Abstract
Background
Gastrointestinal decompression is generally applied to a non-strangulated acute small bowel obstruction (NSASBO). Although long tube (LT) placement and administration of Gastrografin through a nasogastric tube (NGT-G) have shown advantages over NGT alone in previous studies, no studies appear to have compared LT and NGT-G.
Methods
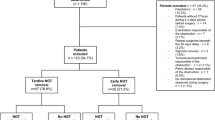
In this multicenter, randomized controlled trial, patients with NSASBO were randomly assigned to receive LT or NGT-G between July 2016 and November 2018 at 11 Japanese institutions. The primary endpoint was non-inferiority of NGT-G compared to LT for non-surgery rate, and the lower limit of the 95% confidence interval for the non-surgery rate (−15%) was set as the lower margin for inferiority of NGT-G compared to LT.
Results
In total, 223 patients (LT group, n = 111; NGT-G group, n = 112) were analyzed in the present trial. The non-surgery rate was 87.4% in the LT group and 91.1% in the NGT-G group, with a 3.7% difference between NGT-G and LT (95.3%CI − 5.55 to 12.91; non-inferiority P = 0.00002923). On the other hand, the non-surgery rate with pure NGT-G alone (76.8%) that represents non-cross-over NGT-G without subsequent LT was significantly lower than that with LT (P = 0.039). Median procedure time was significantly shorter with NGT-G (1 min) than with LT (25 min; P < 0.001), whereas no significant differences in mortality or hospital stay were noted between groups.
Conclusion
NGT-G is an effective alternative to LT as a first-line treatment for NSASBO. A sequential strategy comprising NGT-G followed by LT might offer a new standard for NSASBO.
Clinical trials registration
This trial was registered with the University Hospital Medical Information Network Clinical Trials Registry (umin.ac.jp/ctr Identifier: UMIN000022669) prior to the start of this trial.


Similar content being viewed by others
References
Ten Broek RPG, Krielen P, Di Saverio S, et al. Bologna guidelines for diagnosis and management of adhesive small bowel obstruction (ASBO): 2017 update of the evidence-based guidelines from the world society of emergency surgery ASBO working group. World J Emerg Surg. 2018;13:24.
Fevang BT, Jensen D, Svanes K, et al. Early operation or conservative management of patients with small bowel obstruction? Eur J Surg. 2002;168:475–81.
Fleshner PR, Siegman MG, Slater GI, et al. A prospective, randomized trial of short versus long tubes in adhesive small-bowel obstruction. Am J Surg. 1995;170:366–70.
Chen XL, Ji F, Lin Q, et al. A prospective randomized trial of transnasal ileus tube vs nasogastric tube for adhesive small bowel obstruction. World J Gastroenterol. 2012;18:1968–74.
Branco BC, Barmparas G, Schnuriger B, et al. Systematic review and meta-analysis of the diagnostic and therapeutic role of water-soluble contrast agent in adhesive small bowel obstruction. Br J Surg. 2010;97:470–8.
Burge J, Abbas SM, Roadley G, et al. Randomized controlled trial of Gastrografin in adhesive small bowel obstruction. ANZ J Surg. 2005;75:672–4.
Di Saverio S, Catena F, Ansaloni L, et al. Water-soluble contrast medium (gastrografin) value in adhesive small intestine obstruction (ASIO): a prospective, randomized, controlled, clinical trial. World J Surg. 2008;32:2293–304.
Haule C, Ongom PA, Kimuli T. Efficacy of Gastrografin((R)) compared with standard conservative treatment in management of adhesive small bowel obstruction at Mulago National Referral Hospital. J Clin Trials. 2013;3:4.
Farid M, Fikry A, El Nakeeb A, et al. Clinical impacts of oral gastrografin follow-through in adhesive small bowel obstruction (SBO). J Surg Res. 2010;162:170–6.
Ishizuka M, Shibuya N, Takagi K, et al. Gastrografin reduces the need for additional surgery in postoperative small bowel obstruction patients without long tube insertion: a meta-analysis. Ann Gastroenterol Surg. 2019;3:187–94.
Biondo S, Pares D, Mora L, et al. Randomized clinical study of Gastrografin administration in patients with adhesive small bowel obstruction. Br J Surg. 2003;90:542–6.
Ceresoli M, Coccolini F, Catena F, et al. Water-soluble contrast agent in adhesive small bowel obstruction: a systematic review and meta-analysis of diagnostic and therapeutic value. Am J Surg. 2016;211:1114–25.
Brochwicz-Lewinski MJ, Paterson-Brown S, Murchison JT. Small bowel obstruction–the water-soluble follow-through revisited. Clin Radiol. 2003;58:393–7.
Schulz KF, Altman DG, Moher D, et al. CONSORT 2010 statement: updated guidelines for reporting parallel group randomized trials. Ann Intern Med. 2010;152:726–32.
Scotte M, Mauvais F, Bubenheim M, et al. Use of water-soluble contrast medium (gastrografin) does not decrease the need for operative intervention nor the duration of hospital stay in uncomplicated acute adhesive small bowel obstruction? A multicenter, randomized, clinical trial (Adhesive Small Bowel Obstruction Study) and systematic review. Surgery. 2017;161:1315–25.
Ceresoli M, Coccolini F, Montori G, et al. Water-soluble contrast agent in adhesive small bowel obstruction: the game is still on. Surgery. 2017;162:199–200.
Kumar P, Kaman L, Singh G, et al. Therapeutic role of oral water soluble iodinated contrast agent in postoperative small bowel obstruction. Singapore Med J. 2009;50:360–4.
Keenan JE, Turley RS, McCoy CC, et al. Trials of nonoperative management exceeding 3 days are associated with increased morbidity in patients undergoing surgery for uncomplicated adhesive small bowel obstruction. J Trauma Acute Care Surg. 2014;76:1367–72.
Thornblade LW, Verdial FC, Bartek MA, et al. The safety of expectant management for adhesive small bowel obstruction: a systematic review. J Gastrointest Surg. 2019;23:846–59.
Schraufnagel D, Rajaee S, Millham FH. How many sunsets? Timing of surgery in adhesive small bowel obstruction: a study of the Nationwide Inpatient Sample. J Trauma Acute Care Surg. 2013;74:181–7.
Acknowledgments
We are grateful to all physicians and institutions who participated in the present study. We also thank Yumi Yoshida (Division of Endoscopy, Nagoya City University Hospital) for creating a computer-aided randomization system, Takako Onodera (Department of Gastroenterology and Metabolism, Nagoya City University Graduate School of Medical Sciences) for data management of this clinical trial, and Dr. Satoshi Ohsaga (Clinical Research Management Center, Nagoya City University Hospital) for statistical design and analysis. This study was supported, in part, by multicenter collaborative clinical research funding from the Japanese Gastroenterological Association (to T. Shimura) that had no such involvement to this trial.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Katano, T., Shimura, T., Nishie, H. et al. The first management using intubation of a nasogastric tube with Gastrografin enterography or long tube for non-strangulated acute small bowel obstruction: a multicenter, randomized controlled trial. J Gastroenterol 55, 858–867 (2020). https://doi.org/10.1007/s00535-020-01708-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-020-01708-5