Abstract
Background
The prognosis of autoimmune acute liver failure (ALF) without liver transplantation (LT) is poor worldwide. We subanalyzed infectious complications of autoimmune ALF using data of nationwide surveys between 2010 and 2015 retrospectively and tried to determine when to evaluate the efficacy of corticosteroid (CS) treatment or abandon it for LT based on objective data.
Methods
One hundred and forty-four patients with autoimmune ALF, comprising 79 ALF with coma ≤ I, 52 ALF with coma ≥ II and 13 late onset hepatic failure (LOHF), were analyzed.
Results
CS was administered to 140 (97%) patients. Thirty-seven (26%) patients had infectious complications. Patients with infection revealed more advanced disease type (p < 0.001) and poorer spontaneous survival (p < 0.001) than those without infection. Median (interquartile range) duration between diagnosis of ALF and onset of infection was 18.5 (11–36) days, and that between introduction of CS and onset of infection was 17 (10.5–36) days. Seventy-nine (55%) recovered without LT, 14 (10%) received LT and 51 (35%) died without LT. Dead or transplanted patients were older (p = 0.0057), and revealed more advanced liver failure (p < 0.001) and more occurrence of infection (p < 0.001).
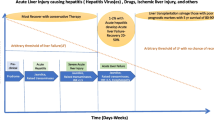
Conclusions
A critical point for evaluating the efficacy of CS treatment and switching to LT is at most 2-week after diagnosis of ALF and introduction of CS. More important, we should accelerate the point and prepare for LT in cases of ALF with coma ≥ II and LOHF, and we should have performed LT by then at the latest in case of failure to improve.


Similar content being viewed by others
Abbreviations
- AIH:
-
Autoimmune hepatitis
- ALF:
-
Acute liver failure
- LOHF:
-
Late onset hepatic failure
- CS:
-
Corticosteroid
- LT:
-
Liver transplantation
References
Czaja AJ. Autoimmune hepatitis: evolving concepts and treatment strategies. Dig Dis Sci. 1995;40:435–56.
Nikias GA, Batts KP, Czaja AJ. The nature and prognostic implications of autoimmune hepatitis with an acute presentation. J Hepatol. 1994;21:866–71.
Kessler WR, Cummings OW, Eckert G, et al. Fulminant hepatic failure as the initial presentation of acute autoimmune hepatitis. Clin Gastroenterol Hepatol. 2004;2:625–31.
Czaja AJ. Acute and acute severe (fulminant) autoimmune hepatitis. Dig Dis Sci. 2013;58:897–914.
Alvarez F, Berg PA, Bianchi FB, et al. International Autoimmune Hepatitis Group report: review of criteria for diagnosis of autoimmune hepatitis. J Hepatol. 1999;31:929–38.
Stravitz RT, Lefkowitch JH, Fontana RJ, et al. Autoimmune acute liver failure: proposed clinical and histological criteria. Hepatology. 2011;53:517–26.
Yasui S, Fujiwara K, Yonemitsu Y, et al. Clinicopathological features of severe and fulminant forms of autoimmune hepatitis. J Gastroenterol. 2011;46:378–90.
Fujiwara K, Yasui S, Tawada A, et al. Diagnostic value and utility of the simplified International Autoimmune Hepatitis Group criteria in acute onset autoimmune hepatitis. Liver Int. 2011;31:1013–20.
Onji M, Zeniya M, Yamamoto K, et al. Autoimmune hepatitis: diagnosis and treatment guide in Japan, 2013. Hepatol Res. 2014;44:368–70.
Manns MP, Czaja AJ, Gorham JD, et al. Diagnosis and management of autoimmune hepatitis. Hepatology. 2010;51:2193–213.
Lee WM, Squires RH Jr, Nyberg SL, et al. Acute liver failure: summary of a workshop. Hepatology. 2008;47:1401–15.
Fujiwara K, Mochida S, Matsui A, et al. Fulminant hepatitis and late onset hepatic failure in Japan. Hepatol Res. 2008;38:646–57.
Oketani M, Ido A, Nakayama N, et al. Etiology and prognosis of fulminant hepatitis and late-onset hepatic failure in Japan: summary of the annual nationwide survey between 2004 and 2009. Hepatol Res. 2013;43:97–105.
Nakao M, Nakayama N, Uchida Y, et al. Nationwide survey for acute liver failure and late-onset hepatic failure in Japan. J Gastroenterol. 2018;53:752–69.
Mochida S, Nakao M, Nakayama N, et al. Nationwide prospective and retrospective surveys for hepatitis B virus reactivation during immunosuppressive therapies. J Gastroenterol. 2016;51:999–1010.
Nakao M, Nakayama N, Uchida Y, et al. Deteriorated outcome of recent patients with acute liver failure and late-onset hepatic failure caused by infection with hepatitis A virus: a subanalysis of patients seen between 1998 and 2015 and enrolled in nationwide surveys in Japan. Hepatol Res. 2019;49:844–52.
Fujiwara K, Yasui S, Tawada A, et al. Autoimmune fulminant liver failure in adults. Experience in a Japanese center. Hepatol Res. 2011;41:133–41.
Fujiwara K, Yasui S, Yokosuka O. Autoimmune acute liver failure: the emerging etiology for acute liver failure. Hepatol Int. 2013;7:335–46.
Rolando N, Harvey F, Brahm J, et al. Prospective study of bacterial infection in acute liver failure: an analysis of fifty patients. Hepatology. 1990;11:49–53.
Rolando N, Harvey F, Brahm J, et al. Fungal infection: a common, unrecognised complication of acute liver failure. J Hepatol. 1991;12:1–9.
Yeoman AD, Westbrook RH, Zen Y, et al. Prognosis of acute severe autoimmune hepatitis (AS-AIH): the role of corticosteroids in modifying outcome. J Hepatol. 2014;61:876–82.
Mack Adams D, Assis DN, et al. Diagnosis and management of autoimmune hepatitis in adults and children: 2019 practice guidance and guidelines from the American Association for the Study of Liver Diseases. Hepatology. 2019. https://doi.org/10.1002/hep.31065(Epub ahead of print).
Fujiwara K, Yasui S, Yokosuka O. Letter: treatment of autoimmune acute liver failure beyond consensus guidelines. Aliment Pharmacol Ther. 2013;38:1143–4.
Fujiwara K, Yasui S, Yonemitsu Y, et al. Analysis of infectious complications and timing for emergency liver transplantation in autoimmune acute liver failure. J Hepatobiliary Pancreat Sci. 2016;23:212–9.
Mochida S, Takikawa Y, Nakayama N, et al. Diagnostic criteria of acute liver failure: a report by the Intractable Hepato-Biliary Diseases Study Group of Japan. Hepatol Res. 2011;41:805–12.
The Proceedings of the 12th Inuyama Symposium. Hepatitis Type A and Fulminant Hepatitis. Tokyo: Chugai Igaku-Sha, 1982.
Mochida S, Nakayama N, Ido A, et al. Revised criteria for classification of the etiologies of acute liver failure and late-onset hepatic failure in Japan: a report by the Intractable Hepato-biliary Diseases Study Group of Japan in 2015. Hepatol Res. 2016;46:369–71.
Yasui S, Fujiwara K, Okitsu K, et al. Importance of computed tomography imaging features for the diagnosis of autoimmune acute liver failure. Hepatol Res. 2012;42:42–50.
Fujiwara K, Oda S, Abe R, et al. On-line hemodiafiltration or high-flow continuous hemodiafiltration is one of the most effective artificial liver support devices for acute liver failure in Japan. J Hepatobiliary Pancreat Sci. 2015;22:246–7.
Fujiwara K, Abe R, Yasui S, et al. High recovery rate of consciousness by high-volume filtrate hemodiafiltration for fulminant hepatitis. Hepatol Res. 2019;49:224–31.
Abe M, Mashiba T, Zeniya M, et al. Present status of autoimmune hepatitis in Japan: a nationwide survey. J Gastroenterol. 2011;46:1136–41.
Joshita S, Yoshizawa K, Umemura T, Japan Autoimmune Hepatitis Study Group (JAIHSG), et al. Clinical features of autoimmune hepatitis with acute presentation: a Japanese nationwide survey. J Gastroenterol. 2018;53:1079–88.
Bernsmeier C, Antoniades CG, Wendon J. What’s new in acute liver failure? Intensive Care Med. 2014;40:1545–8.
Antoniades CG, Berry PA, Wendon JA, et al. The importance of immune dysfunction in determining outcome in acute liver failure. J Hepatol. 2008;49:845–61.
Czaja AJ. Difficult treatment decisions in autoimmune hepatitis. World J Gastroenterol. 2010;16:934–47.
Ichai P, Duclos-Vallée JC, Guettier C, et al. Usefulness of corticosteroids for the treatment of severe and fulminant forms of autoimmune hepatitis. Liver Transpl. 2007;13:996–1003.
Karkhanis J, Verna EC, Chang MS, et al. Steroid use in acute liver failure. Hepatology. 2014;59:612–21.
Fujiwara K, Hida S, Yasui S, et al. Corticosteroid might reduce serum levels of pro-inflammatory cytokines in fulminant hepatitis: a case series. Hepatol Res. 2018;48:106–12.
Zachou K, Arvaniti P, Azariadis K, et al. Prompt initiation of high-dose i.v. corticosteroids seems to prevent progression to liver failure in patients with original acute severe autoimmune hepatitis. Hepatol Res. 2019;49:96–104.
Czaja AJ. Corticosteroids or not in severe acute or fulminant autoimmune hepatitis: therapeutic brinksmanship and the point beyond salvation. Liver Transpl. 2007;13:953–5.
Stravitz RT, Lee WM. Acute liver failure. Lancet. 2019;394:869–81.
Rahim MN, Liberal R, Miquel R, et al. Acute severe autoimmune hepatitis: corticosteroids or liver transplantation? Liver Transpl. 2019;25:946–59.
Fujiwara K, Fukuda Y, Yokosuka O. Precise histological evaluation of liver biopsy specimen is indispensable for diagnosis and treatment of acute onset autoimmune hepatitis. J Gastroenterol. 2008;43:951–8.
Fujiwara K, Nakano M, Yasui S, et al. Advanced histology and impaired liver regeneration are associated with disease severity in acute onset autoimmune hepatitis. Histopathology. 2011;58:693–704.
Fujiwara K, Yasui S, Nakano M, et al. Severe and fulminant hepatitis of indeterminate etiology in a Japanese center. Hepatol Res. 2015;45:E141–9.
Fujiwara K, Yasui S, Yokosuka O, et al. Acute severe autoimmune hepatitis: corticosteroids or liver transplantation? Liver Transpl. 2019;25:1455–6.
Acknowledgements
We thank doctors for their cooperation to enroll patients (Supplementary Table 1). This study was performed with the support of the Ministry of Health, Labour and Welfare as an official project by the Intractable Hepato-Biliary Diseases Study Group of Japan.
Author information
Authors and Affiliations
Consortia
Contributions
All authors participated in the interpretation of study results, and in the drafting, critical revision, and approval of the final version of the manuscript. All are members of the Intractable Hepato-Biliary Diseases Study Group of Japan. NN and SM performed annual nationwide surveys and constructed the original database as official projects by the Intractable Hepato-Biliary Diseases Study Group of Japan. KF was involved in the data collection, extracted the data of autoimmune acute liver failure from the original database, analyzed data and wrote the manuscript. NK and OY were involved in the data collection and are co-workers of KF in the Study Group. Hirohito Tsubouchi was involved in the data collection and is the former Chair of the Intractable Hepato-Biliary Diseases Study Group of Japan, and Hajime Takikawa was involved in the data collection and is the present Chair of the Intractable Hepato-Biliary Diseases Study Group of Japan. SM was investigator in the study, involved in the data collection and is the Chair of the Fulminant Hepatitis Study Group, subgroup of the Intractable Hepato-Biliary Diseases Study Group of Japan.
Corresponding author
Ethics declarations
Conflicts of interest
Satoshi Mochida received lecture fees from Abbvie GK, Gilead Sciences Inc., MSD K.K., Ohtsuka Parmaceutical Co. Ltd., Bristol-Myers Squibb Co., Dainihon-Sumitomo Pharma Co. Ltd., Asuka Pharmaceutical Co. Ltd., Torey Medical, Asahikasei Pharma Co., Kyowa Hakko Kirin Co. Ltd., consigned/joint research expenses from Gilead Sciences Inc., EA Pharma Co., Ltd., Janssen Pharmaceutical K.K., Kowa Co. Ltd., MSD K.K., Abbvie GK, and scholarship donations from Abbvie GK, MSD K.K., Dainihon-Sumitomo Pharma Co. Ltd., Mochida Pharmaceutical Co. Ltd., Daiich Sankyo Co. Ltd., Torey Medical, Chugai Pharmaceutical Co., SRL inc., EA Pharma Co., Ltd., Japan Blood Products Organization. The other authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fujiwara, K., Nakayama, N., Kato, N. et al. Infectious complications and timing for liver transplantation in autoimmune acute liver failure in Japan: a subanalysis based on nationwide surveys between 2010 and 2015. J Gastroenterol 55, 888–898 (2020). https://doi.org/10.1007/s00535-020-01699-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-020-01699-3