Abstract
Background
Numerous biomarkers have been developed for assessing the presence and severity of liver fibrosis associated with non-alcoholic fatty liver disease (NAFLD). Fibrosis can be assessed by liver stiffness measurement (LSM) using vibration-controlled transient elastography (VCTE). Here we examined whether diagnostic accuracy and applicability can be further improved by combining various biomarker measurements with LSM.
Methods
A total of 278 patients with biopsy-confirmed Japanese NAFLD patients were enrolled. Area under the receiver operator characteristic curve (AUROC) was evaluated for obtaining the optimum interpretation criteria for LSM by VCTE and comparing various biomarkers alone and in combination with LSM.
Results
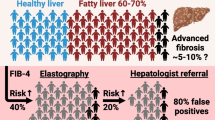
Liver stiffness measurements including cases with interquartile range (IQR)/median (M) < 30% or LSM ≤ 7.1 kPa demonstrated high applicability (90% of patients with NAFLD) and accuracy (AUROC: 0.891) for predicting stage ≥ 3 fibrosis. For all biomarkers tested, the AUROC values for predicting stage ≥ 3 fibrosis were increased when combined with LSM [platelet count, 0.734 vs. 0.912; type-4 collagen 7s (T4C7s), 0.894 vs. 0.921; aspartate aminotransferase to alanine aminotransferase ratio (AST/ALT), 0.774 vs. 0.906; AST to platelet ratio index, 0.789 vs. 0.902; FIB-4 index, 0.828 vs. 0.922; NAFLD fibrosis score, 0.800 vs. 0.906; CA index-fibrosis, 0.884 vs. 0.913; FM-fibro index, 0.920 vs. 0.943; FIB-4 index + T4C7s, 0.901 vs. 0.930], demonstrating the advantage of concurrent LSM.
Conclusions
While VCTE has slightly limited applicability (90%) for patients with NAFLD, concurrent measurement with certain biomarkers (especially FM-fibro, T4C7s, and FIB-4) greatly improves the diagnostic accuracy.


Similar content being viewed by others
Abbreviations
- AUROC:
-
The area under the receiver operator characteristic curve
- cfNRI:
-
Category-free net reclassification improvement
- IDI:
-
Integrated discrimination improvement
- IQR/M:
-
Interquartile range/median
- LSM:
-
Liver stiffness measurement
- NAFLD:
-
Non-alcoholic fatty liver disease
- NFS:
-
NAFLD fibrosis score
- NPV:
-
Negative predictive value
- PLT:
-
Platelet count
- PPV:
-
Positive predictive value
- T4C7s:
-
Type-4 collagen 7s
- VCTE:
-
Vibration-controlled transient elastography
References
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the study of liver diseases. Hepatology. 2018;67:328–57.
Angulo P, Kleiner DE, Dam-Larsen S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149(389–97):e10.
Kim D, Kim WR, Kim HJ, et al. Association between noninvasive fibrosis markers and mortality among adults with nonalcoholic fatty liver disease in the United States. Hepatology. 2013;57:1357–65.
Dulai PS, Singh S, Patel J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology. 2017;65:1557–65.
Yoshimura K, Okanoue T, Ebise H, et al. Identification of novel noninvasive markers for diagnosing nonalcoholic steatohepatitis and related fibrosis by data mining. Hepatology. 2016;63:462–73.
Okanoue T, Ebise H, Kai T, et al. A simple scoring system using type IV collagen 7s and aspartate aminotransferase for diagnosing nonalcoholic steatohepatitis and related fibrosis. J Gastroenterol. 2018;53:129–39.
Kwok R, Tse YK, Wong GL, et al. Systematic review with meta-analysis: non-invasive assessment of non-alcoholic fatty liver disease—the role of transient elastography and plasma cytokeratin-18 fragments. Aliment Pharmacol Ther. 2014;39:254–69.
Yoneda M, Imajo K, Takahashi H, et al. Clinical strategy of diagnosing and following patients with nonalcoholic fatty liver disease based on invasive and noninvasive methods. J Gastroenterol. 2018;53:181–96.
Younossi ZM, Loomba R, Anstee QM, et al. Diagnostic modalities for nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and associated fibrosis. Hepatology. 2018;68:349–60.
Vilar-Gomez E, Chalasani N. Non-invasive assessment of non-alcoholic fatty liver disease: clinical prediction rules and blood-based biomarkers. J Hepatol. 2018;68:305–15.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Matteoni CA, Younossi ZM, Gramlich T, et al. Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology. 1999;116:1413–9.
Kruger FC, Daniels CR, Kidd M, et al. Apri: a simple bedside marker for advanced fibrosis that can avoid liver biopsy in patients with Nafld/Nash. S Afr Med J. 2011;101:477–80.
Shah AG, Lydecker A, Murray K, et al. Comparison of noninvasive markers of fibrosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2009;7:1104–12.
Angulo P, Hui JM, Marchesini G, et al. The Nafld fibrosis score: a noninvasive system that identifies liver fibrosis in patients with Nafld. Hepatology. 2007;45:846–54.
Kanda Y. Investigation of the freely available easy-to-use software ‘Ezr’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Kanda Y. Free Statistical Software: Ezr (Easy R) on R Commander.
Hanley JA, McNeil BJ. The meaning and use of the area under a receiver operating characteristic (Roc) curve. Radiology. 1982;143:29–36.
DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44:837–45.
Pencina MJ, D’Agostino RB Sr, D’Agostino RB Jr, et al. Evaluating the added predictive ability of a new marker: from area under the roc curve to reclassification and beyond. Stat Med. 2008;27:157–72 (discussion 207–212).
Pencina MJ, D’Agostino RB Sr, Steyerberg EW. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat Med. 2011;30:11–21.
Boursier J, Zarski JP, de Ledinghen V, et al. Determination of reliability criteria for liver stiffness evaluation by transient elastography. Hepatology. 2013;57:1182–91.
Dietrich CF, Bamber J, Berzigotti A, et al. Efsumb guidelines and recommendations on the clinical use of liver ultrasound elastography, update 2017 (long version). Ultraschall Med. 2017;38:e16–47.
Singh S, Muir AJ, Dieterich DT, et al. American gastroenterological association institute technical review on the role of elastography in chronic liver diseases. Gastroenterology. 2017;152:1544–77.
Castera L, Foucher J, Bernard PH, et al. Pitfalls of liver stiffness measurement: a 5-year prospective study of 13,369 examinations. Hepatology. 2010;51:828–35.
European Association for Study of L, Asociacion Latinoamericana para el Estudio del H. Easl-Aleh clinical practice guidelines: non-invasive tests for evaluation of liver disease severity and prognosis. J Hepatol. 2015;63:237–64.
Sandrin L, Fourquet B, Hasquenoph JM, et al. Transient elastography: a new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med Biol. 2003;29:1705–13.
Arena U, Vizzutti F, Corti G, et al. Acute viral hepatitis increases liver stiffness values measured by transient elastography. Hepatology. 2008;47:380–4.
Millonig G, Reimann FM, Friedrich S, et al. Extrahepatic cholestasis increases liver stiffness (Fibroscan) irrespective of fibrosis. Hepatology. 2008;48:1718–23.
Xiao G, Zhu S, Xiao X, et al. Comparison of laboratory tests, ultrasound, or magnetic resonance elastography to detect fibrosis in patients with nonalcoholic fatty liver disease: a meta-analysis. Hepatology. 2017;66:1486–501.
Yoshioka K, Hashimoto S, Kawabe N. Measurement of liver stiffness as a non-invasive method for diagnosis of non-alcoholic fatty liver disease. Hepatol Res. 2015;45:142–51.
Guha IN, Parkes J, Roderick P, et al. Noninvasive markers of fibrosis in nonalcoholic fatty liver disease: validating the European liver fibrosis panel and exploring simple markers. Hepatology. 2008;47:455–60.
Rosenberg WM, Voelker M, Thiel R, et al. Serum markers detect the presence of liver fibrosis: a cohort study. Gastroenterology. 2004;127:1704–13.
Chen J, Yin M, Talwalkar JA, et al. Diagnostic performance of Mr elastography and vibration-controlled transient elastography in the detection of hepatic fibrosis in patients with severe to morbid obesity. Radiology. 2017;283:418–28.
Hsu C, Caussy C, Imajo K, et al. Magnetic resonance vs transient elastography analysis of patients with nonalcoholic fatty liver disease: a systematic review and pooled analysis of individual participants. Clin Gastroenterol Hepatol. 2018.
Cui J, Heba E, Hernandez C, et al. Magnetic resonance elastography is superior to acoustic radiation force impulse for the diagnosis of fibrosis in patients with biopsy-proven nonalcoholic fatty liver disease: a prospective study. Hepatology. 2016;63:453–61.
Imajo K, Kessoku T, Honda Y, et al. Magnetic resonance imaging more accurately classifies steatosis and fibrosis in patients with nonalcoholic fatty liver disease than transient elastography. Gastroenterology. 2016;150(626–37):e7.
Park CC, Nguyen P, Hernandez C, et al. Magnetic resonance elastography vs transient elastography in detection of fibrosis and noninvasive measurement of steatosis in patients with biopsy-proven nonalcoholic fatty liver disease. Gastroenterology. 2017;152(598–607):e2.
Petta S, Vanni E, Bugianesi E, et al. The combination of liver stiffness measurement and Nafld fibrosis score improves the noninvasive diagnostic accuracy for severe liver fibrosis in patients with nonalcoholic fatty liver disease. Liver Int. 2015;35:1566–73.
Boursier J, Vergniol J, Sawadogo A, et al. The combination of a blood test and fibroscan improves the non-invasive diagnosis of liver fibrosis. Liver Int. 2009;29:1507–15.
Ducancelle A, Leroy V, Vergniol J, et al. A single test combining blood markers and elastography is more accurate than other fibrosis tests in the main causes of chronic liver diseases. J Clin Gastroenterol. 2017;51:639–49.
Heo JY, Kim BK, Park JY, et al. Combination of transient elastography and an enhanced liver fibrosis test to assess the degree of liver fibrosis in patients with chronic hepatitis B. Gut Liver. 2018;12:190–200.
Boursier J, de Ledinghen V, Zarski JP, et al. A new combination of blood test and fibroscan for accurate non-invasive diagnosis of liver fibrosis stages in chronic hepatitis C. Am J Gastroenterol. 2011;106:1255–63.
Cales P, Boursier J, Ducancelle A, et al. Improved fibrosis staging by elastometry and blood test in chronic hepatitis C. Liver Int. 2014;34:907–17.
Voican CS, Louvet A, Trabut JB, et al. Transient elastography alone and in combination with Fibrotest((R)) for the diagnosis of hepatic fibrosis in alcoholic liver disease. Liver Int. 2017;37:1697–705.
Dincses E, Yilmaz Y. Diagnostic usefulness of fibrometer Vcte for hepatic fibrosis in patients with nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2015;27:1149–53.
Loong TC, Wei JL, Leung JC, et al. Application of the combined fibrometer vibration-controlled transient elastography algorithm in chinese patients with non-alcoholic fatty liver disease. J Gastroenterol Hepatol. 2017;32:1363–9.
Myers RP, Pomier-Layrargues G, Kirsch R, et al. Feasibility and diagnostic performance of the Fibroscan Xl probe for liver stiffness measurement in overweight and obese patients. Hepatology. 2012;55:199–208.
de Ledinghen V, Wong VW, Vergniol J, et al. Diagnosis of liver fibrosis and cirrhosis using liver stiffness measurement: comparison between M and Xl probe of Fibroscan(R). J Hepatol. 2012;56:833–9.
Vuppalanchi R, Siddiqui MS, Van Natta ML, et al. Performance characteristics of vibration-controlled transient elastography for evaluation of nonalcoholic fatty liver disease. Hepatology. 2018;67:134–44.
Funding
This research is supported by AMED under Grant Number JP19fk0210040 (T.O.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T. Shima has received research funding from Bristol-Myers Squibb. All authors have no conflicts of interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shima, T., Sakai, K., Oya, H. et al. Diagnostic accuracy of combined biomarker measurements and vibration-controlled transient elastography (VCTE) for predicting fibrosis stage of non-alcoholic fatty liver disease. J Gastroenterol 55, 100–112 (2020). https://doi.org/10.1007/s00535-019-01626-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-019-01626-1