Abstract
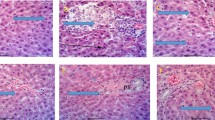
The aim of this study was to determine the subchronic effect of Crotalaria lachnosema Stapf. (Fabaceae) extract (MLECL) on biochemical and oxidative stress biomarkers in male Wistar rats. The rats were assigned into four groups designated as groups 1(control), 2, 3, and 4, of ten rats each, and were exposed to dose levels of 0, 40, 200, and 1000 mg of MLECL extract per kilogram in feed, respectively, for a period of 6 weeks. At the end of the study period, the surviving rats were sacrificed and blood was collected from each rat via jugular venesection and used for biochemical analysis. Postmortem examination was carried out on euthanized rats after which tissue sections of various organs were harvested for histopathological examination. Exposure to the extract was found to significantly (p < 0.0001) increased the values of urea and creatinine in groups 2, 3, and 4 (urea, 4.32 ± 0.15, 3.92 ± 0.32, 4.14 ± 0.09 mmol/l and creatinine, 68.60 ± 1.99, 89.40 ± 5.61, 71.60 ± 3.67 μmol/l, respectively) when compared with group 1 (urea, 2.62 ± 0.10 mmol/l, creatinine, 42.80 ± 1.59 μmol/l, respectively). Similarly, the extract was found to significantly affect the serum levels of liver enzymes only in group 4 (ALT, 52.80 ± 2.11 IU/L; AST, 49.40 ± 2.25 IU/L; ALP, 92.20 ± 8.24 IU/L), (p < 0.05) than the corresponding values in the control group (ALT, 45.00 ± 1.52 IU/L; AST, 38.00 ± 1.52 IU/L; ALP, 61.60 ± 6.43 IU/L). Exposure to the extract had variable effects on the levels of the oxidative stress biomarkers, Superoxide dismutase (SOD) and catalase (CAT). The SOD levels were significantly (p < 0.05) lower in groups 2 and 3 with mean values of 1.96 ± 0.12 and 1.80 ± 0.07 IU/L, respectively, when compared to the control value of 2.36 ± 0.08 IU/L. The serum CAT value (40.20 ± 1.24 IU/L) was significantly (p < 0.05) lower only in group 3, when compared to the control value (45.80 ± 1.16 IU/L). Microscopic lesions observed in the MLECL-exposed groups were hepatic megalocytosis and severe congestion in the kidneys and lungs. Results from this study showed variable toxic effects induced by MLECL administration to male Wistar rats, especially low dose exposure to rats in groups 2 and 3. This could be attributed to pyrrolizidine alkaloid in MLECL.







Similar content being viewed by others
References
Adaudi AO (2005) Studies on the toxicity and some pharmacological properties of methanolic extract of Tephrosiavogelii Hook (Fabaceae). Ph.D Thesis, Ahmadu Bello University, Zaria, pp 174
Arzt J, Mount ME (1999) Hepatotoxicity associated with pyrrolizidine alkaloid (Crotalaria species) ingestion in horse on Easter Island. Vet Hum Toxicol 41:96–99
Barros S, Ropke CD, Sawada TCH, Silva VVD, Pereira SMM, Barros SBDM (2005) Assessment of acute and subchronic oral toxicity of ethanolic extract of Pothomorpheumbellata L. Miq (Pariparoba). Braz J Pharma Sci 41(1):53–61
BEIR (1980) Biological effects of ionizing radiation. The effects on population of exposure to low levels of ionizing radiation. National Academy Press, Washington DC, pp 21–25
Bonda C, Soares JCM, Cecim M, Lopes STDA, Graça DL, Da Rocha RX (2005) Oxidative stress in the erythrocytes of cattle intoxicated with Senecio sp. Vet Clin Pathol 34(4):353–357
Bras G, Brooks SE, Watler DC (1961) Cirrhosis of the liver in Jamaica. J Pathol Bacteriol 82(2):503–512
Caisey JT, King DJ (1980) Clinical chemical values for some common laboratory animals. Clin Chem 26(13):1877–1879
Culvenor CCJ, Edgar JA, Smith LW (1981) Pyrrolizidine alkaloids in honey from Echiumplantagineum L. J Agric Food Chem 29:958–960
Deinzer ML, Thomson PA, Burgett DM, Isaacson DL (1977) Pyrrolizidine alkaloids: their occurrence in honey from tansy ragwort (Senecio jacobaea L.). Science 195:497–499
Dickinson JO, Cooke MP, King RR, Mohamed PA (1976) Milk transfer of pyrrolizidine alkaloids in cattle. J Am Vet Med Assoc 169:1192–1196
Fu PP, Xia Q, Lin G (2004) Pyrrolizidine alkaloids – genotoxicity, metabolism enzymes, metabolic activation, and mechanism. Drug Metab Rev 36:1–55
Hartmann T, Witte L (1995) Pyrrolizidine alkaloids: chemical, biological and chemoecological aspects. In: Pelletier SW (ed) Alkaloids: chemical and biological perspectives, vol 9. Pergamon Press, Oxford, pp 155–233
Holstege DM, Seiber JN, Galey FD (1995) Rapid multiresidue screen for alkaloids in plant material and biological samples. J Agric Food Chem 43:690–699
Ibrahim AJ, Makinde O, Ibekwe NN (2012) Pharmacognostic, physicochemical standardization and phytochemical analysis of leaves of cultivated Crotalaria lachnosemastapf. J App Pharm Sci 2(9):067–070
Langel D, Ober D, Pelser PB (2011) The evolution of pyrrolizidine alkaloid biosynthesis and diversity in the Senecioneae. Phytochem Rev 10:3–74
Levine BS (2002) Animal clinical pathology. In: Derelanko MJ, Hollinger MA (eds) Handbook of toxicology, 2nd edn. CRC Press, Boca Raton, pp 742–768
Luna LG (1960) In Manual of histologic staining of the Armed Forces Institute of Pathology, 94–95
Majak W, Brooke BM, Ogilvie RT (2008) Stock-poisoning plants of Western Canada. “Agriculture and Agric-food Green Cover Project”, Grey-wooded forage Association of Rocky Mountain House, AB., Canada, pp 75–76
Mattocks AR (1986) Chemistry and toxicology of Pyrrolizidine Alkaloids. Academic, London
Mattocks AR, Nwude N (1988) Pyrrolizidine alkaloids from Crotalaria lachnosema and Crotalaria naragutensis. Phytochemisty 27(10):3289–3291
McGavin MD, Zachary JF (2007) Pathologic basis of Veterinary disease fourth edition. Mosby Elsevier, Maryland Heights, pp 444–445
Nash RJ, Beaumont J, Veitch NC, Reynolds T, Benner J, Hughes CNG, Dring JV, Bennett RN, Dellar JE (1992) Phenylethylamine and piperidine alkaloids in Aloe species. PlantaMedica 58(1):84–87
Nuhu H, Abdulrrahman EM, Shok M (2009) Ethnobotanical studies on Crotalaria species found in Zaria, Northern Nigeria. Nig Journ Pharm Sci 8:46–53
OECD (2010) Organization for Economic Co-operation and Development. Draft guidance document No .116, on the design and conduct of chronic toxicity and carcinogenicity studies supporting TG 451,452,453, pp 3
Okopido IT, Ogunbiyi AO (1976) Investigation into the toxicity of Crotalaria retusa and C. lachnosema. Student Veterinaria, Zaria 7:6–7
Poli G, Leonarduzzi G, Biasi F, Chiarpotto E (2004) Oxidative stress and cell signalling. Curr Med Chem 11(9):1163–1182
Prakash AS, Pereira TN, Reilly EB, Seawright AA (1999) Pyrrolizidine alkaloids in human diet. Mutat Res 443:53–67
Singleton VL (1981) Naturally occurin food toxicants; phenolic substances of plant origin common in foods. Adv Food Res 27:149–221
Smith LW, Culvenor CCJ (1981) Plant sources of hepatotoxic pyrrolizidine alkaloids. J Nat Prod 44:129–152
Stewart MJ, Steenkamp V (2001) Pyrrolizidine poisoning: a neglected area in human toxicology. Ther Drug Monit 23(6):698–708
Trease GE, Evans WC (1983) Drugs of biological origin. In: Pharmacognosy, 12th edn. London, Balliere Tindall, pp 309–540
Wester PW, Krajanc EI, van der Heijden CA (1986) Chronic toxicity and carcinogenicity study with bisltri-n-bulyltin oxide (TBTO) in rats. In: toxicology and analytics of tributyltins, the present status. Proceedings ORTEP Association Berlin, pp 54–56
Woo J, Sigurdsson ST, Hopkins PB (1993) DNA Interstrand cross-linking reactions of pyrrol-derived bifunctional electrophiles: evidence for a common target site in DNA. J Am Chem Soc 115:3407–3415
Woodman DD (1996) Assessment of hepatotoxicity. In: Evans GO (ed) Animal clinical chemistry: a primer for toxicologist. Taylor and Francis, London, pp 71–86
World Health Organization (1978) Acute, subchronic and chronic test. In: principles and methods for evaluating the toxicity of chemicals. Part 1 Environmental Health Criteria, No. 6, pp 95–115
World Health Organization (1988) Report on pyrrolizidine alkaloids. International program on chemical safety. Environmental Health Criteria, No. 6, pp 75–83
World Health Organization (2000) General guidelines for methodologies on research and evaluation of traditional medicine. WHO Press, World Health Organization, Geneva
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baba, N.D., Uchendu, C. & Njoku, C.O.I. Effect of Crotalaria lachnosema Stapf. (Fabaceae) extract on biomarkers of hepatic and renal function and oxidative stress in male Wistar rats. Comp Clin Pathol 28, 1259–1266 (2019). https://doi.org/10.1007/s00580-019-02906-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-019-02906-1