Abstract
Background
Secondary hyperparathyroidism (SHPT) may persist after renal transplantation (RTx), inducing hypophosphatemia and hypercalcemia that precludes the use of vitamin D analogs. The calcimimetic cinacalcet improved plasma calcium and parathyroid hormone (PTH) levels in randomized controlled trials in adults after RTx, but pediatric data are scarce.
Methods
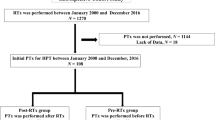
In this retrospective study, we analyzed 20 pediatric patients from the Cooperative European Paediatric Renal TransplAnt Initiative (CERTAIN) Registry who received cinacalcet after RTx. The results are presented as median and interquartile range (25th–75th percentile).
Results
At 13.7 (11.0–16.5) years of age, 20 pediatric patients received a renal allograft. Cinacalcet was introduced at 0.4 (0.3–2.7) years post-transplant at an estimated glomerular filtration rate (eGFR) of 50 (34–66) mL/min/1.73 m2, plasma calcium of 2.58 (2.39–2.71) mmol/L, age-standardized (z score) phosphate of − 1.7 (− 2.7−− 0.4), and PTH of 136 (95–236) ng/L. The starting dose of cinacalcet was 0.5 (0.3–0.8) mg/kg per day, with a maximum dose of 1.1 (0.5–1.3) mg/kg per day. With a follow-up of 3.0 (1.5–3.6) years on cinacalcet therapy, eGFR remained stable; PTH levels decreased to 66 (56–124) ng/L at the last follow-up (p = 0.015). One patient displayed hypocalcemia (1.8 mmol/L). Cinacalcet was withdrawn in three patients (hypocalcemia, parathyroidectomy, incompliance). Nephrocalcinosis of the graft was not reported.
Conclusions
This pilot study suggests that cinacalcet as off-label therapy for SHPT after pediatric RTx is efficacious in controlling post-transplant SHPT with acceptable tolerability. Continuing cinacalcet even with normal PTH can lead to dangerous life-threatening hypocalcemia. Therefore, at each subsequent visit, the need to continue cinacalcet must be assessed.





Similar content being viewed by others
References
Moe SM, Drüeke T (2008) Improving global outcomes in mineral and bone disorders. Clin J Am Soc Nephrol 3:S127–S130. https://doi.org/10.2215/CJN.04331206
Bacchetta J, Harambat J, Cochat P, Salusky IB, Wesseling-Perry K (2012) The consequences of chronic kidney disease on bone metabolism and growth in children. Nephrol Dial Transplant 27:3063–3071. https://doi.org/10.1093/ndt/gfs299
Wheeler DC, Winkelmayer WC (2017) KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD) foreword. Kidney Int Suppl 7:1–59
Sohn WY, Portale AA, Salusky IB, Zhang H, Yan LL, Ertik B, Shahinfar S, Lee E, Dehmel B, Warady BA (2019) An open-label, single-dose study to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of cinacalcet in pediatric subjects aged 28 days to < 6 years with chronic kidney disease receiving dialysis. Pediatr Nephrol 34:145–154. https://doi.org/10.1007/s00467-018-4054-8
Bacchetta J, Schmitt CP, Ariceta G, Bakkaloglu SA, Groothoft J, Wan M, Vervloet M, Shroff R, Haffner D, on belhalf of the European Society for Paediatric Nephrology and the Chronic Kidney Disease-Mineral and Bone Disorders and Dialysis Working Group of the ERA-EDTA (2020) Cinacalcet use in paediatric dialysis: a position statement from the European Society for Paediatric Nephrology and the Chronic Kidney Disease-Mineral and Bone Disorders Working Group of the ERA-EDTA. Nephrol Dial Transplant 35:47–64. https://doi.org/10.1093/ndt/gfz159
Serra AL, Savoca R, Huber AR, Hepp U, Delsignore A, Hersberger M, Wüthrich RP (2007) Effective control of persistent hyperparathyroidism with cinacalcet in renal allograft recipients. Nephrol Dial Transplant 22:577–583. https://doi.org/10.1093/ndt/gfl560
El-Amm J-M, Doshi MD, Singh A, Migdal S, Moranwski K, Sternbauer D, Cincotta E, West MS, Losanoff JE, Gruber SA (2007) Preliminary experience with cinacalcet use in persistent secondary hyperparathyroidism after kidney transplantation. Transplantation 83:546–549. https://doi.org/10.1097/01.tp.0000253429.33198.2f
Gómez Marqués G, Obrador Mulet A, Vilar Gimeno A, Pascual Felip MJ, Alarcon Zurita A, Molina Guasch M, Uriol Rivera M, Losada Gonzalez P (2009) Treatment with cinacalcet of secondary hyperparathyroidism after renal transplantation. Transplant Proc 41:2139–2143. https://doi.org/10.1016/j.transproceed.2009.06.090
Copley JB, Germain M, Stern L,Pankewcz O, Katznelson S, Shah T, Wang O, Turner SA, Sprague SM (2010) Evaluation of cinacalcet HCl treatment after kidney transplantation. Transplant Proc 42:2503–2508. https://doi.org/10.1016/j.transproceed.2010.04.052
Cho ME, Duan Z, Chamberlain CE, Reynols JC, Ring MS, Mannon RB (2010) Cinacalcet improves bone density in post-kidney transplant hyperparathyroidism. Transplant Proc 42:3554–3558. https://doi.org/10.1016/j.transproceed.2010.06.027
Courbebaisse M, Diet C, Timsit M-O, Mamzer MF, Thervet E, Noel LH, Legendre C, Friedlnder G, Martinez F, Prié D (2012) Effects of cinacalcet in renal transplant patients with hyperparathyroidism. Am J Nephrol 35:341–348. https://doi.org/10.1159/000337526
Paschoalin RP, Torregrosa J-V, Sánchez-Escuredo A, Barros X, Duran CE, Campistol JM (2012) Cinacalcet treatment for stable kidney transplantation patients with hypercalcemia due to persistent secondary hyperparathyroidism: a long-term follow-up. Transplant Proc 44:2588–2589. https://doi.org/10.1016/j.transproceed.2012.09.049
Torregrosa JV, Morales E, Díaz JM, Crespo J, Bravo J, Gomez G, Gentil MA, Rodriguez-Benot A, Rodriguez-Garcia M, Lopez-Jimenez V, Gutiérrez-Dalmau A, Jimeno L, Pérez-Saez MJ, Romero R, Gomez-Alamillo C, Grupo de Estudio CINAREN (2014) Cinacalcet in the management of normocalcaemic secondary hyperparathyroidism after kidney transplantation: one-year follow-up multicentre study. Nefrologia 34:62–68. https://doi.org/10.3265/Nefrologia.pre2013.Aug.12183
Ważna-Jabłońska E, Gałązka Z, Durlik M (2016) Treatment of persistent hypercalcemia and hyperparathyroidism with cinacalcet after successful kidney transplantation. Transplant Proc 48:1623–1625. https://doi.org/10.1016/j.transproceed.2016.01.044
Soliman AR, Maamoun HA, Soliman MA, Daewish H, Elbanna E (2016) Cinacalcet versus parathyroidectomy in the treatment of secondary hyperparathyroidism post renal transplantation. Rom J Intern Med 54:184–189. https://doi.org/10.1515/rjim-2016-0027
Mawad H, Bouchard H, Tran D, Ouimet D, Lafrance JP, Bell RB, Bezzaoucha S, Boucher A, Collette S, Pichette V, Senécal L, Vallée M (2017) Retrospective study looking at cinacalcet in the management of hyperparathyroidism after kidney transplantation. J Transp Secur 2017. https://doi.org/10.1155/2017/8720283
Kruse AE, Eisenberger U, Frey FJ, Mohaupt MG (2005) The calcimimetic cinacalcet normalizes serum calcium in renal transplant patients with persistent hyperparathyroidism. Nephrol Dial Transplant 20:1311–1314. https://doi.org/10.1093/ndt/gfh924
Apostolou T, Kollia K, Damianou L, Kaitsioti H, Kotsiev V, Dracopoulos S, Vougas V, Hadjiconstantinou V (2006) Hypercalcemia due to resistant hyperparathyroidism in renal transplant patients treated with the calcimimetic agent cinacalcet. Transplant Proc 38:3514–3516. https://doi.org/10.1016/j.transproceed.2006.10.133
Kamar N, Gennero I, Spataru L, Esposito L, Guitard J, Lavayssière L, Cointault O, Gandia O, Durand D, Rostaing L (2008) Pharmacodynamic effects of cinacalcet after kidney transplantation: once- versus twice-daily dose. Nephrol Dial Transplant 23:3720–3726. https://doi.org/10.1093/ndt/gfn345
Bergua C, Torregrosa J-V, Fuster D, Gutierrez-Dalmau A, Oppenheimer F, Campistol JM (2008) Effect of cinacalcet on hypercalcemia and bone mineral density in renal transplanted patients with secondary hyperparathyroidism. Transplantation 86:413–417. https://doi.org/10.1097/TP.0b013e31817c13e1
Borchhardt KA, Heinzl H, Mayerwöger E, Hörl WH, Haas M, Sunder-Plassmann G (2008) Cinacalcet increases calcium excretion in hypercalcemic hyperparathyroidism after kidney transplantation. Transplantation 86:919–924. https://doi.org/10.1097/TP.0b013e318186b7fb
Serra AL, Wuhrmann C, Wüthrich RP (2008) Phosphatemic effect of cinacalcet in kidney transplant recipients with persistent hyperparathyroidism. Am J Kidney Dis 52:1151–1157. https://doi.org/10.1053/j.ajkd.2008.08.012
Carrasco FR, Pérez-Flores I, Calvo N, Ridao A, Sanchez A, Barrientos A (2009) Treatment of persistent hyperparathyroidism in renal transplant patients with cinacalcet improves control of blood pressure. Transplant Proc 41:2385–2387. https://doi.org/10.1016/j.transproceed.2009.06.167
López V, Toledo R, Sola E, Guttiérrez C, Sujan S, Rodriguez MA, Cabello M, Burgos D, Gonzalez Molina M, Hernandez D (2009) Treatment with cinacalcet in 29 kidney transplant patients with persistent hyperparathyroidism. Transplant Proc 41:2394–2395. https://doi.org/10.1016/j.transproceed.2009.06.055
Guerra R, Auyanet I, Fernández EJ, Pérez MA, Bosch E, Ramirez A, Suria S, Checa MD (2011) Hypercalcemia secondary to persistent hyperparathyroidism in kidney transplant patients: analysis after a year with cinacalcet. J Nephrol 24:78–82
Aalten J, Wetzels JFM, Hoitsma AJ (2010) Continuation of cinacalcet immediately after renal transplantation: a prospective cohort study. Clin Nephrol 74:433–439
Schwarz A, Merkel S, Leitolf H, Haller H (2011) The effect of cinacalcet on bone remodeling and renal function in transplant patients with persistent hyperparathyroidism. Transplantation 91:560–565. https://doi.org/10.1097/TP.0b013e3182079431
Pinho LRS, Ribeiro Santos MJC, Pestana Vasconcelos M (2011) Cinacalcet in the treatment of persistent hyperparathyroidism after kidney transplantation. Clin Nephrol 75:263–268
Boulanger H, Haymann J-P, Fouqueray B, Mansouri R, Metivier F, Mercadal L, Attaf D, Flamant M, Glotz D (2012) Cinacalcet impact on calcium homeostasis and bone remodeling in 13 renal transplanted patients with hyperparathyroidism and hypercalcaemia. Nephrol Ther 8:47–53. https://doi.org/10.1016/j.nephro.2011.04.007
Zitt E, Woess E, Mayer G, Lhotta K (2011) Effect of cinacalcet on renal electrolyte handling and systemic arterial blood pressure in kidney transplant patients with persistent hyperparathyroidism. Transplantation 92:883–889. https://doi.org/10.1097/TP.0b013e31822d87e8
Evenepoel P, Sprangers B, Lerut E, Bammens B, Claes K, Kuypers, Meijers B, Varenterghem Y (2012) Mineral metabolism in renal transplant recipients discontinuing cinacalcet at the time of transplantation: a prospective observational study. Clin Transpl 26:393–402. https://doi.org/10.1111/j.1399-0012.2011.01524.x
Cohen JB, Gordon CE, Balk EM, Francis JM (2012) Cinacalcet for the treatment of hyperparathyroidism in kidney transplant recipients: a systematic review and meta-analysis. Transplantation 94:1041–1048. https://doi.org/10.1097/TP.0b013e31826c3968
Evenepoel P, Cooper K, Holdaas H, Messa P, Mourad G, Olgaard K, Ruthkowski B, Schaefer H, Deng H, Torregrosa JV, Wuthrich RP, Yue S (2014) A randomized study evaluating cinacalcet to treat hypercalcemia in renal transplant recipients with persistent hyperparathyroidism. Am J Transplant 14:2545–2555. https://doi.org/10.1111/ajt.12911
Thiem U, Gessl A, Borchhardt K (2015) Long-term clinical practice experience with cinacalcet for treatment of hypercalcemic hyperparathyroidism after kidney transplantation. Biomed Res Int 2015:292654. https://doi.org/10.1155/2015/292654
Cruzado JM, Moreno P, Torregrosa JV, Taco O, Mast R, Gomez-Vaquero C, Polo C, Revuelta I, Francos J, Torras J Garcia-Barrasa A, Bestard O, Grinyo JM (2016) A Randomized study comparing parathyroidectomy with cinacalcet for treating hypercalcemia in kidney allograft recipients with hyperparathyroidism. - PubMed - NCBI. https://www-ncbi-nlm-nih-gov.gate2.inist.fr/pubmed/26647424. Accessed 28 Sep 2018
Zavvos V, Fyssa L, Papasotiriou M, Papachristou E, Ntrinias T, Savviadaki E, Goumenos DS (2018) Long-term use of cinacalcet in kidney transplant recipients with hypercalcemic secondary hyperparathyroidism: a single-center prospective study. Exp Clin Transplant 16:287–293. https://doi.org/10.6002/ect.2016.0342
Borchhardt KA, Diarra D, Sulzbacher I, Benesch T, Haas M, Sunder-Plassmann G (2010) Cinacalcet decreases bone formation rate in hypercalcemic hyperparathyroidism after kidney transplantation. Am J Nephrol 31:482–489. https://doi.org/10.1159/000304180
Borstnar S, Erzen B, Gmeiner Stopar T, Kocjan T, Arnol M, Kandus D, Kovac D (2010) Treatment of hyperparathyroidism with cinacalcet in kidney transplant recipients. Transplant Proc 42:4078–4082. https://doi.org/10.1016/j.transproceed.2010.09.059
Henschkowski J, Bischoff-Ferrari HA, Wüthrich RP, Serra AL (2011) Renal function in patients treated with cinacalcet for persistent hyperparathyroidism after kidney transplantation. Kidney Blood Press Res 34:97–103. https://doi.org/10.1159/000323902
Niel O, Maisin A, Macher M-A, Peuchmaur M, Deschênes G (2016) Cinacalcet in hyperparathyroidism management after pediatric renal transplantation. CEN Case Rep 5:141–143. https://doi.org/10.1007/s13730-015-0211-0
Plotnicki L, Kohl CD, Höcker B, Krupka K, Rahmel A, Pape L, Hoyer P, Marks SD, Webb NJA, Söylemezoglu O, Topaloglu R, Szabo AJ, Seeman T, Marlies Cornelissen EA, Knops N, Grenda R, Tönshoff B (2013) The CERTAIN Registry: a novel, web-based registry and research platform for pediatric renal transplantation in Europe. Transplant Proc 45:1414–1417. https://doi.org/10.1016/j.transproceed.2013.01.007
Köster L, Krupka K, Höcker B, Rahmel A, Samuel U, Zanen W, Opelz G, Süsal C, Döhler B, Plotnicki L, Kohl CD, Knaup P, Tönshoff B (2015) Integrating data from multiple sources for data completeness in a web-based registry for pediatric renal transplantation--the CERTAIN Registry. Stud Health Technol Inform 216:1049
Schwartz GJ, Work DF (2009) Measurement and estimation of GFR in children and adolescents. Clin J Am Soc Nephrol 4:1832–1843. https://doi.org/10.2215/CJN.01640309
Habbig S, Beck BB, Hoppe B (2011) Nephrocalcinosis and urolithiasis in children. Kidney Int 80:1278–1291. https://doi.org/10.1038/ki.2011.336
Nanmoku K, Shinzato T, Kubo T, Shimizu T, Yagisawa T (2019) Prevalence and predictors of early hypercalcemia after kidney transplantation: a nested case–control study within a cohort of 100 patients. Clin Exp Nephrol 23:268–274. https://doi.org/10.1007/s10157-018-1627-6
Gulleroglu K, Baskin E, Moray G, Haberal M (2016) Low-grade persistent hyperparathyroidism after pediatric renal transplant. Exp Clin Transplant 14:294–298. https://doi.org/10.6002/ect.2014.0157
Kim DH, Lee JH, Han DJ, Park YS (2018) Risk factors for persistent hyperparathyroidism in children with stable renal function after kidney transplantation. Pediatr Transplant 22:e13238. https://doi.org/10.1111/petr.13238
Wesseling-Perry K, Pereira RC, Tsai E, Ettenger R, Jüppner H, Salusky IB (2013) FGF23 and mineral metabolism in the early post-renal transplantation period. Pediatr Nephrol 28:2207–2215. https://doi.org/10.1007/s00467-013-2547-z
Warady BA, Iles JN, Ariceta G, Dehmel B, Hidalgo G, Jiang X, Laskin B, Shahinfar S, Vande Walle J, Schaefer F (2019) A randomized, double-blind, placebo-controlled study to assess the efficacy and safety of cinacalcet in pediatric patients with chronic kidney disease and secondary hyperparathyroidism receiving dialysis. Pediatr Nephrol 34:475–486. https://doi.org/10.1007/s00467-018-4116-y
Conflict of interest
Justine Bacchetta has been an investigator for the Amgen-sponsored trials on the use of cinacalcet in pediatric dialysis. Justine Bacchetta is the coordinator of the Amgen-sponsored international registry on the use of cinacalcet in pediatric dialysis, and the national principal investigator for the Amgen-sponsored trial on the use of etelcalcetide in pediatric dialysis. She received a research grant for experimental studies by Amgen.
Claus Peter Schmitt received financial support from Amgen for investigator-initiated experimental research, a travel grant, and is national principal investigator for the Amgen-sponsored trial on the use of etelcalcetide in pediatric dialysis.
No other relevant conflicts of interest are reported.
Funding
The authors received financial support from the CERTAIN Registry by a grant from the Dietmar Hopp Stiftung, the European Society for Paediatric Nephrology (ESPN), and the German Society for Paediatric Nephrology (GPN), and by grants from the pharmaceutical companies Astellas and Novartis.
The non-profit pediatric association of CHU de Nice (ADHPUN) paid for statistical analyses, performed by SARL Green Grow Scientific.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
For research purposes, an analysis of the multinational dataset requires approval of the predefined study protocol by the CERTAIN Registry Steering Committee. Written informed consent was obtained from all parents/guardians to participate in the registry, with assent from patients as appropriate for their age. The CERTAIN Registry has been approved by the ethics committee of each contributing center and is kept in full accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bernardor, J., Schmitt, C.P., Oh, J. et al. The use of cinacalcet after pediatric renal transplantation: an international CERTAIN Registry analysis. Pediatr Nephrol 35, 1707–1718 (2020). https://doi.org/10.1007/s00467-020-04558-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-020-04558-8