Abstract
Purpose
To investigate the effectiveness of physiotherapy interventions compared to control conditions on fecal incontinence (FI) and quality of life (QoL) following colorectal surgery.
Methods
Electronic searches in English-language (Scopus, Web of Science, Embase, AMED, CENTRAL, CINAHL, MEDLINE, Ovid, and PEDro) and Chinese-language (CNKI, Wanfang Data) databases were conducted. Trials comparing physiotherapy interventions against control conditions and assessing FI and QoL outcomes were included in the review.
Results
Ten trials were included. Meta-analysis revealed statistically significant improvements in lifestyle (0.54; 95% CI 0.03, 1.05; p = 0.04), coping behavior (MD 1.136; 95% CI 0.24, 2.04; p = 0.01), and embarrassment (0.417; 95% CI 0.14, 0.70; p = 0.00) components of QoL among individuals receiving pelvic floor muscle training (PFMT) compared with those receiving usual care (UC). Meta-analysis showed biofeedback to be significantly more effective than UC in enhancing anal resting pressure (ARP; 9.551; 95% CI 2.60, 16.51; p = 0.007), maximum squeeze pressure (MSP; 25.29; 95% CI 4.08, 48.50; p = 0.02), and rectal resting pressure (RRP; 0.51; 95% CI 0.10, 0.9; p = 0.02). Meta-analysis also found PFMT combined with biofeedback to be significantly more effective than PFMT alone for ARP (3.00; 95% CI 0.40, 5.60; p = 0.02), MSP (9.35, 95% CI 0.17, 18.53; p = 0.05), and RRP (1.54; 95% CI 0.60, 2.47; p = 0.00).
Conclusions
PFMT combined with biofeedback was more effective than PFMT alone, but both interventions delivered alone were superior to UC. Future studies remain necessary to optimize and standardize the PFMT parameters for improving QoL among individuals who experience FI following CRC surgery.
Review registration
This systematic review is registered in the PROSPERO registry (Ref: CRD42022337084).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Colorectal cancer (CRC) is more common among men than women, and as of 2020, CRC had a 5-year prevalence rate of 51.9 per 100,000 people in Asia and an incidence rate of 68 per 100,000 in Hong Kong [1, 2]. Surgical methods are commonly applied to remove tumors from the colon or rectum, including low anterior resection (LAR), laparoscopy, abdominoperineal resection, and transanal microsurgery. However, fecal incontinence (FI), defined as the “involuntary loss of feces when feces are solid and/or liquid,” commonly develops following surgical management of CRC and chemoradiation [3]. Following surgery, FI can develop due to damage to muscular, fascial, or neural tissues during surgery [4]. Seventy to 90% of the patients who undergo sphincter preserving surgery experience FI in addition to other symptoms such as incontinence for flatus, increased intestinal gas, and rectal urgency (commonly referred to known as LAR syndrome) [5]. The reported incidence of FI in patients treated with pelvic chemoradiation ranges between 3 and 53% [6, 7]. FI has a significant negative impact on the quality of life (QoL) among CRC survivors [8]. The inability to control the involuntary leakage of stool can cause embarrassment and fear of such episodes may hinder social participation or physical activity, adversely affecting mental health [9]. Therefore, the post-surgical management of FI is necessary.
Treatments for FI include both pharmaceutical and non-pharmaceutical interventions. Non-pharmaceutical treatments may include diet adjustments, anal plugs, or physiotherapy [10, 11]. Physiotherapy interventions for the treatment of FI include pelvic floor muscle training (PFMT) with or without biofeedback, neuromuscular electrical stimulation (NMES), and acupuncture [12]. A recent review by Kim and Oh [13] evaluated the effectiveness of PFMT on bowel function and health-related QoL among patients who have undergone LAR and included studies published until 2019. Since then, further studies have been published on physiotherapy interventions in the management of FI, and therefore, the review requires updating. The objective of this review is to investigate the effectiveness of physiotherapy interventions on FI and the QoL following colorectal surgery.
Methods
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [14] and was registered in the International Prospective Register of Systematic Reviews (PROSPERO, CRD42022337084).
Search strategy
Electronic databases of English-language articles, including the Allied and Complementary Medicine Database (AMED), The Cochrane Central Register of Controlled Trials (CENTRAL), the Cumulative Index of Nursing and Allied Health (CINAHL), Embase, MEDLINE, Ovid, the Physiotherapy Evidence Database (PEDro), Scopus, and Web of Science, and electronic databases of Chinese-language articles, including Chinese National Knowledge Infrastructure (CNKI) and Wanfang Data, were searched to identify potentially relevant articles. Searches were conducted from database inception to December 2021 and updated in November 2022. Searches were conducted by two independent review authors (P.C. and C.T.C). The specific search strategy applied to the Medline database is presented in Supplementary Appendix 1. Four themes, “physiotherapy intervention,” “colorectal cancer and surgery,” “bowel incontinence,” and “randomized controlled trials,” were utilized to identify potentially relevant articles. Related terms associated with each theme were combined using the Boolean operator “OR.” All four themes were combined using the Boolean operator “AND.” EndNote 20 citation management software was used to archive and organize the search results and remove duplicates. Manual searches were also performed by examining the reference lists for each included trial and relevant systematic reviews to identify additional candidate trials.
Study eligibility criteria
Trials were included if they (1) were randomized controlled trials (RCTs, including pilot, cluster, or crossover trials) comparing physiotherapy interventions (acupuncture, biofeedback, electrical stimulation, aerobic exercises, resistance exercises, stretching exercises, manual therapy, PFMT, or yoga) with a control condition (no treatment, usual care [UC], placebo, or active control) on FI and QoL outcomes following colorectal surgery; (2) measured FI using the Cleveland Clinic Incontinence Score (CCIS, also known as the Wexner score), the low anterior resection syndrome (LARS) score, or anorectal manometry (ARM) or measured QoL using the Fecal Incontinence Quality of Life (FIQL) scale, the EuroQoL 5-Dimension (EQ-5D) scale, the European Organization for Research and Treatment of Cancer (EORTC), Quality of Life Questionnaire (QLQ) module for colorectal cancer (QLQ-CR29), the EORTC QLQ module for cancer (QLQ-C30), or the 36-Item Short Form Survey (SF-36); and (3) were available in full-text format in either English or Chinese (traditional or simplified). Unpublished theses were also included in the review if they met the review criteria described above. Studies were excluded if they were systematic reviews, RCT/systematic review protocols, or case reports. Trials that included subjects with FI associated with causes other than colorectal surgery or radiotherapy for CRC (such as labor, prostate cancer, malabsorption, spinal surgery, or congenital disorders) were excluded. Trials reporting data as median and interquartile range, trials that included subjects with FI secondary to other medical conditions, such as neurological diseases, and trials that evaluated the effectiveness of non-physiotherapy treatments (such as anal plugs, Chinese medication, herb medication, injection, acupressure, moxibustion) were also excluded.
Article screening
Articles identified through the electronic searches underwent a three-stage screening process, including title, abstract, and full-text screening. Studies were screened for inclusion by two reviewers (P.M.Y. and L.P.H.). Disagreements between reviewers were resolved by discussion, and a third reviewer (P.K.) was consulted if the disagreement remained unresolved after discussion.
Data extraction
Data extraction was conducted independently by three authors (P.C., P.K., and C.K.H). The following data were extracted from each included study: last name of the first author, publication year, country of origin, sample size, mean age/age range of study subjects, intervention and control, outcome measures, and pre- and post-treatment data (mean and standard deviation) of ARM measured by anal resting pressure (ARP) and maximum squeeze pressure (MSP) for FI, and lifestyle, coping behavior, depression, and embarrassment components of QoL. For post-treatment data, only the data associated with the longest follow-up period was extracted.
Quality assessment
Five authors (P.C., P.K., C.K.H., P.M.Y., and L.P.H) independently assessed the methodological quality of the included trials using the Revised Cochrane Risk-of-Bias (RoB 2) tool for randomized trials [15]. Disagreements between reviewers were resolved by discussion. An additional reviewer (PK) was consulted for any unresolved disagreements. The RoB 2 categorizes the overall RoB level as high, moderate, or low, based on the RoB identified in five domains: (1) randomization process, (2) deviation from intended interventions, (3) missing outcome data, (4) outcome measurements, and (5) the selection of the reported results [15]. The overall RoB evaluation was guided by signaling questions in the five domains [15].
Data synthesis and statistical analysis
All meta-analyses were performed using Comprehensive Meta-Analysis software (CMA version 3.3.070, Biostat Inc., Englewood, NJ, USA). Statistical heterogeneity was measured using the I2 test. A random-effect model was applied for high heterogeneity (I2 > 50%); otherwise, a fixed-effect model was utilized [16]. Trials evaluating similar physiotherapy interventions, control conditions, and outcome measures were grouped together for meta-analysis. For continuous data, the weighted mean difference (WMD) and 95% confidence interval (CI) were calculated. WMD was chosen because similar units of measurement were used for outcome measures in the included trials [17]. In the meta-analyses and throughout the “Results” section, all data for ARP and MSP reported by Chen [18] were converted from mmHg to kPa (by multiplying by 0.133) [19] to standardize the unit for the calculation of WMD. Significance was defined as a p-value ≤ 0.05.
Results
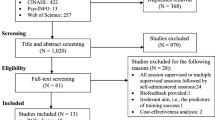
The study selection process, which followed the PRISMA approach, is summarized in Fig. 1. The studies that were excluded at the full-text screening stage and the reasons for exclusion are listed in Supplementary Appendix 2. A total of 4413 articles were identified via electronic and manual searches. After all screening stages were applied, only 10 trials met the inclusion criteria for the meta-analytic review.
Characteristics of the included trials
Table 1 summarizes the characteristics of the included trials. Across all 10 trials, data were extracted for 608 subjects. The sample sizes of the included trials ranged from 12 to 100. The mean age of participants in the included trials ranged from 45.6 to 66.8 years. All subjects in the included studies had fecal incontinence following surgery for CRC. The interventions assessed in the included trials were PFMT (n = 2), biofeedback (n = 4), and biofeedback combined with PFMT (n = 4). Among the 10 included trials, three trials reported QoL outcomes, and eight trials reported FI outcomes.
Risk of bias in the included trials
Figure 2A shows the distributions of RoB levels across each domain, and Fig. 2B shows the overall RoB levels assessed for all included trials. Of the 10 included trials, three (30%) reported adequate randomization processes, one (10%) had few to no deviations from the intended intervention, six (60%) were free of missing outcome data, seven (70%) blinded the outcome assessors, and eight (80%) had reported all planned outcomes. Of the 10 included trials, four had moderate RoB, and six had high RoB.
Effects of interventions on QoL among individuals with FI after CRC surgery
PFMT versus UC
Two trials compared the effects of PFMT with those of UC on QoL [20, 21]. The RoB for these two trials was high. Both trials [20, 21] measured QoL using the FIQL and reported the lifestyle, coping behavior, depression, and embarrassment components. PFMT was performed daily in both trials, but the protocols varied. In Hung, Lin (20), participants performed four PMFT sessions per day, with each session consisting of 20 contractions. In Xia et al. [21], participants performed three PMFT sessions per day, with each session consisting of 20 contractions. The duration of PFMT ranged from 3 to 9 months. Meta-analysis of data from the two trials [20, 21] (n = 112) revealed significant effects of the intervention compared with UC for the lifestyle (WMD 0.54; 95% CI 0.03, 1.05; p = 0.04; Fig. 3A), coping behavior (WMD 1.14; 95% CI 0.24, 2.04; p = 0.01; Fig. 3B), and embarrassment (WMD 0.417; 95% CI 0.14, 0.70; p = 0.00; Fig. 3C) components of the FIQL; however, no significant effect was observed for PFMT compared with UC on the depression component (WMD 0.424; 95% CI − 0.24, 1.09; p = 0.21; Fig. 3D).
Effect of interventions on FI
Biofeedback versus UC
Three trials compared the effects of biofeedback with those of UC on FI, measured by ARM [22,23,24]. The RoB for these three trials ranged from moderate to high. ARP and MSP were reported in all three trials, but only two trials reported RRP [22, 24]. In all three trials [22,23,24], electromyography (EMG) biofeedback therapy was implemented after colorectal surgery. One [22] of the three trials used an anal electrode inserted into the lower rectum, with adhesive electrodes placed on the external oblique muscles, forming a circuit to enable the detection of muscle activities during bowel movements. In this trial, daily biofeedback therapy, provided for 45–60 min per session, was performed for 15 days. Two trials [23, 24] provided insufficient descriptions of the method used to detect electrical activity during bowel movements or the treatment parameters. The meta-analysis of all three trials [23, 24] (n = 226) demonstrated a significant effect for biofeedback compared with UC for improving ARP (WMD 9.55; 95% CI 2.59, 16.51; p = 0.01; Fig. 4A) and MSP (WMD 25.29; 95% CI 4.08, 48.50; p = 0.02; Fig. 4B). The meta-analysis of the two trials reporting RRP [22, 24] (n = 152) found a significant effect for biofeedback compared with UC on RRP improvement (WMD 0.51; 95% CI 0.10, 0.92; p = 0.02; Fig. 4C).
PFMT plus biofeedback versus PFMT
The effects of PFMT alone compared with the effects of PFMT combined with biofeedback on anorectal dynamics were examined in three trials [18, 25, 26]. The RoB of these three trials varied from moderate to high. All three trials [25, 26] reported ARP and MSP as outcomes, but only two trials [25, 26] reported RRP. Chen (18) and Zheng, Wu (26) included EMG biofeedback therapy in addition to PFMT. The electrical activities of the pelvic floor muscles were measured by anal electrodes in all three trials. Zheng, Wu (26) also used adhesive electrodes to detect the electrical activity of external oblique muscles. Biofeedback therapy was performed two to three times per week for 20–30 min each time, with the total training period ranging from 3 to 13 months. In the trials by Chen (18) and Zheng, Wu (26), PFMT was performed daily, consisting of five sets of 10 repetitions consisting of contractions lasting 5–10 s per repetition, with 10 s of rest between repetitions. Subjects in the study by Yang, Wang (25) performed PFMT by contracting pelvic floor muscles for 10 s. The total PMFT training period varied from 16 months to the time subjects needed to be discharged [18, 25, 26]. The meta-analysis of the three trials [18, 25, 26] (n = 211) showed a significant effect for biofeedback combined with PFMT compared with PMFT alone on ARP (WMD 3.00; 95% CI 0.40, 5.59; p = 0.02, Fig. 5A) and MSP (WMD 9.35; 95% CI 0.17, 18.53; p = 0.05, Fig. 5B). The meta-analysis of the two trials reporting RRP [25, 26] (n = 135) revealed a significant effect for biofeedback combined with PMFT compared with PMFT alone on RRP (WMD 1.54; 95% CI 0.60, 2.48; p = 0.00, Fig. 5C).
Biofeedback versus PFMT
The effects of biofeedback therapy alone were compared with the effects of PFMT alone on FI using the CCIS in two trials [27, 28]. The RoB for the two trials varied from moderate to high. Both trials provided biofeedback therapy for the experimental group, whereas the control group performed Kegel exercises. Cho, Kim (28) provided the experimental group with biofeedback training designed to strengthen their external anal sphincter. Subjects were educated to slowly contract and relax the pelvic floor muscles and were presented with visual or audible signals proportional to their anal squeezing pressure. The training was provided one to two times per week for 6 months. Kim, Jeon (27) provided the experimental group with biofeedback training in which an anorectal probe was used to train subjects to achieve adequate squeeze pressure using a visual feedback display. Each training session lasted 10 to 30 min, and subjects were encouraged to repeat the exercises five times each day. The PFMT dosage used for the control groups was not specified in either trial [27, 28]. The meta-analysis of data from both trials [27, 28] (n = 168) showed a non-significant effect for biofeedback alone compared with PFMT alone on the CCIS (WMD 0.49; 95% CI − 1.68, 2.66; p = 0.66; Fig. 6).
Discussion
This systematic review and meta-analysis investigated the effectiveness of physiotherapy interventions on FI and QoL following colorectal surgery. The literature searches identified 4413 potentially relevant articles indexed in both English- and Chinese-language databases; however, only 10 trials met the pre-defined inclusion criteria for the meta-analysis. The interventions examined in the included trials were PFMT alone, biofeedback therapy alone, and the combination of PFMT and biofeedback therapy. The RoB of the included trials ranged from moderate to high.
Meta-analysis of data from two trials [20, 21] comparing PFMT alone with UC revealed a significant effect of the intervention on QoL components, including lifestyle, coping behavior, and embarrassment, as measured using the FIQL. The minimal clinically important difference (MCID) values reported for lifestyle, coping behavior, and embarrassment are 0.2, 0.3, and 0.2, respectively, among the noncancerous population [29]. The mean estimated effects obtained in the studies included in the current review surpassed the MCID for lifestyle (0.54), coping behavior (1.14), and embarrassment (0.43), indicating that these effects might be clinically meaningful. Despite the significant results obtained for QoL in the current review, the findings are limited by the high RoB, the limited number of pooled trials (n = 2), and the varying PFMT protocols used by each included study. Nevertheless, considering the effect size and safety of PFMT [30], this intervention should be considered a potential treatment option for improving QoL among individuals who experience FI following CRC surgery.
Meta-analysis of data from trials with moderate to high RoB comparing biofeedback alone with UC revealed significant effects of the intervention on the ARP, MSP [22,23,24], and RRP [22, 24] measures of ARM. Meta-analysis of data from trials [18, 25, 26] with moderate to high RoB identified similar significant effects on FI parameters when PFMT combined with biofeedback was compared with PFMT alone. ARM is a non-invasive procedure used to objectively quantify anorectal function and has been found to be clinically relevant for assessing the severity of FI in both children and adults [31,32,33]. No MCID has been established for ARM, preventing interpretation of the estimated effect size. However, the 95% CI values for both interventions (PFMT plus biofeedback and biofeedback alone) were below the MCID, indicating the potential for clinically trivial effects. Additional data examining the effects of these interventions would narrow the 95% CI and provide more precise estimates of the average effects of PFMT plus biofeedback and biofeedback alone for the treatment of FI following CRC surgery.
The results obtained in the current study for PFMT plus biofeedback agree with results reported in previous systematic reviews [34, 35] examining the effects of pelvic floor rehabilitation, including PFMT plus biofeedback, on improving anorectal function following rectal resection surgery. However, these prior systematic reviews [34, 35] did not include quantitative analyses, and both reviews included non-RCTs. By contrast, the current review quantitatively evaluated the efficacy of PFMT plus biofeedback and only included RCTs, offering a higher level of evidence [36].
Although the current review found significant effects for biofeedback alone and PFMT plus biofeedback on FI as measured by ARM, these findings are limited by a considerably high RoB, large variations in the PFMT and biofeedback protocols described in the included trials, and the small number of trials included in the meta-analysis. However, considering the non-invasive nature of PFMT and the minimally invasive nature of biofeedback, both PFMT combined with biofeedback and biofeedback alone should be considered potential interventions for improving FI following CRC surgery. Future studies should investigate additional ARM other than ARP, MSP, and RRP, such as urge volume and volume of first sensation [37], to obtain a more holistic understanding of the effects of these interventions on FI.
Meta-analysis of data from two trials [27, 28] comparing biofeedback alone with a PMFT alone revealed a non-significant effect of the intervention on FI following colorectal surgery. Based on these results, no recommendations can be made regarding the effectiveness of biofeedback alone compared with PFMT alone. Future studies of high methodological rigor are required to confirm the results obtained in this review for biofeedback alone compared with PFMT alone.
Strengths and limitations of the review
The current review has several strengths, including being the first review to include meta-analyses evaluating the effectiveness of various physiotherapy interventions on FI and QoL following CRC surgery. A comprehensive search strategy was applied to identify RCTs evaluating the effectiveness of various physiotherapy interventions for the treatment of FI following CRC surgery. Meta-analyses revealed significant effects for PFMT alone, biofeedback alone, and the combination of PFMT with biofeedback for improving QoL and FI following CRC surgery.
Our review also has some limitations. More than half of the included studies (6 of 10) were identified as having a high RoB. Other limitations are the inclusion of unpublished theses, which might hinder the study quality because unpublished studies may be of lower methodological quality than published studies [38], heterogeneity in terms of PFMT and biofeedback protocols utilized in the included trials, which minimizes the applicability of these findings to clinical settings; a high degree of statistical heterogeneity was evident across the pooled estimates as indicated by large I2 values, small sample sizes in some of the included trial;, and the small number of trials included in meta-analyses.
Conclusion
The current systematic review and meta-analysis identified significant improvements in the lifestyle, coping behavior, and embarrassment components of the FIQL among patients who received PFMT compared with those who received UC. Considering the non-invasive nature of PFMT and the sizes of the effects obtained for this intervention on different QoL components, PFMT should be considered an intervention that may improve QoL among individuals who experience FI following CRC surgery. Meta-analysis revealed that biofeedback alone was superior to UC and that PFMT plus biofeedback was superior to PFMT alone, with both superior interventions resulting in significant improvements in ARP, MSP, and RRP when assessed using ARM. Biofeedback is a minimally invasive intervention that can be applied alone or in combination with PFMT to treat FI following CRC surgery. However, the efficacy of biofeedback alone compared with PFMT alone remains inconclusive. Future high-quality RCTs remain necessary to standardize and optimize PFMT and biofeedback parameters for FI rehabilitation following CRC surgery and to confirm the results obtained in this review.
References
Wong MC, Ding H, Wang J, Chan PS, Huang J (2019) Prevalence and risk factors of colorectal cancer in Asia. Intest Res 17(3):317–329
Hong Kong Cancer Registry, Hospital Authority (2021) Top ten common cancers in Hong Kong in 2021. Retrieved Oct 2021, from https://www3.ha.org.hk/cancereg/tc/topten.html
D’Ancona C, Haylen B, Oelke M, Abranches-Monteiro L, Arnold E, Goldman H et al (2019) The International Continence Society (ICS) report on the terminology for adult male lower urinary tract and pelvic floor symptoms and dysfunction. Neurourol Urodyn 38(2):433–477
Chan K, Suen M, Coulson S, Vardy JL (2021) Efficacy of pelvic floor rehabilitation for bowel dysfunction after anterior resection for colorectal cancer: a systematic review. Support Care Cancer 29(4):1795–1809
Pieniowski E, Nordenvall C, Palmer G, Johar A, TumlinEkelund S, Lagergren P et al (2020) Prevalence of low anterior resection syndrome and impact on quality of life after rectal cancer surgery: population-based study. BJS open 4(5):935–942
CJH cjh van_de_velde@ lumc. nl cciotDTMEtLMdDMBEMCPKRHKKEMCvdV (2007) Risk factors for faecal incontinence after rectal cancer treatment. J Br Surg 94(10):1278–84
Bentzen AG, Guren MG, Vonen B, Wanderås EH, Frykholm G, Wilsgaard T et al (2013) Faecal incontinence after chemoradiotherapy in anal cancer survivors: long-term results of a national cohort. Radiother Oncol 108(1):55–60
Whitehead W, Borrud PH, Goode PS, Meikle S, Mueller R, Tuteja A et al (2009) Fecal incontinence in U.S. Adults: epidemiology and risk factors for the pelvic floor disorders network. National health and nutrition examination survey (NHANES) of fecal incontinence: Prevalence by sex, age, race, marital status, education, and income. Gastroenterology 137(2):512–7
Rockwood TH, Church JM, Fleshman JW, Kane RL, Mavrantonis C, Thorson AG, Wexner SD, Bliss D, Lowry AC (2000) Fecal Incontinence Quality of Life Scale: quality of life instrument for patients with fecal incontinence. Dis Colon Rectum 43:9–16
Norton C, Kamm MA (2001) Anal plug for faecal incontinence. Colorectal Dis 3(5):323–327
Norton C, Whitehead W, Bliss D, Harari D, Lang J (2010) Management of fecal incontinence in adults. Neurourol Urodyn: Off J Int Continence Soc 29(1):199–206
Bates F, Bliss DZ, Bardsely A, Yeung WKW (2018) Management of fecal incontinence in community-living adults. Springer, Management of fecal incontinence for the advanced practice nurse, pp 93–126
Kim YM, Oh EG (2023) Effectiveness of pelvic floor muscle training for patients following low anterior resection: a systematic review and meta-analysis. J Wound Ostomy Continence Nurs 50(2):142–150
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6(7):e1000100-e
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I et al (2019) RoB 2: A revised tool for assessing risk of bias in randomised trials. Br Med J 366:l4898-1
Borenstein M, Hedges LV, Higgins JPT, Rothstein HR (2010) A basic introduction to fixed-effect and random-effects models for meta-analysis. Res Synth Methods 1(2):97–111
Khan S (2020) Meta-analysis of weighted mean difference. In: Khan S (ed) Meta-analysis: methods for health and experimental studies. Springer Singapore, Singapore, pp 195–216
Chen MF (2021) Effect of biofeedback training combined with pelvic floor muscle training in promoting the recovery of anorectal function in patients with middle and low rectal cancer after sphincter preservation surgery. Med J Chin People’s Health 33(18):81–82
Thompson A, Taylor BN (2008) Guide for the use of the international system of units (SI). NIST Special Publication, Gaithersburg
Hung S-L, Lin Y-H, Yang H-Y, Kao C-C, Tung H-Y, Wei L-H (2016) Pelvic floor muscle exercise for fecal incontinence quality of life after coloanal anastomosis. J Clin Nurs 25(17–18):2658–2668
Xia B, Zeng Y, Zhang Q (2016) Application value of Kegel pelvic floor muscle exercises combined with nursing intervention in the treatment of anterior resection syndrome. China Med Herald 13(34):178–181
Zhang CZ, Zhang YL, WZ F, (2016) Kinetics after defecation based biofeedback to lower colorectal cancer between internal and external anal sphincter resection. Chin J Surg Integrated Traditional Western Med 22:235–238
Chai R, Ni X, Chen S, Wan Z, Chen B, Tu S (2018) Study on anorectal dynamics of biofeedback therapy for fecal incontinence in patients with low rectal cancer after restorative resection. Chin J Exp Surg 35(2):226–229
You S, Guan Y (2018) Evaluation of anal function after anus preserving operation for rectal cancer and the research of comprehensive treatment for promoting recovery of anal function. Anhui Med Pharm J 22(9):1747–1751
Yang J-m, Wang S-x, Wang Z-x (2020) Effect of biofeedback training combined with pelvic floor muscle exercise on anal canal function and rectal function in patients with middle and low rectal cancer. J Clin Nurs 19(5):51–53
Zheng M-C, Wu X-D, Jiang W, Wen Y-S, Yang X, Pan Z-Z (2019) Effects of biofeedback training on prevention of anterior resection syndrome in rectal cancer patients underwent anus-preserving surgery. Chin J Nurs 54(07):1032–1037
Kim JK, Jeon BG, Song YS, Seo MS, Kwon YH, Park JIW et al (2015) Biofeedback therapy before ileostomy closure in patients undergoing sphincter-saving surgery for rectal cancer: a pilot study. Ann Coloproctol 31(4):138–143
Cho HM, Kim H, Yoo R, Kim G, Kye BH (2021) Effect of biofeedback therapy during temporary stoma period in rectal cancer patients: a prospective randomized trial. J Clin Med 10(21):5172
Tejedor P, Bodega-Quiroga I, Plaza J, Ortega-López M, Gutiérrez C, García-Olmo D et al (2019) Quality of life and 3D-EUS assessment for anal incontinence after childbirth. Rev Esp Enferm Dig 111(6):453–459
Boyle R, Hay-Smith EJ, Cody JD, Mørkved S (2012) Pelvic floor muscle training for prevention and treatment of urinary and faecal incontinence in antenatal and postnatal women. Cochrane Database Syst Rev 10:Cd007471
Senel E, Demirbag S, Tiryaki T, Erdogan D, Cetinkursun S, Cakmak O (2007) Postoperative anorectal manometric evaluation of patients with anorectal malformation. Pediatr Int 49(2):210–214
Keshtgar AS, Athanasakos E, Clayden GS, Ward HC (2008) Evaluation of outcome of anorectal anomaly in childhood: the role of anorectal manometry and endosonography. Pediatr Surg Int 24(8):885–892
Bjørsum-Meyer T, Christensen P, Jakobsen M, Baatrup G, Qvist N (2020) Correlation of anorectal manometry measures to severity of fecal incontinence in patients with anorectal malformations–a cross-sectional study. Sci Rep 10(1):1–8
Visser WS, Te Riele WW, Boerma D, Van Ramshorst B, Van Westreenen HL (2014) Pelvic floor rehabilitation to improve functional outcome after a low anterior resection: a systematic review. Ann Coloproctol 30(3):109
Maris A, Devreese AM, D’Hoore A, Penninckx F, Staes F (2013) Treatment options to improve anorectal function following rectal resection: a systematic review. Colorectal Dis 15(2):e67-78
Burns PB, Rohrich RJ, Chung KC (2011) The levels of evidence and their role in evidence-based medicine. Plast Reconstr Surg 128(1):305–310
Lee TH, Bharucha AE (2016) How to perform and interpret a high-resolution anorectal manometry test. J Neurogastroenterol Motil 22(1):46–59
Shuster J (2011) Review: cochrane handbook for systematic reviews for interventions, version 5.1.0, published 3/2011. In: Higgins JPT, Green S (eds) Res Synth Methods, vol 2, pp. 126–130
Acknowledgements
The research team acknowledges Mr. Muhammad Usman Ali for his assistance and support.
Funding
Open access funding provided by The Hong Kong Polytechnic University.
Author information
Authors and Affiliations
Contributions
P.K. contributed to the study’s conception and design.
P.C. and C.T.C performed electronic and hand searches.
P.M.Y. and L.P.H. completed the study screening.
P.C., P.K., and C.K.H performed data extraction. All authors conducted the risk of bias assessment and read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pun, M.Y., Leung, P.H., Chan, T.C. et al. The effectiveness of physiotherapy interventions on fecal incontinence and quality of life following colorectal surgery: a systematic review and meta-analysis of randomized controlled trials. Support Care Cancer 32, 103 (2024). https://doi.org/10.1007/s00520-023-08294-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-08294-1