this study, 27% are in the category of ‘expandable anchors’. This is remarkable since this method is not used to increase the fixation strength in commercially available intrapedicular bone anchors. Expandable structures inside the vertebral body are, however, used in vertebroplasty. During this type of surgery, a collapsed vertebra is reinforced using an expandable structure in combination with bone cement. A possible explanation for the high number of expandable anchors may be the versatile design of such anchors. There are many ways to achieve expansion, and each can be patented individually. The category ‘cement augmented anchors’ only account for 6% of the included patents, and the designs described in these patents are much more straightforward as they all contain a central channel through which the cement is transported. The other categories ‘threaded anchors’, ‘curved anchors’ and ‘bone ingrowth anchors’ account for 26%, 14% and 27%, respectively. Figure 7 shows the temporal distribution of the included patents classified per fixation method. There is an overall increase in the number of patents published over time especially when taking into account that only patents published till February 2022 are included in this review. There is no clear trend towards one of the five identified fixation methods but in the last six years, there has been a clear increase in patents that describe anchors that initiate bone ingrowth.
Comparison anchor placement
Safe anchor placement is an important issue when designing spinal bone anchors to avoid damage to the vertebra itself and the surrounding anatomical structures such as nerve roots, blood vessels and the spinal cord during anchor placement. When using threaded anchors, the aim is to achieve maximal cortical bone purchase during the placement. The highest fixation strength is achieved by anchors that have a maximum outer diameter and length that can be placed without the occurrence of cortical breach [78]. The trade-off between maximal bone purchase and avoiding damage has led to the recommendation to use a screw diameter that is 80% of the pedicle width [79]. This results in a placement accuracy that ranges from 60 to 97.5% in the lumbar spine, and from 27.6 to 96.5% in the thoracic spine using conventional pedicle screws with the free-hand method [80].
Bone anchors that are cement augmented or that have a coating to induce bone ingrowth have a similar placement strategy as the conventional pedicle screw. Although no literature could be found on the placement accuracy with such anchors, it is likely that their placement accuracy is similar to conventional pedicle screws. Additional risks that arise during placement of cement augmented bone anchors are thermal and chemical bone necrosis due to the polymerisation of the bone cement [81] and cement leakage out of the vertebral body via veins or cortical defects [82]. Although cement leakage can result in serious complications such as pulmonary embolisms, the risks of clinically relevant complications are slim: in a study of 98 patients with 474 cement augmented pedicle screws, leakage occurred in 93.6% of the patients but all cases were asymptomatic [83].
For curved or expandable bone anchors, placement is more critical as the placement of these anchors is more difficult to predict and to could potentially cause more harm. We think that real-time feedback of the anchor location within the vertebra is a necessity to prevent injury to surrounding structures when using curved or expandable anchors. 3D navigation or other systems such as electrical conductivity or diffuse reflectance spectroscopy (DRS) should be used to reliably detect cortical breaches [84].
Comparison anchor fixation
The fixation strength of bone anchors is often measured by the pull-out force; the force required to axially pull the anchor out of the vertebra. In this section we will give an indication for the fixation strength for each of the five identified fixation methods. These findings should be taken as a rough estimate for the fixation strength as differences in design, while using the same anchoring method, could result in significant differences in fixations strength.
The use of double threaded screws significantly increases the pull-out strength with 20% as compared to conventional single-threaded screws [85]. To our knowledge there is no data available on the fixation strength of curved anchors. Yet, placing the screw along a well-selected curved path as compared to the straight path of a conventional pedicle screw is likely to increase the fixation strength. Bi-cortical fixation, where the distal tip of a straight screw engages in the opposite cortex improves the pull-out force to 120% compared to conventional screw placement [8]. Similar effects might be reached when using curved intrapedicular bone anchors. Anchors with a hydroxyapatite-coating that promotes bone ingrowth can increase the pull-out strength 1.5 times as compared to the pull-out strength of conventional screws [86]. It takes, however, days to weeks for the bone to grow and increase the fixation strength. Cement augmented anchors improve the fixation even more, as the load is transferred to the vertebral body via the cement [87]. The pull-out strength of cement augmented screws can almost double the pull-out strength compared to the conventional pedicle screws that are not cement augmented [8]. Lastly, expandable bone anchors can also improve the pull-out strength compared to conventional pedicle screws [88]. The study of Wu et al. [89] shows that the used anchors became loose in 4.1% of the cases while with conventional pedicle screws, loosening occurred in 12.9% of the cases, showing clearly the advantage of the expandable anchors in terms of loosening. However, the same study showed that 0.4% of the expandable anchors broke while none of the conventional pedicle screws broke, which is a clear drawback of expandable anchors. A possible explanation is that expandable bone anchors consist of multiple moving parts that makes them more fragile.
Comparison anchor removal
Causes for implant removal are pain, discomfort or deep infection, which makes removal of bone anchors a necessity. However, the screws must be fixated well such that fusion of the vertebrae can take place. It was found that 15% of the pedicle screws are not functional anymore within one year, for instance, due to loosening [90]. In the study of Sandén et al. [91] it was found that threaded anchors were already loose after 10–22 months or could be removed by a maximum torque of only 25 Ncm. It must be noted that the fusion of the vertebra is expected to take place within the first six months after surgery. During these six months, good fixation of the screws is a necessity to allow for the desired fusion, as even minor motions between the vertebrae can prevent fusion, but afterwards, the screws are redundant. Removal of expandable anchors can be challenging depending on the design, as bone ingrowth can occur within the open spaces of the expanded anchor. This could prevent folding of the anchor into the original state and subsequently damage the pedicle during the removal of the anchor [89]. The removal of cement augmented anchors with a screw thread is also a challenge as the cement creates a firm fixation by flowing into the pores of the cancellous bone. The anchor can be removed similarly to a threaded anchor by rotating it out, but the removal of the remaining cement may cause damage to the vertebral body or the pedicle [8]. Removal of anchors with a hydroxyapatite-coating is also challenging. The study of Sandén et al. showed that the required torque to remove a hydroxyapatite-coated anchor could exceed 600 Ncm [91].
Limitations and future research
This review provides an overview of the bone anchors found in patent literature. Even though patents merely describe novel ideas without giving many details about suitability for surgical practice, we have included this information in this review to the best of our knowledge. The patent search was carried out within two patent classes: A61B17/70 and A61B17/7001, which both focus on anchors for spinal stabilisation. It would be interesting to expand this study with a patent review on bone anchors for other anatomical sites as these may also be applicable in the spine. Furthermore, only patent literature was included in this review to provide insights into the development of future intrapedicular bone anchors. Including anchor designs presented in scientific literature could also be interesting for future research.
This study provides an overview of the currently patented means to improve the fixation strength of intrapedicular bone anchors. The overview can provide insights into the future direction of technologies for spine fixation and could serve as a source of inspiration for the design of new anchors that allow for safe placement and removal while increasing the fixation strength of the anchor in the vertebra.
Conclusion
Due to ageing of the population, an increase in spinal fusion surgeries will be performed in osteoporotic bone. To achieve reliable fixation in this weaker bone, alternatives to the conventional pedicle screw, which is now the gold standard for spine fixation, are desirable. A patent search has been performed to map what lies beyond the pedicle screw. Five means to increase the fixation of a bone anchor have been identified: (1) anchors that use threading, (2) anchors that utilize a curved path through the vertebra, (3) anchors that (partly) expand, (4) anchors that use cement and (5) anchors that are designed to initiate bone ingrowth. This overview provides insights into worldwide patented creative ideas by which the fixation strength of bone anchors that run through the pedicle can be improved, which may also serve to improve fixation strength of bone anchors in general.
Availability of data and material
Data are available upon request.
References
Tukkapuram VR, Kuniyoshi A, Ito M (2019) A review of the historical evolution, biomechanical advantage, clinical applications, and safe insertion techniques of cervical pedicle screw fixation. Spine Surg Relat Res 3(2):126–135. https://doi.org/10.22603/ssrr.2018-0055
iData Research (2018) How many spinal fusions are performed each year in the United States?’, iData Research. https://idataresearch.com/how-many-instrumented-spinal-fusions-are-performed-each-year-in-the-united-states/. Accessed 24 Sep 2021
Cowan JA Jr, Dimick JB, Wainess R, Upchurch GR Jr, Chandler WF, La Marca F (2006) Changes in utilization of spinal fusion in the united states. Neurosurgery 59(1):15–20. https://doi.org/10.1227/01.neu.0000243276.09094.5f
Kobayashi K, Ando K, Nishida Y, Ishiguro N, Imagama S (2018) Epidemiological trends in spine surgery over 10 years in a multicenter database. Eur Spine J 27(8):1698–1703. https://doi.org/10.1007/s00586-018-5513-4
Cortesi PA et al (2017) Epidemiologic and economic burden attributable to first spinal fusion surgery: analysis from an Italian administrative database. Spine 42(18):1398–1404. https://doi.org/10.1097/BRS.0000000000002118
Thirukumaran CP, Raudenbush B, Li Y, Molinari R, Rubery P, Mesfin A (2016) National trends in the surgical management of adult lumbar isthmic spondylolisthesis: 1998 to 2011. Spine 41(6):490–501. https://doi.org/10.1097/BRS.0000000000001238
Rajaee SS, Bae HW, Kanim LEA, Delamarter RB (2012) Spinal fusion in the United States: analysis of trends from 1998 to 2008. Spine 37(1):67–76. https://doi.org/10.1097/BRS.0b013e31820cccfb
Zindrick MR et al (1986) A biomechanical study of intrapeduncular screw fixation in the lumbosacral spine. Clin Orthop Relat Res 203:99–112
Hibbs RA (1912) An operation for Pott’s disease of the spine. J Am Med Assoc 59(6):433–436. https://doi.org/10.1001/jama.1912.04270080115010
King D (1944) Internal fixation for lumbosacral fusion. Am J Surg 66(3):357–361. https://doi.org/10.1016/S0002-9610(44)90201-1
Boucher HH (1959) A method of spinal fusion. J Bone Jt Surg Br 41-B(2):248–259. https://doi.org/10.1302/0301-620X.41B2.248
Harrington PR (1962) Treatment of scoliosis correction and internal fixation by spine instrumentation. J Bone Jt Surg Am 44-A:591–610
Roy-Camille R, Saillant G, Berteaux D, Salgado V (1976) Osteosynthesis of thoraco-lumbar spine fractures with metal plates screwed through the vertebral pedicles. Reconstr Surg Traumatol 15:2–16
Suk S-I, Kim W-J, Lee S-M, Kim J-H, Chung E-R (2001) Thoracic pedicle screw fixation in spinal deformities: Are they really safe? Spine 26(18):2049–2057. https://doi.org/10.1097/00007632-200109150-00022
Wegener B, Birkenmaier C, Fottner A, Jansson V, Dürr HR (2008) Delayed perforation of the aorta by a thoracic pedicle screw. Eur Spine J 17(2):351–354. https://doi.org/10.1007/s00586-008-0715-9
Hussain I et al (2020) Evolving navigation, robotics, and augmented reality in minimally invasive spine surgery. Glob Spine J 10(2_Suppl):22S-33S. https://doi.org/10.1177/2192568220907896
Hirano T et al (1997) Structural characteristics of the pedicle and its role in screw stability. Spine 22(21):2504–2510. https://doi.org/10.1097/00007632-199711010-00007
Brantley AG, Mayfield JK, Koeneman JB, Clark KR (1994) The effects of pedicle screw fit an in vitro study. Spine 19(15):1752–1758. https://doi.org/10.1097/00007632-199408000-00016
Law M, Tencer A, Anderson P (1993) Caudo-cephalad loading of pedicle screws: mechanisms of loosening and methods of augmentation. Spine 18(16):2438–2443. https://doi.org/10.1097/00007632-199312000-00012
Wu J-C et al (2011) Pedicle screw loosening in dynamic stabilization: incidence, risk, and outcome in 126 patients. Neurosurg Focus 31(4):E9. https://doi.org/10.3171/2011.7.FOCUS11125
Burval DJ, McLain RF, Milks R, Inceoglu S (2007) Primary pedicle screw augmentation in osteoporotic lumbar vertebrae: biomechanical analysis of pedicle fixation strength. Spine 32(10):1077–1083. https://doi.org/10.1097/01.brs.0000261566.38422.40
Crook D, Harris P, Sharps C (2011) Triple lead bone screw. US2011152948A1
Yoon HW, Lee KH (2019) Pedicle screw with quadruple screw thread. US10512494B2
Bar Y, Hewko B, Shoham M, Zehavi E (2010) Double threaded orthopedic screw. CA2746032A1
Jo ML, Kim SW, Ryu HS, Seo JH, Song DR (2015) Implant for spine fixation. KR20150066158A
Deng X, Song Z, Yang J (2019) Pedicle screw. CN109620382A
Hess M, Schlapfer FJ (2000) Screw. ZA997344B
Burgess I, Thompson AA (2000) Improvements in and relating to bone Fixins. WO0015128A1
Qian J, Zhou B (2020) Pedicle screw capable of preventing angle loss. CN112043369A
Denis F et al (2007) Multi-thread bone screw. WO2007095447A1
Zheng S, Liao H, Zhao G, Zhou P, Yang D (2021) Spinal screw. CN112220545A
Cole C (2018) Spinal fixation device. US2018368889A1
Jung EM, Kim DW, Kim SH, Park JK (2017) Pedicle screw. KR101731421B1
Alon G (2021) Orthopedic fastener and associated systems and methods. US11000326B1
Casutt S (2007) Pedicle screw. EP1865862A1
Biedermann L, Fischer B, Matthis W (2015) Bone screw and method for providing bone screw. JP2015073905A
Ben-Arye A, Epstein A, Shezifi Y (2010) Bone anchoring system. US2010305700A1
Matityahu AM, Mcclellan RT, Dillin WH (2011) Posterior spinal fastener. EP2299921A1
Gonzalez-Blohm SA, Doulgeris JJ, Aghayev K, and Vrionis FD (2019) Transdiscal screw. US10314631B2
Errico T, Harwell B, Newton P, Shufflebarger H (2018) Fixation device and method of using the same. WO2018183837A1
Meek RN, Thaler CA, Dimmer SC (2020) Bone-fixation device and system. WO2020077457A1
Saidha S, White M (2012) Flexible helical fixation device. WO2012121705A1
Glerum C, Weiman M, Hessler T, Hill A, Sullivan M (2021) Pedicle-based intradiscal fixation devices and methods. US2021307924A1
Banouskou E, Oglaza J-F (2015) Universal anchor for bone fixation. US2015045841A1
Kim KT (2021) Screw anchor assembly and method of using the same in pedicle screw fixation. US11083509B2
Griffiths B et al (2012) Expandable fixation assemblies. EP2451373A1
Cho KJ, Shin MS, Kim JS (2008) A spinal screw module. WO2008146981A1
Gooch HL (2007) Anchor for augmentation of screw purchase and improvement of screw safety in pedicle screw fixation and bone fracture fixation systems. US2007118131A1
Hawkins JR, Grinberg A, Michielli M (2016) Revisable orthopedic anchor and methods of use. US2016008033A1
Maestretti G, Ratron Y-A, Oglaza J-F (2020) Expansible intravertebral implant system with posterior pedicle fixation. US10603080B2
Biedermann L, Matthis W, Rapp H (2016) Bone anchoring element. US2016361104A1
Wang H et al (2016) Telescopic double-threaded pull-out resistant pedicle screw assembly for medical use. WO2016054951A1
Feng Y, Lei W, Shi L, Wu Z, Yan Y, Zhang Y (2014) Medical anchor type vertebral pedicle screw. CN103860251A
Kim MS (2011) Screw for fixing vertebra. WO2011025098A1
Nijenbanning G (2008) Medical device for treating broken bones or fixing stabilising elements to bone parts. EP1937172A1
Mcdonald R, Thornes B (2020) A bolt apparatus for vertebral fixation. WO2020178409A1
Chan BM, Chirico PE, Pakbaz SR (2010) Devices for stabilizing bone compatible for use with bone screws. EP2173268A1
Georgy B, Ghobial EKR (2009) Device and method for orthopedic fracture fixation. WO2009086024A1
Froehlich M (2009) Bone anchor system. US2009099609A1
Li B, Su H, Wang J, Xu B, Zhang G (2019) Porous screw. CN109124748A
Luo Z, Wang Z, Yang H, Zhang W (2017) Combined-type perfusable pedicle screw system. WO2017197868A1
Zhou J (2014) Minimally-invasive hollow multi-side-pore pedicle screw. CN103815956A
Kohm AC, Ferdinand AE (2008) Apparatuses and methods for bone screw augmentation. WO2008121608A2
Huwais S (2015) Implant/anchor for cellular and visco-elastic materials. US2015297275A1
Juszczyk M, Kelnberger A, Wecker H (2016) Screw with an Elliptical longitudinal and cross section. EP3041435A1
Kohketsu M, Ojima S (1996) Anchor for fixing a screw in bone. GB2301535A
Andersson G, Carter A, Mcrury I, Patt B (2019) Demineralized bone fiber composition for augmentation of fixation. WO2019033082A1
Arnin U (2008) Pedicle screw surface treatment for improving bone-implant interface. CA2669250A1
Gao Y, Hao D, Wang H, Yang S, Zhao F (2015) Pedicle screw. CN104398297A
Liu Y, Yang D (2015) Spine screw. CN104840243A
Kyle K, Patel S (2018) Implants for tissue fixation and fusion. WO2018209177A1
Castro F (2020) Surgical fastener. WO2020040862A1
Bergeron B et al (2019) Material directing orthopedic anchor. WO2019152737A1
Tempco DA, Ballard RR, Miller KE, Rezach WA (2021) Bone screw and method of manufacture. US10993753B2
Aebi M, Steiner B (2000) Self cutting hollow cylindrical bone anchor. ZA994846B
Eastlack RK et al (2020) Implants for Spinal fixation and or fusion. US2020261240A1
Mehl DT, Mesiwala AH (2021) Bone screw implant for sacroiliac joint fusion. US11045238B2
Cho W, Cho SK, Wu C (2010) The biomechanics of pedicle screw-based instrumentation. J Bone Jt Surg Br 92-B(8):1061–1065. https://doi.org/10.1302/0301-620X.92B8.24237
Solitro GF, Whitlock K, Amirouche F, Mehta AI, Mcdonnell A (2019) Currently adopted criteria for pedicle screw diameter selection. Int J Spine Surg 13(2):132–145. https://doi.org/10.14444/6018
Kosmopoulos V, Schizas C (2007) Pedicle screw placement accuracy: a meta-analysis. Spine 32(3):E111. https://doi.org/10.1097/01.brs.0000254048.79024.8b
Stańczyk M, van Rietbergen B (2004) Thermal analysis of bone cement polymerisation at the cement–bone interface. J Biomech 37(12):1803–1810. https://doi.org/10.1016/j.jbiomech.2004.03.002
Yeom JS, Kim WJ, Choy WS, Lee CK, Chang B-S, Kang JW (2003) Leakage of cement in percutaneous transpedicular vertebroplasty for painful osteoporotic compression fractures. J bone jt surg Br 85(1):83–89. https://doi.org/10.1302/0301-620x.85b1.13026
Mueller JU, Baldauf J, Marx S, Kirsch M, Schroeder HW, Pillich DT (2016) Cement leakage in pedicle screw augmentation: a prospective analysis of 98 patients and 474 augmented pedicle screws. J Neurosurg Spine 25(1):103–109. https://doi.org/10.3171/2015.10.SPINE15511
Burström G et al (2019) Diffuse reflectance spectroscopy accurately identifies the pre-cortical zone to avoid impending pedicle screw breach in spinal fixation surgery. Biomed Opt Expr 10(11):5905–5920. https://doi.org/10.1364/BOE.10.005905
Seng WRD, Chou SM, Siddiqui SS, Oh JYL (2019) ‘Pedicle screw designs in spinal surgery: Is there a difference? a biomechanical study on primary and revision pull-out strength. Spine 44(3):E144. https://doi.org/10.1097/BRS.0000000000002789
Hasegawa T, Inufusa A, Imai Y, Mikawa Y, Lim T-H, An HS (2005) Hydroxyapatite-coating of pedicle screws improves resistance against pull-out force in the osteoporotic canine lumbar spine model: a pilot study. Spine J 5(3):239–243. https://doi.org/10.1016/j.spinee.2004.11.010
Becker S et al (2008) Assessment of different screw augmentation techniques and screw designs in osteoporotic spines. Eur Spine J 17(11):1462–1469. https://doi.org/10.1007/s00586-008-0769-8
Vishnubhotla S, McGarry WB, Mahar AT, Gelb DE (2011) A titanium expandable pedicle screw improves initial pullout strength as compared with standard pedicle screws. Spine J 11(8):777–781. https://doi.org/10.1016/j.spinee.2011.06.006
Wu Z et al (2012) A comparative study on screw loosening in osteoporotic lumbar spine fusion between expandable and conventional pedicle screws. Arch Orthop Trauma Surg 132(4):471–476. https://doi.org/10.1007/s00402-011-1439-6
Ohlin A, Karlsson M, Düppe H, Hasserius R, Redlund-Johnell I (1994) Complications after transpedicular stabilization of the spine a survivorship analysis of 163 cases. Spine 19(24):2774–2779. https://doi.org/10.1097/00007632-199412150-00007
Sandén B, Olerud C, Johansson C, Larsson S (2000) Improved extraction torque of hydroxyapatite-coated pedicle screws. Eur Spine J 9(6):534–537. https://doi.org/10.1007/s005860000180
‘척추임플란트’. http://www.mantiz.net/. Accessed 19 Oct 2021
Barber FA, Herbert MA (2013) Cyclic loading biomechanical analysis of the pullout strengths of rotator cuff and glenoid anchors update. Arthrosc J Arthrosc Relat Surg 29(5):832–844. https://doi.org/10.1016/j.arthro.2013.01.028
‘Technology-SpineAlign Medical’. http://www.spinealignmedical.com/technology.html. Accessed 19 Oct 2021
Anselmetti GC et al (2014) Vertebral augmentation with nitinol endoprosthesis: clinical experience in 40 patients with 1-year follow-up. Cardiovasc Intervent Radiol 37(1):193–202. https://doi.org/10.1007/s00270-013-0623-1
‘X-Bolt | SPINE’. https://www.x-bolt.com/products/spine/. Accessed 19 Oct 2021
Griffin XL, Achten J, Sones W, Cook J, Costa ML (2018) Randomised controlled trial of the sliding hip screw versus X-Bolt dynamic hip plating system for the fixation of trochanteric fractures of the hip in adults: a protocol study for WHiTE 4 (WHiTE4). BMJ Open 8(1):e019944. https://doi.org/10.1136/bmjopen-2017-019944
Griffin XL, Achten J, O’Connor HM, Cook JA, Costa ML, WHiTE Four Investigators (2021) Effect on health-related quality of life of the X-Bolt dynamic plating system versus the sliding hip screw for the fixation of trochanteric fractures of the hip in adults: the WHiTE Four randomized clinical trial. Bone Joint J 103-B(2):256–263. https://doi.org/10.1302/0301-620X.103B.BJJ-2020-1404.R1
Goh EL, Lerner RG, Achten J, Parsons N, Griffin XL, Costa PML (2020) Complications following hip fracture: results from the world hip trauma evaluation cohort study. Injury 51(6):1331–1336. https://doi.org/10.1016/j.injury.2020.03.031
Kahane S, Vaghela KR, Stammers J, Goldberg A, Smitham P (2019) biomechanical study comparing cut-out resistance of the X-Bolt® and dynamic hip screw at various tip-apex distances. Surg Technol Int 35:395–401
Clarke AD, Herron JBT, McVie JL (2017) X-Bolt unforeseen placement complication: case report. Ann Royal Coll Surg Engl 99(8):e227–e229. https://doi.org/10.1308/rcsann.2017.0152
Gosiewski JD, Holsgrove TP, Gill HS (2017) The efficacy of rotational control designs in promoting torsional stability of hip fracture fixation. Bone Jt Res 6(5):270–276. https://doi.org/10.1302/2046-3758.65.BJR-2017-0287.R1
Fernandez MA, Aquilina A, Achten J, Parsons N, Costa ML, Griffin XL (2017) The tip-apex distance in the X-Bolt dynamic plating system. Bone Jt Res 6(4):204–207. https://doi.org/10.1302/2046-3758.64.BJR-2015-0016.R2
Griffin XL, Parsons N, McArthur J, Achten J, Costa ML (2016) The warwick hip trauma evaluation one: a randomised pilot trial comparing the X-Bolt dynamic hip plating system with sliding hip screw fixation in complex extracapsular hip fractures: WHiTE (One). Bone Jt J 98-B(5):686–689. https://doi.org/10.1302/0301-620X.98B5.37350
Griffin XL, McArthur J, Achten J, Parsons N, Costa ML (2013) The warwick hip trauma evaluation one–an abridged protocol for the WHiTE one study. Bone Jt Res 2(10):206–209. https://doi.org/10.1302/2046-3758.210.2000183
O’Neill F, McGloughlin T, Lenehan B, Condon F, Coffey JC, Walsh M (2013) Influence of implant design on the method of failure for three implants designed for use in the treatment of intertrochanteric fractures: the dynamic hip screw (DHS), DHS blade and X-BOLT. Eur J Trauma Emerg Surg 39(3):249–255. https://doi.org/10.1007/s00068-013-0257-7
Costa ML et al (2018) Intramedullary nail fixation versus locking plate fixation for adults with a fracture of the distal tibia: the UK FixDT RCT. Health Technol Assess 22(25):1–148. https://doi.org/10.3310/hta22250
‘TheraCell: Technologies-Fiber Matrix TechnologyTM-Fiber AnchorTM’. https://www.theracellinc.com/product-fiberanchor.php. Accessed 19 Oct 2021
Walsh WR et al (2017) Critical size bone defect healing using collagen-calcium phosphate bone graft materials. PLoS ONE 12(1):e0168883. https://doi.org/10.1371/journal.pone.0168883
‘Solid Screw Fixation by Pedicle Screw-Based ProMISTM System Implants’, Premia Spine. https://premiaspine.com/promistm-system/pedicle-screw/. Accessed 19 Oct 2021
Schwartz Z et al (2008) Effect of micrometer-scale roughness of the surface of Ti6Al4V pedicle screws in vitro and in vivo. J Bone Jt Surg Am 90(11):2485–2498. https://doi.org/10.2106/JBJS.G.00499
‘Lateral Sacroiliac Joint Fusion-SIrosTM 3D Printed’, Genesys Spine. https://www.genesysspine.com/products/sacral/lateral-sacroiliac-joint-fusion-siros-3d-printed/. Accessed 20 Oct 2021
‘iFuse: The Triangle-Shaped Implant Designed Specifically for the SI Joint’, SI-BONE. https://si-bone.com/providers/solutions/ifuse. Accessed 20 Oct 2021
Berlin C, Patel P, Lieberman I, Shaffrey M, Buchholz A (2021) Robotic sacroiliac fixation technique for triangular titanium implant in adult degenerative scoliosis surgery: 2-dimensional operative video. Oper Neurosurg (Hagerstown) 21(6):E555–E556. https://doi.org/10.1093/ons/opab326
de Andrada Pereira B et al (2021) Biomechanics of a laterally placed sacroiliac joint fusion device supplemental to S2 alar-iliac fixation in a long-segment adult spinal deformity construct: a cadaveric study of stability and strain distribution. J Neurosurg Spine. https://doi.org/10.3171/2021.3.SPINE202175
de Andrada Pereira B et al (2021) Biomechanical effects of a novel posteriorly placed sacroiliac joint fusion device integrated with traditional lumbopelvic long-construct instrumentation. J Neurosurg Spine. https://doi.org/10.3171/2020.11.SPINE201540
Novák V, Wanek T, Hrabálek L, Stejskal P (2021) Minimally invasive sacroiliac joint stabilization. Acta Chir Orthop Traumatol Cech 88(1):35–38
Dale M, Evans J, Carter K, O’Connell S, Morgan H, Carolan-Rees G (2020) iFuse implant system for treating chronic sacroiliac joint pain: a NICE medical technology guidance. Appl Health Econ Health Policy 18(3):363–373. https://doi.org/10.1007/s40258-019-00539-7
Tran ZV, Ivashchenko A, Brooks L (2019) Sacroiliac joint fusion methodology-minimally invasive compared to screw-type surgeries: a systematic review and meta-analysis. Pain Phys 22(1):29–40
Jeong JH, Leasure JM, Park J (2018) Assessment of biomechanical changes after sacroiliac joint fusion by application of the 3-dimensional motion analysis technique. World Neurosurg 117:e538–e543. https://doi.org/10.1016/j.wneu.2018.06.072
Lindsey DP, Kiapour A, Yerby SA, Goel VK (2018) Sacroiliac joint stability: finite element analysis of implant number, orientation, and superior implant length. World J Orthop 9(3):14–23. https://doi.org/10.5312/wjo.v9.i3.14
Bornemann R et al (2017) Two-year clinical results of patients with sacroiliac joint syndrome treated by arthrodesis using a triangular implant system. Technol Health Care 25(2):319–325. https://doi.org/10.3233/THC-161272
Sturesson B, Kools D, Pflugmacher R, Gasbarrini A, Prestamburgo D, Dengler J (2017) Six-month outcomes from a randomized controlled trial of minimally invasive SI joint fusion with triangular titanium implants versus conservative management. Eur Spine J 26(3):708–719. https://doi.org/10.1007/s00586-016-4599-9
Bornemann R et al (2017) Diagnosis of patients with painful sacroiliac joint syndrome. Z Orthop Unfall 155(3):281–287. https://doi.org/10.1055/s-0042-124417
Bornemann R et al (2016) Clinical trial to test the iFuse implant system® in patients with sacroiliac joint syndrome: one year results. Z Orthop Unfall 154(6):601–605. https://doi.org/10.1055/s-0042-110207
Dengler J et al (2016) Referred leg pain originating from the sacroiliac joint: 6-month outcomes from the prospective randomized controlled iMIA trial. Acta Neurochir (Wien) 158(11):2219–2224. https://doi.org/10.1007/s00701-016-2953-7
Saavoss JD, Koenig L, Cher DJ (2016) Productivity benefits of minimally invasive surgery in patients with chronic sacroiliac joint dysfunction. Clinicoecon Outcomes Res 8:77–85. https://doi.org/10.2147/CEOR.S101607
Lindsey DP, Kiapour A, Yerby SA, Goel VK (2015) Sacroiliac joint fusion minimally affects adjacent lumbar segment motion: a finite element study. Int J Spine Surg 9:64. https://doi.org/10.14444/2064
Cher DJ, Reckling WC, Capobianco RA (2015) Implant survivorship analysis after minimally invasive sacroiliac joint fusion using the iFuse implant System(®). Med Devices (Auckl) 8:485–492. https://doi.org/10.2147/MDER.S94885
Schroeder JE, Cunningham ME, Ross T, Boachie-Adjei O (2014) Early results of sacro-iliac joint fixation following long fusion to the sacrum in adult spine deformity. HSS J 10(1):30–35. https://doi.org/10.1007/s11420-013-9374-4
Lindsey DP et al (2014) Evaluation of a minimally invasive procedure for sacroiliac joint fusion-an in vitro biomechanical analysis of initial and cycled properties. Med Devices (Auckl) 7:131–137. https://doi.org/10.2147/MDER.S63499
Gaetani P et al (2013) Percutaneous arthrodesis of sacro-iliac joint: a pilot study. J Neurosurg Sci 57(4):297–301
Sachs D, Capobianco R (2013) Minimally invasive sacroiliac joint fusion: one-year outcomes in 40 patients. Adv Orthop 2013:536128. https://doi.org/10.1155/2013/536128
Rudolf L (2013) MIS Fusion of the SI joint: does prior lumbar spinal fusion affect patient outcomes? Open Orthop J 7:163–168. https://doi.org/10.2174/1874325001307010163
Miller LE, Reckling WC, Block JE (2013) Analysis of postmarket complaints database for the iFuse SI Joint fusion system®: a minimally invasive treatment for degenerative sacroiliitis and sacroiliac joint disruption. Med Devices (Auckl) 6:77–84. https://doi.org/10.2147/MDER.S44690
Duhon BS, Cher DJ, Wine KD, Lockstadt H, Kovalsky D, Soo C-L (2013) Safety and 6-month effectiveness of minimally invasive sacroiliac joint fusion: a prospective study. Med Devices (Auckl) 6:219–229. https://doi.org/10.2147/MDER.S55197
‘SImpact®’, Life Spine. https://lifespine.com/simpact/. Accessed 21 Oct 2021
Funding
This research is supported by the Netherlands Organisation for Scientific Research (NWO), domain Applied and Engineering Sciences (TTW), project number 17553.
Author information
Authors and Affiliations
Contributions
Literature search was contributed by EdK, writing of the article was contributed by EdK. Medical context was contributed by EE, AE-T and GK. Revision of the article was contributed by EE, AE-T, AS and PB.
Corresponding author
Ethics declarations
Conflicts of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Kater, E.P., Sakes, A., Edström, E. et al. Beyond the pedicle screw–a patent review. Eur Spine J 31, 1553–1565 (2022). https://doi.org/10.1007/s00586-022-07193-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-022-07193-z