Abstract
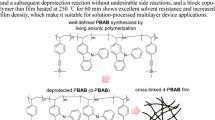
A new thiophene monomer design was demonstrated by exploiting the flexibility and bulkiness of trisiloxane units to produce “soft” polythiophene materials. In practice, the thiophene monomer having trisiloxane units could be polymerized by Kumada Catalyst-Transfer Polymerization (KCTP) to afford well-defined poly(1,1,1,3,3,5,5-heptametyl-5-(6′-(thien-3″-yl)hexyl)trisiloxane-2″,5″-diyl) (P3SiHT) with low dispersity, which was viscous material with fluidity. To the best of our knowledge, it is first time to develop “liquid” polythiophene at room temperature. In addition, we are successful in synthesizing novel all-conjugated hard-soft-hard ABA triblock copolymer, where A and B were poly(3-hexylthiophene) (P3HT) and P3SiHT, respectively, based on KCTP method and sequential monomer addition technique. The livingness of the KCTP system was confirmed by size exclusion chromatography (SEC) UV traces. The formation of phase separated P3HT and P3SiHT domains in the triblock copolymer (P3HT-b-P3SiHT-b-P3HT) was supported by differential scanning calorimetry (DSC), although the existence of miscible domains was also implied.








Similar content being viewed by others
References
Dou L, You J, Hong Z, Xu Z, Li G, Street RA, Yang Y (2013) 25th anniversary article: a decade of organic/polymeric photovoltaic research. Adv Mater 25:6642–6671. doi:10.1002/adma.201302563
Gather MC, Köhnen A, Meerholz K (2011) White organic light-emitting diodes. Adv Mater 23:233–248. doi:10.1002/adma.201002636
Guo Y, Yu G, Liu Y (2010) Functional organic field-effect transistors. Adv Mater 22:4427–4447. doi:10.1002/adma.201000740
Hammock ML, Chortos A, Tee BC-K, Tok JB-H, Bao Z (2013) 25th anniversary article: the evolution of electronic skin(E-Skin): a brief history, design considerations, and recent progress. Adv Mater 25:5997–6038. doi:10.1002/adma.201302240
Higashihara T, Goto E (2014) Controlled synthesis of low-polydisperse regioregular poly(3-hexylthiophene) and related materials by zincate-complex metathesis polymerization. Polym J 46:381–390. doi:10.1038/pj.2014.14
Higashihara T, Ueda M (2013) Precision synthesis of tailor-made polythiophene-based materials and their application to organic solar cells. Macromol Res 21:257–271. doi:10.1007/s13233-013-1123-5
Hirao A, Hayashi M, Higashihara T, Hadjichristidis N (2011) Recent synthetic developments in miktoarm star polymers with more than three different arms, in complex macromolecular architectures: synthesis, characterization, and self-assembly. In: Hadjichristidis N, Hirao A, Tezuka A, Prez F D (eds) John Wiley & Sons (Asia) Pte Ltd, Singapore. doi:10.1002/9780470825150.ch4
Kaltenbrunner M, White MS, Głowacki ED, Sekitani T, Someya T, Sariciftci NS, Bauer S (2012) Ultrathin and lightweight organic solar cells with high flexibility. Nature Commun 3:770. doi:10.1038/ncomms1772
Lessing J, Morin SA, Keplinger C, Tayi AS, Whitesides GM (2015) Stretchable conductive composites based on metal wools for use as electrical vias in soft devices. Adv Funct Mater 25:1418–1425. doi:10.1002/adfm.201403396
Lipomi DJ, Tee BC-K, Vosgueritchian M, Bao Z (2011) Stretchable organic solar cells. Adv Mater 23:1771–1775. doi:10.1002/adma.201004426
Lipomi DJ, Chongb H, Vosgueritchiana M, Meia J, Bao Z (2012) Toward mechanically robust and intrinsically stretchable organic solar cells: evolution of photovoltaic properties with tensile strain. Sol Energy Mater Sol Cells 107:355–365. doi:10.1016/j.solmat.2012.07.013
Mark JE (2004) Some interesting things about polysiloxanes. Acc Chem Res 37:946–953. doi:10.1021/ar030279z
Miyakoshi R, Yokoyama A, Yokozawa T (2008) Development of catalyst-transfer condensation polymerization. Synthesis of π-conjugated polymers with controlled molecular weight and low polydispersity. J Polym Sci Part A Polym Chem 46:753–765. doi:10.1002/pola.22389
Müller C, Goffri S, Breiby DW, Andreasen JW, Chanzy HD, Janssen RAJ, Nielsen MM, Radano CP, Sirringhaus H, Smith P, Stingelin-Stutzmann N (2007) Adv Funct Mater 17:2674–2679. doi:10.1002/adfm.200601248
O’Connor B, Chan EP, Chan C, Conrad BR, Richter LJ, Kline RJ, Heeney M, McCulloch I, Soles CL, DeLongchamp DM (2010) Correlations between mechanical and electrical properties of polythiophenes. ACS Nano 4:7538–7544. doi:10.1021/nn1018768
Peng R, Pang B, Hu D, Chen M, Zhang G, Wang X, Lu H, Choc K, Qiu L (2015) An ABA triblock copolymer strategy for intrinsically stretchable semiconductors. J Mater Chem C 3:3599–3606. doi:10.1039/c4tc02476a
Rogers JA, Someya T, Huang Y (2010) Materials and mechanics for stretchable electronics. Science 327:1603–1607. doi:10.1126/science.1182383
Savagatrup S, Makaram AS, Burke DJ, Lipomi DJ (2014) Mechanical properties of conjugated polymers and polymer-fullerene composites as a function of molecular structure. Adv Funct Mater 24:1169–1181. doi:10.1002/adfm.201302646
Sekitani T, Nakajima H, Maeda H, Fukushima T, Aida T, Hata K, Someya T (2009) Stretchable active-matrix organic light-emitting diode display using printable elastic conductors. Nature Mater 8:494–499. doi:10.1038/nmat2459
Sheina EE, Liu J, Iovu MC, Laird DW, McCullough RD (2004) Chain growth mechanism for regioregular nickel-Initiated cross-coupling polymerizations. Macromolecules 37:3526–3528. doi:10.1021/ma0357063
Wang J, Higashihara T (2013) Synthesis of all-conjugated donor–acceptor block copolymers and their application in all-polymer solar cells. Polym Chem 4:5518–5526. doi:10.1039/C3PY00979C
Wu H, Kustra S, Gates EM, Bettinger CJ (2013) Topographic substrates as strain relief features in stretchable organic thin film transistors. Org Electron 14:1636–1642. doi:10.1016/j.orgel.2013.02.037
Yokoyama A, Miyakoshi R, Yokozawa T (2004) Chain-growth polymerization for poly(3-hexylthiophene) with a defined molecular weight and a low polydispersity. Macromolecules 37:1169–1171. doi:10.1021/ma035396o
Yokozawa T, Yokoyama A (2009) Chain-growth condensation polymerization for the synthesis of well-defined condensation polymers and π-conjugated polymers. Chem Rev 109:5595–5619. doi:10.1021/cr900041c
Acknowledgments
This study was supported by KAKENHI (#26620172), Japan Society for the Promotion of Science (JSPS). S. Miyane thanks Innovative Flex Course for Frontier Organic Material Systems (iFront) at Yamagata University for him financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyane, S., Mori, H. & Higashihara, T. Synthesis and characterization of all-conjugated hard-soft-hard ABA triblock copolythiophenes. Microsyst Technol 22, 3–10 (2016). https://doi.org/10.1007/s00542-015-2627-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00542-015-2627-z