Abstract
Purpose
Adjuvant endocrine therapy reduces the recurrence and mortality of early hormone receptor-positive breast cancer in both pre- and postmenopausal women. The aim of this study was to investigate adjuvant tamoxifen adherence and associated factors in breast cancer survivors.
Methods
This descriptive, prospective study was conducted in 2019–2020 with the participation of 531 women who survived breast cancer and were under follow-up at the Senology Institute of a hospital in Istanbul. Inclusion criteria were having completed treatment for early hormone receptor-positive breast cancer, being prescribed tamoxifen, and being 18 years or older. Data were collected using a patient information form and the Morisky Medication Adherence Scale-8 (MMAS-8).
Results
The mean age of the participants was 44.9 ± 6.5 years, and the mean duration of tamoxifen use was 834.4 ± 685.7 days. The women’s mean MMAS-8 score was 6.86 ± 1.39. Medication adherence was significantly positively correlated with current age (p = 0.006) and age at diagnosis (p = 0.002). There was a statistically significant difference between tamoxifen adherence according to participants’ employment status (p = 0.028), chronic disease status (p = 0.018), loss of libido (p = 0.012), treatment-related changes in mood changes (p = 0.004), and having negative effects affecting daily life (p < 0.001).
Conclusion
Overall, breast cancer survivors in this study reported moderate adherence to tamoxifen. The women’s individual characteristics and the adverse effects of treatment influenced medication adherence. Healthcare professionals can help increase adherence to this treatment, which reduces the risk of mortality, by explaining the importance of the medication, identifying and eliminating barriers to adherence, and informing women about evidence-based interventions to increase medication compliance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Treatment with adjuvant endocrine therapy (AET) reduces breast cancer recurrence and improves overall survival in patients with hormone receptor-positive breast cancer [1, 2]. Standard AETs used for hormone receptor-positive breast cancers include selective estrogen receptor modulators (SERMs) (e.g., tamoxifen) and aromatase inhibitors (AIs) (e.g., anastrozole, letrozole, and exemestane), which inhibit estrogen synthesis [3]. To obtain the desired therapeutic benefit, it is recommended to use these treatments for 5 to 10 years, depending on the patient’s condition and risk of recurrence [2]. The risk of breast cancer recurrence and mortality is high in patients who do not adhere to or prematurely discontinue treatment [4, 5]. Despite its known efficacy, non-adherence is reported to be common among women receiving AET [6].
Negative treatment-related experiences are the main factor influencing patients’ medication adherence [7, 8]. Adverse effects associated with include hot flushes, depression, sexual dysfunction, weight gain, musculoskeletal symptoms, bone problems, cognitive dysfunction, and fatigue [9, 10]. The persistence of adverse effects over the long-term results in impaired quality of life and reduced treatment adherence [11]. In premenopausal women, ovarian function suppression increases the adverse effects of AET. Premenopausal women undergoing tamoxifen therapy with ovarian function suppression were found to have significantly increased hot flushes and night sweats [12]. In a study conducted in premenopausal women with breast cancer, 16% of women prescribed tamoxifen were found to be noncompliant with treatment in biochemistry tests performed 1 year later, and it was also determined that these women had worse cancer-related outcomes [13]. Although postmenopausal women are reported to have lower rates of non-adherence, impaired quality of life and subsequent treatment discontinuation has been observed [14, 15]. Patients who are not adequately informed about the adverse effects of hormone therapy are more likely to discontinue treatment early [8]. In one qualitative study, women who prematurely discontinued AET had more negative beliefs about the effectiveness of AET, perceived the continuation of AET as a barrier to normalization of their lives, and stated they valued quality over length of life. These women also reported having less support from health care professionals than women who continued treatment [16].
Defining the factors that influence adherence to AET may promote treatment continuity. Ensuring medication adherence is a key point in maintaining long-term survival in breast cancer [17,18,19]. The aim of this study was to investigate adherence to adjuvant tamoxifen and associated factors in women breast cancer survivors.
Methods
Study design
This was a prospective and descriptive study.
Sample selection and patient characteristics
The study data were collected in the Senology Institute of a hospital in Istanbul, Turkey between the years 2019 and 2020. The sample consisted of 531 women over 18 years of age using adjuvant tamoxifen. Patients with neurological and psychological problems were not included in the study.
Data collection tools
Data were collected using the Morisky Medication Adherence Scale-8 (MMAS-8) and a patient information form prepared by the researchers. Women were invited to participate in the study when they presented to the breast outpatient clinic for follow-up visits or came to the hospital for treatment. The data were collected in face-to-face interviews with participating women.
Patient information form: This form was prepared by the researchers and consisted of two parts: sociodemographic characteristics (5 questions) and disease-related characteristics (13 questions).
Morisky medication adherence scale: This scale was first developed by Morisky et al. in 1986 as a 4-item scale and was later supported with additional items addressing the conditions surrounding adherence behavior. Morisky et al. also performed the validity and reliability analyses of the MMAS-8 [20], and Sayiner et al. conducted the validity and reliability study of the Turkish version [21]. The MMAS-8 consists of 7 closed-ended items with two options (yes/no) and 1 closed-ended question with five options. In the first 7 items, responses associated with greater adherence are scored as 1 point, while responses associated with poorer adherence receive 0 points. Item 8 uses a Likert-type scale of frequency in which the response “never/rarely” is scored 1 point and the other four response options are given 0 points. Patients with an MMAS-8 score of ≥ 6 are considered treatment adherent [21]. In the sample of this study, the Cronbach’s alpha value of the scale was calculated as 0.680.
Statistical analysis
Statistical analyses were performed using R version 2.15.3 software (R Core Team, 2013). Data were reported as minimum, maximum, mean, standard deviation, median, interquartile range (IQR; first quartile–third quartile), frequency, and percentage. Quantitative data were evaluated for normal distribution using Shapiro–Wilk test and graphical analysis. Independent groups t test was used for between-group comparisons of normally distributed variables. Mann–Whitney U test was used for between-group comparisons of non-normally distributed variables. Comparisons between more than two groups were done using one-way analysis of variance (ANOVA) for normally distributed variables and Kruskal–Wallis test for non-normally distributed variables. Pearson correlation analysis was used to evaluate correlations between quantitative variables. The KR-20 (Kuder-Richardson 20) coefficient was used to assess internal consistency. Statistical significance was accepted at p < 0.05.
Results
The women ranged in age from 27 to 82, with a mean age of 44.92 ± 6.50 years. Of the women, 63.8% (n = 339) held an undergraduate or postgraduate degree and 59.3% (n = 315) were employed (Table 1).
Their age at breast cancer diagnosis ranged from 24 to 76, with a mean of 42.26 ± 6.51 years. The mean duration of tamoxifen use was 834.4 ± 685.72 days (Table 1).
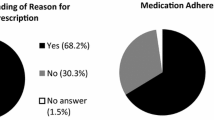
The mean total MMAS-8 score was 6.86 ± 1.39. There was a statistically significant positive correlation between tamoxifen adherence levels and current age (r = 0.119, p = 0.006) and age at diagnosis (r = 0.137, p = 0.002) (Table 1). Tamoxifen adherence increased as the age of women increased also tamoxifen adherence was better for women diagnosed at a later age.
MMAS-8 scores were not correlated with the women’s education level, who they lived with, or duration of tamoxifen use (p > 0.05) (Table 1).
However, MMAS-8 scores differed significantly according to employment status (p = 0.028). Women who were actively working had lower adherence than retired women and homemakers (p = 0.043 and p = 0.034, respectively) (Table 1).
At least one chronic disease was present in 16.9% (n = 90) of the women. The most common chronic diseases were hypertension in 3.4% (n = 18) and diabetes in 3% (n = 16). Those with chronic comorbidity had higher adherence (p = 0.018) (Table 1).
Most women (n = 503, 94.7%) reported adverse effects related to tamoxifen. Of the women, 68.9% (n = 366) reported hot flushes, 49.5% (n = 263) reported mood swings, 48.8% (n = 259) reported night sweats, 45.4% (n = 241) reported changes in weight, and 40.1% (n = 213) reported loss of libido. Tamoxifen adherence was significantly lower in women who reported loss of libido and mood swings as tamoxifen-related side effects (p = 0.012 and p = 0.004, respectively) (Table 2).
Of the women, 41.4% (n = 213) stated that the adverse effects of tamoxifen considerably affected their daily lives. These women had significantly lower tamoxifen adherence than those who stated that the adverse effects of treatment did not affect or only somewhat affected their lives (p < 0.001 and p < 0.001, respectively) (Table 2).
In the majority of women (n = 312, 58.8%), the extent of disease was limited to the breast. Almost all of the women (n = 529, 99.6%) had undergone surgical treatment, 57.6% (n = 306) had completed chemotherapy, 74.2% (n = 394) completed radiotherapy, and 12.8% (n = 68) completed targeted medication therapies. There were no significant differences in MMAS-8 scores according to extent of disease and metastasis status of the patients (p > 0.05) (Table 2).
Of the women who participated in the study, 53.9% (n = 286) regularly used at least one medication other than tamoxifen. Most women (n = 423, 79.7%) used tamoxifen at a dose of 20 mg once a day. There was no significant difference in tamoxifen adherence scores based on whether the women had completed treatment, were under ongoing treatment, were using medications other than tamoxifen, or their tamoxifen dose (p > 0.05) (Table 2).
Discussion
The aim of this study was to investigate adjuvant tamoxifen adherence and associated factors in breast cancer survivors. The mean MMAS-8 score in our sample was 6.86 ± 1.39. An MMAS score of ≥ 6 is defined as medication-compliant. Therefore, it can be said that the patients’ scores were at the lower limit of adherence. He et al. (2015) found that among patients who started AET, 14% had discontinued treatment by the end of the first year and 36% by the end of the third year. At 5 years after AET was prescribed, the rate of discontinuation was reported to be close to 54% [7]. Similarly, a systematic review showed that 5-year discontinuation rates were between 31 and 73% [22]. As mentioned earlier, one study showed that 16% of premenopausal women with breast cancer were noncompliant with tamoxifen therapy at 1 year after initiation and had poorer cancer-related outcomes [13]. Despite the high risk of recurrence and death, treatment non-adherence is seen to be common among breast cancer survivors.
There are many factors that influence treatment adherence to tamoxifen [7]. In our study, these factors included the women’s age, the age at diagnosed with cancer, their employment status, comorbidities, and the adverse effects of treatment.
We observed that adherence to tamoxifen increased with the women’s age. This suggests that tamoxifen adherence is more difficult in young women. A study conducted in Japan also showed that women over the age of 40 had higher treatment compliance [23].
In a study conducted in Sweden, it was reported that apart from the adverse effects of treatment, poorer adherence was associated with the presence of comorbidity and being younger than 40 or older than 64 years of age at diagnosis [7]. In our study, age at diagnosis was positively correlated with medication adherence scores, leading to the conclusion that women over 40 years of age had better adherence. However, in contrast to the findings of He (2015) [7], we observed that tamoxifen adherence was higher among women with chronic diseases. Comorbidity also leads to the use of multiple drugs. Therefore, these patients may take tamoxifen regularly along with the other medications taken to manage their chronic diseases. In our study, most women used the full dose of the tamoxifen at one time (20 mg/day), a small number of women took the tamoxifen dose by dividing into two doses per a day (10 mg twice daily). Taking the dose in full or divided were not change the effectiveness of the treatment. This varies according to the promotion of the patient and the physician.
In our study, the age of tamoxifen use was in a wide range (27–82 years). Physicians seem to prefer tamoxifen especially in postmenopausal women, since the side effects of anti-aromatase drugs are quite high. Although the side effects of tamoxifen were considered to be less, 94.7% of women experienced tamoxifen-related adverse effects. Women who stated that these side effects had a considerable effect on their daily lives had lower treatment adherence than those who reported no or less pronounced impact. Patients and oncologists perceive adverse effects to be a major deterrent to AET adherence [16]. Side effects are strongly associated with adherence to adjuvant therapy, and the mitigation and control of adverse effects is one way to increase adherence rates. In a survey of 622 postmenopausal women, 30% of patients discontinued hormone therapy, with a very high proportion of these patients (84%) citing adverse effects as the reason for discontinuation [24]. In one study, it was reported that the rate of AET discontinuation was higher among survivors who used symptom-relieving medications [7].
It was stated that hot flushes, not receiving chemotherapy, and not undergoing mastectomy were risk factors for discontinuing treatment. Demographic variables such as age and education level were not significant factors in treatment discontinuation [25].
It is known that AETs that inhibit estrogen production in women can lead to sexual dysfunction by worsening the symptoms associated with menopause (low libido, vaginal dryness and pain) [26]. In our study, it was found that women who reported mood swings and loss of libido had lower medication adherence scores. In a study conducted with 167 breast cancer survivors, 72% reported psychological effects and 34.7% reported sexual dysfunction. In the same study, the frequency of psychological disorders was shown to correlate with the severity of sexual dysfunction, and these problems were found to reduce the women’s quality of life [27]. This may explain why in our study, women who reported mood swings and loss of libido had similarly low medication adherence, given the correlation between these factors. In addition, as stated in the study by Pumo et al. (2012), these two factors may reduce treatment adherence because they impair women’s quality of life [27].
In our study, actively working women had lower medication adherence scores than women who did not work or were retired. This suggests that the active social and professional lives of younger women may negatively affect their adherence to medication. Younger age was also associated with lower adherence in AET in a systematic review of studies on women with breast cancer using AET [28].
Strengths and limitations
This study has several limitations. Firstly, it was a single-center study. The data collection period also overlaps with the COVID-19 pandemic. Fewer women came to the hospital during this period. In addition, this study did not include an evaluation of the women’s qualitative opinions influencing their medication adherence or objective parameters such as drug level in the blood. As the study data were cross-sectional, we could not identify patients who prematurely discontinued treatment before the start of the study or after the study data were obtained.
Conclusion
The women participating in this study were at the lower limit of medication adherence. Factors found to be associated with medication adherence were the women’s current age, age at cancer diagnosis, employment status, presence of chronic disease, and treatment-related adverse effects. Of these adverse effects, loss of libido and mood swings were identified as factors that reduced the women’s medication adherence.
To increase adherence to adjuvant tamoxifen, which reduces the risk of recurrence and mortality in breast cancer survivors, nurses should be aware of the individual factors affecting medication adherence and inform women about evidence-based interventions that promote treatment adherence. Multiple strategies can be implemented to ensure medication adherence, such as telephone checks, pill containers, and analyzing when prescriptions are filled. Changing the medication time (from morning to night) to reduce adverse effects and using email or message reminders are other options. Patients should also be educated on how to manage the adverse effects of treatment using pharmacological and nonpharmacological methods.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Early Breast Cancer Trialists’ Collaborative Group (2015) Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. The Lancet Lancet Publishing Group 386:1341–1352
Early Breast Cancer Trialists’ Collaborative Group (2011) Relevance of breast cancer hormone receptors and other factors to the effi cacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378:771–784
Ekinci E, Nathoo S, Korattyil T, Vadhariya A, Zaghloul HA, Niravath PA et al (2018) Interventions to improve endocrine therapy adherence in breast cancer survivors: what is the evidence? J Cancer Surviv 12:348–356
Hershman DL, Shao T, Kushi LH, Buono D, Yann Tsai W, Fehrenbacher L et al (2011) Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat 126:529–537
He W, Eriksson L, Törnberg S, Strand F, Czene K (2019) Discontinuation of adjuvant hormone therapy among breast cancer patients not previously attending mammography screening. BMC Med 17:1–9
Paranjpe R, John G, Trivedi M, Abughosh S (2019) Identifying adherence barriers to oral endocrine therapy among breast cancer survivors. Breast Cancer Res Treat 174:297–305
He W, Fang F, Varnum C, Eriksson M, Hall P, Czene K (2015) Predictors of discontinuation of adjuvant hormone therapy in patients with breast cancer. Journal of Clinical Oncology. World J Clin Oncol 33:2262–9
Kahn KL, Schneider EC, Malin JL, Adams JL, Epstein AM (2007) Patient centered experiences in breast cancer: predicting long-term adherence to tamoxifen use. Med Care 45:431–439
Berkowitz MJ, Thompson CK, Zibecchi LT, Lee MK, Streja E, Berkowitz JS et al (2021) How patients experience endocrine therapy for breast cancer: an online survey of side effects, adherence, and medical team support. J Cancer Surviv 15:29–39
Amir E, Seruga B, Niraula S, Carlsson L, Ocaña A (2011) Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst. J Natl Cancer Inst 103:1299–309
Ferreira AR, Di Meglio A, Pistilli B, Gbenou AS, El-Mouhebb M, Dauchy S et al (2019) Differential impact of endocrine therapy and chemotherapy on quality of life of breast cancer survivors: a prospective patient-reported outcomes analysis. Annals of Oncology Elsevier Ltd 30:1784–1795
Bernhard J, Luo W, Ribi K, Colleoni M, Burstein HJ, Tondini C, et al Adjuvant exemestane versus tamoxifen in premenopausal women with early breast cancer undergoing ovarian suppression: patient-reported outcomes in the TEXT and SOFT randomised trials
Pistilli B, Paci A, Ferreira AR, Antonio Meglio D, Poinsignon V et al (2020) Serum detection of nonadherence to adjuvant tamoxifen and breast cancer recurrence risk. J Clin Oncol 38:2762–72
Howard-Anderson J, Ganz PA, Bower JE, Stanton AL (2012) Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst. J Natl Cancer Inst 104:386–405
Ziller V, Kalder M, Albert US, Holzhauer W, Ziller M, Wagner U et al (2009) Adherence to adjuvant endocrine therapy in postmenopausal women with breast cancer. Annals of Oncology Elsevier 20:431–436
Brett J, Boulton M, Fenlon D, Hulbert-Williams J, Walter FM, Donnelly P, et al (2018) Adjuvant endocrine therapy after breast cancer: a qualitative study of factors associated with adherence. Patient Prefer Adherence. 12–291
Graetz I, Mckillop CN, Stepanski E, Vidal GA, Anderson JN, Schwartzberg LS (2018) Use of a web-based app to improve breast cancer symptom management and adherence for aromatase inhibitors: a randomized controlled feasibility trial. J. Cancer Surviv. 12:431–40
Farias AJ, Du XL (2017) Racial differences in adjuvant endocrine therapy use and discontinuation in association with mortality among Medicare breast cancer patients by receptor status. Cancer Epidemiology. Biomarkers & Prevention. NIH Public Access 26:1266–75
Heery M, Corbett P, Zelkowitz R (2018) Precautions for patients taking tamoxifen. J Adv Pract Oncol 9:78–83
Morisky DE, Ang A, Krousel-Wood M, Ward HJ (2008) Predictive validity of a medication adherence measure in an outpatient setting. The Journal Of Clinical Hypertension 10:348–354
Sayiner ZA, Savas E, Kul S, Morisky DE (2020) Validity and reliability of the Turkish version of the 8-ıtem morisky medication adherence scale in patients with type 2 diabetes. European Journal of Therapeutics Cordus 26:47–52
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat [Internet]. NIH Public Access; 2012 [cited 2022 Sep 5];134:459. Available from: /pmc/articles/PMC3607286/
Taketani K, Tokunaga E, Yamashita N, Tanaka K, Akiyoshi S, Okada S et al (2014) The early discontinuation of adjuvant hormone therapy is associated with a poor prognosis in Japanese breast cancer patients. Surgery Today: Official Journal of the Japan Surgical Society 44:1841–1846
Salgado VAB, Zivian M, Salgado-neglet B (2007) Aromatase inhibitor: side effects reported by 622 women. Cancer Res Treat. 168
Kemp A, Preen DB, Saunders C, Boyle F, Bulsara M, Malacova E et al (2014) Early discontinuation of endocrine therapy for breast cancer: who is at risk in clinical practice? Springerplus 3:1–10
Franzoi MA, Agostinetto E, Perachino M, Del Mastro L, de Azambuja E, Vaz-Luis I et al (2021) Evidence-based approaches for the management of side-effects of adjuvant endocrine therapy in patients with breast cancer. Lancet Oncol Lancet Publishing Group 22:e303–e313
Pumo V, Milone G, Iacono M, Giuliano SR, Di A, Lopiano MC et al (2012) Psychological and sexual disorders in long-term breast cancer survivors. Cancer Manag Res 2012:61–65
Yussof I, Mohd Tahir NA, Hatah E, Mohamed SN (2022) Factors influencing five-year adherence to adjuvant endocrine therapy in breast cancer patients: a systematic review. Breast Churchill Livingstone 62:22–35
Acknowledgements
We thank patients who generously gave their time to participate in the study.
Author information
Authors and Affiliations
Contributions
YU: data curation, formal analysis, writing—original draft, writing—review and editing, and visualization. VK: conceptualization, funding acquisition, investigation, writing—review and editing. DSS: data curation, resources, writing—review and editing. CU: conceptualization, funding acquisition, and investigation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Acibadem University Medical Research Ethic Committee (decision number 2019–17/48). Informed consent was obtained from all individual participants included in the study. The research was conducted in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uslu, Y., Kocatepe, V., Sezgin, D.S. et al. Adherence to adjuvant tamoxifen and associated factors in breast cancer survivors. Support Care Cancer 31, 285 (2023). https://doi.org/10.1007/s00520-023-07742-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-07742-2