Abstract
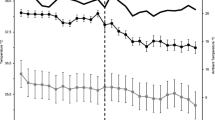
The objective of this study was to investigate the effect of acute and chronic heat load events on scrotal temperature (ST), body temperature (BT) and bull behaviour, and to examine the interrelationship between these parameters; the underlying hypothesis was that adverse heat treatments delivered in a temperature controlled environment will lead to thermoregulatory dysfunction of the bull scrotum. Six sexually mature Wagyu bulls were used in this study with data loggers surgically implanted into the abdominal cavity and scrotum. Body temperate and ST were recorded at 30-min intervals for the duration of the study. There were two housing locations used throughout the study, outdoor pens and climate control rooms. The study was designed as a four-phase crossover design with two heat treatments: (1) a 5-day acute challenge, and (2) a 14-day chronic challenge. The study was also blocked by phase to control for systematic change between phases with a thermoneutral (TN) phase in outdoor pens between each heat challenge. Observations within the climate rooms were conducted at 1-h intervals and data on panting scores (PS), respiration rate (RR), posture (standing or lying) and general behaviours (feeding, drinking, ruminating) recorded. Ambient temperature (AT, °C) and relative humidity (RH, %) were obtained at 10-min intervals and used to calculate the temperature humidity index (THI). Multiple models were conducted using a linear mixed effects model that contained different permutations of date and time factors and interactions as well as inclusion of an autoregressive parameter. The strongest model based on Akaike’s information criterion (AIC) was selected and further analysed. Ambient conditions during heat treatments were consistent with heat load and bulls showed typical physiological symptoms of the same. Maximum ST for acute and chronic treatments occurred once AT had exceeded 34 °C for at least 3 h (acute 35.59 °C at 1500 h; chronic 35.18 °C at 1400 h), whereas during TN conditions, maximum ST was at 2100 h. All phases showed variation in ST throughout the day. There were strong cross correlations between ST and RR during the heat treatments (acute r = 0.918, P < 0.0001; chronic r = 0.916, P < 0.0001), but not during TN (r = 0.411, P < 0.05). Our results confirmed that the ST of the bulls used in this study was not held at a constant temperature and that there was a possible connection between ST and RR. We have shown that during a period of heat load, the thermoregulatory mechanisms thought responsible for maintaining bovine ST appear to breakdown.






Similar content being viewed by others
References
Barros Adwell C et al (2018) Arterial blood flow is the main source of testicular heat in bulls and higher ambient temperatures significantly increase testicular blood flow. Therio 116:12–16
Blazquez NB, Mallard GJ, Wedd SR (1988) Sweat glands of the scrotum of the bull. J Reprod Fertil 83:673–677
Brito LF, Silva AE, Barbosa RT, Kastelic JP (2004) Testicular thermoregulation in Bos indicus, crossbred and Bos taurus bulls: relationship with scrotal, testicular vascular cone and testicular morphology, and effects on semen quality and sperm production. Therio 61:511–528
Brown-Brandl TM, Eigenberg RA, Nienaber J (2006) Heat stress risk factors of feedlot heifers. Livest Sci 105:57–68
Carrick F, Setchell B (1977) The evolution of the scrotum. In: Calaby J, Tyndale-Biscoe CH (eds) Reproduction and Evolution. Australian Academy of Science, Canberra, pp 165–170
Casady RB, Myers RM, Legates JE (1953) The effect of exposure to high ambient temperature on spermatogenesis in the dairy bull. J Dairy Sci 36:14–23
Castaneda C, Gaughan J, Sakaguchi Y (2004) Relationships between climatic conditions and the behaviour of feedlot cattle. Sci Access 1:33–36
Cook RB, Coulter GH, Kastelic JP (1994) The testicular vascular cone, scrotal thermoregulation, and their relationship to sperm production and seminal quality in beef bulls. Therio 41:653–671
Coulter G, Senger P, Bailey D (1988) Relationship of scrotal surface temperature measured by infrared thermography to subcutaneous and deep testicular temperature in the ram. J Reprod Fertil 84:417–423
De K, Kumar D, Balaganur K, Saxena VK, Thirumurugan P, Naqvi SMK (2017) Effect of thermal exposure on physiological adaptability and seminal attributes of rams under semi-arid environment. J Therm Biol 65:113–118
Durairajanayagam D, Agarwal A, Ong C (2015) Causes and molecular mechanisms of testicular heat stress. Reprod BioMed 30:14–27
Gaughan JB, Goopy JP (2002) Behaviour of lot fed cattle when exposed to hot environmental conditions. In: 15th Conference on Biometeorology and Aerobiology, and 16th Int Congress of Biometeorology
Gaughan J, Mader T (2014) Body temperature and respiratory dynamics in un-shaded beef cattle. Int J Biometeorol 58:1443–1450
Gaughan J, Mader TL, Holt S, Lisle A (2008) A new heat load index for feedlot cattle. J Anim Sci 86:226–234
Hahn GL (1989) Body temperature rhythms in farm animals—a review and reassessment relative to environmental influences. In: Proc. 11th Intl. Soc. Biometeorol. Congr., The Hague, The Netherlands. p 325-337
Hahn G (1999) Dynamic responses of cattle to thermal heat loads. J Anim Sci 77:10–20
Hahn G, Mader T (1997) Heat waves in relation to thermoregulation, feeding behavior and mortality of feedlot cattle. In: Proceedings, Fifth International Livestock Environment Symposium. p 563-571
Hahn GL, Eigenberg R, Nienaber J, Littledike ET (1990) Measuring physiological responses of animals to environmental stressors using a microcomputer-based portable datalogger. J Anim Sci 68:2658–2665
IPCC (2013) Summary for policymakers. In: Stocker TF et al (eds) Climate change 2013: the physical science basis. Contribution of Working Group 1 to the FIfth Assessment Report of the Intergovernmental Panel on Climate Changte. Cambridge Unviersity Press, Cambridge
Johnston J, Naelapaa H, Frye J (1963) Physiological responses of Holstein, Brown Swiss and Red Sindhi crossbred bulls exposed to high temperatures and humidities. J Anim Sci 22:432–436
Kahwage PR, Esteves SN, Jacinto MAC, Junior WB, Pezzopane JRM, de Andrade Pantoja MH, Bosi C, Miguel MCV, Mahlmeister K, Garcia AR (2017) High systemic and testicular thermolytic efficiency during heat tolerance test reflects better semen quality in rams of tropical breeds. Int J Biometeorol 61(10):1819–1829
Karabinus DS, Vogler CJ, Saacke RG, Evenson DP (1997) Chromatin structural changes in sperm after scrotal insulation of Holstein bulls. J Androl 18:549–555
Kastelic JP (1999) Scrotal/testicular thermoregulation in the bull. Annu Rev Biomed Sci 1:87–101
Kastelic J, Cook R, Coulter G (1995) Scrotal/testicular thermoregulation in the bull. In: Proceedings-American Society of Animal Science Western Section. p 227-229
Kastelic J, Cook R, Coulter G (1996a) Contribution of the scrotum and testes to scrotal and testicular thermoregulation in bulls and rams. J Reprod Fertil 108:81–85
Kastelic J, Cook R, Coulter G, Saacke R (1996b) Insulating the scrotal neck affects semen quality and scrotal/testicular temperatures in the bull. Therio 45:935–942
Kastelic JP, Cook RB, Coulter GH (2000) Scrotal/testicular thermoregulation in bulls. In: Chenoweth P (ed) Topics in bull fertility. International Veterinary Information Service, Alberta, pp 16–19
Kastelic J, Rizzoto G, Thundathil J (2018) Testicular vascular cone development and its association with scrotal thermoregulation, semen quality and sperm production in bulls. Animal 12:s133–s141
LCI (1970) Patterns of transit losses. Livestock Conservation, Inc., Omaha
Lees AM, Lees JC, Lisle AT, Sullivan ML, Gaughan JB (2018) Effect of heat stress on rumen temperature of three breeds of cattle. Int J Biometeorol 62:207–215
Lees AM, Sejian V, Lees JC, Sullivanm ML, Lisle AT, Gaughan JB (2019) Evaluating rumen temperature as an estimate of core body temperature in Angus feedlote cattle during summer. Int J Biometeorol 63:939–947
Lees AM, Lees JC, Sejian V, Sullivan ML, Gaughan JB (2020) Influence of shade on panting score and behavioural responses of Bos taurus and Bos indicus feedlot cattle to heat load. Anim Prod Sci 60:305–315
Luceño NL, Angrimani DdSR, de Cássia Bicudo L, Szymanska KJ, Van Poucke M, Demeyere K, Meyer E, Peelman L, Mullaart E, and Broekhuijse ML (2020) Exposing dairy bulls to high temperature-humidity index during spermatogenesis compromises subsequent embryo development in vitro. Theriogenology 141: pp. 16–25
Mader TL (2003) Environmental stress in confined beef cattle. J Anim Sci 81:E110
Mader TL, Kreikemeier WM (2006) Effects of growth-promoting agents and season on blood metabolites and body temperature in heifers. J Anim Sci 84:1030–1037
Mader TL, Holt S, Scott T, Davis S (2001) Restricted feeding strategies for reducing heat load of yearling steers. Nebraska Beef Cattle Reports 307
Mader TL, Davis M, Brown-Brandl T (2006) Environmental factors influencing heat stress in feedlot cattle. J Anim Sci 84:712–719
Mader TL, Davis MS, Gaughan JB (2007) Effect of sprinkling on feedlot microclimate and cattle behavior. Int J Biometeorol 51(6):541–551. https://doi.org/10.1007/s00484-007-0093-8
Mader TL, Gaughan JB, Johnson LJ, Hahn GL (2010) Tympanic temperature in confined beef cattle exposed to excessive heat load. Int J Biometeorol 54:629–635
Maloney SK, Bonomelli JM, DeSouza J (2003) Scrotal heating stimulates panting and reduces body temperature similarly in febrile and non-febrile rams (Ovis aries). Comp Biochem Physiol A Mol Integr Physiol 135(4):565–573
Menegassi SRO, Pereira GR, Dias EA, Koetz C Jr, Lopes FG, Bremm C, Pimentel C, Lopes RB, da Rocha MK, Carvalho HR, Barcellos JOJ (2016) The uses of infrared thermography to evaluate the effects of climatic variables in bull’s reproduction. Int J Biometeorol 60:151–157
Meyerhoeffer DC, Wettemann RP, Coleman SW, Wells ME (1985) Reproductive criteria of beef bulls during and after exposure to increased ambient temperature. J Anim Sci 60:352–357
Minton JE, Wettemann R, Meyerhoeffer D, Hintz R, Turman E (1981) Serum luteinizing hormone and testosterone in bulls during exposure to elevated ambient temperature. J Anim Sci 53:1551–1558
Mitlohner FM, Morrow-Tesch JL, Wilson SC, Dailey JW, McGlone JJ (2001) Behavioral sampling techniques for feedlot cattle. J Anim Sci 79:1189–1193
Morrow-Tesch JL, Woolen N, Hahn GL (1996) Response of gamma delta T-lymphocytes to heat stress in Bos taurus nd Bos indicus crossbred cattle. J Therm Biol 21:101–108
R Core Team (2019) R: a language and environment for statistical computing. R Fondation for Statistical Computing, Vienna
Refinetti R, Menaker M (1992) The circadian rhythm of body temperature. Physiol Behav 51:613–637
Ross AD, Entwistle KW (1979) The effect of scrotal insulation on spermatozoal morphology and the rates of spermatogenesis and epididymal passage of spermatozoa in the bull. Therio 11:111–129
Sealfon AI, Zorgniotti AW (1991) A theoretical model for testis thermoregulation. In: Temperature and Environment Effects on the Testis. Springer. pp. 123–35
Sejian V, Bhatta R, Gaughan J, Dunshea F, Lacetera N (2018) Adaptation of animals to heat stress. Animal 12:s431–s444
Setchell BP (1978) The mammalian testis. Elek, London
Setchell BP (1998) The parkes lecture heat and the testis. J Reprod Fertil 114:179–194
Setchell BP (2006) The effects of heat on the testes of mammals. Anim Reprod 3:81–91
Skinner JD, Louw GN (1966) Heat stress and spermatogenesis in Bos indicus and Bos taurus cattle. J Appl Physiol 21:1784–1790
Sorensen H, Lambrechtsen J, Einer-Jensen N (1991) Efficiency of the countercurrent transfer of heat and 133Xenon between the pampiniform plexus and testicular artery of the bull under in-vitro conditions. Int J Androl 14:232–240
Thom EC (1959) The discomfort index. Weatherwise 12:57–61
Vogler CJ, Bame JH, DeJarnette JM, McGilliard ML, Saacke RG (1993) Effects of elevated testicular temperature on morphology characteristics of ejaculated spermatozoa in the bovine. Therio 40:1207–1219
Waites G (1961) The polypnea evoked by heating the scrotum of the ram. Nature 190:172–173
Waites G (1962) The effect of heating the scrotum of the ram on respiration and body temperature. Exp Physiol 47:314–323
Waites GM (1991) Thermoregulation of the scrotum and testis: studies in animals and significance for man. In: Temperature and environmental effects on the testis. Springer. pp. 9–17
Waites G, Voglmayr JK (1963) The functional activity and control of the apocrine sweat glands of the scrotum of the ram. Aust Agri Res 14:839–851
Waites G, Setchell B, Quinlan D (1973) Effect of local heating of the scrotum, testes and epididymides of rats on cardiac output and regional blood flow. J Reprod Fertil 34:41–49
Wallage AL, Gaughan JB, Lisle AT, Beard L, Collins CW, Johnston SD (2017a) Measurement of bovine body and scrotal temperature using implanted temperature sensitive radio transmitters, data loggers and infrared thermography. Int J Biometeorol 61:1309–1321
Wallage AL, Johnston SD, Lisle AT, Beard L, Lees AM, Collins CW, Gaughan JB (2017b) Thermoregulation of the bovine scrotum 1: measurements of free-ranging animals in a paddock and pen. Int J Biometeorol 61:1381–1387
Acknowledgements
The lead author acknowledges the support received through an Australian Government Research Training Program Scholarship. The authors gratefully acknowledge the assistance provided by Lyn Beard for the preparation of the data loggers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was conducted with the approval of the University of Queensland Animal Ethics committee (SAS/243/10).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wallage, A.L., Lees, A.M., Lisle, A.T. et al. Thermoregulation of the bovine scrotum 2: simulated acute and chronic heat waves reduces the scrotal thermoregulatory capability of Wagyu bulls. Int J Biometeorol 66, 251–262 (2022). https://doi.org/10.1007/s00484-021-02108-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-021-02108-x