Abstract
Background
Despite technical advances in minimally invasive gastrectomy for gastric cancer, an increased incidence of postoperative pancreatic fistula (POPF) has been reported. POPF can cause infectious and bleeding complications, which could lead to surgery-related death; therefore, reduction of the post-gastrectomy POPF risk is crucial. This study aimed to investigate the importance of pancreatic anatomy as a predictor of POPF in patients undergoing laparoscopic or robotic gastrectomy.
Methods
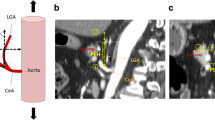
Data were collected from 331 consecutive patients who underwent laparoscopic or robotic gastrectomy for gastric cancer. The thickness of the pancreas anterior to the most ventral level of the splenic artery (TPS) was measured. The correlation between TPS and POPF incidence was investigated using univariate and multivariate analyses.
Results
The cutoff value of TPS was 11.8 mm, which predicted a high drain amylase concentration on postoperative day 1, and patients were categorized into thin (Tn group) and thick TPS groups (Tk group). There was no significant difference in the background characteristics between the two groups, except for sex (P = 0.009) and body mass index (P < 0.001). The incidences of POPF grade B or higher (2% vs. 16%, P < 0.001), all postoperative complications of grade II or higher (12% vs. 28%, P = 0.004), and postoperative intra-abdominal infections of grade II or higher (4% vs. 17%, P = 0.001) were significantly higher in the Tk group. Multivariable analysis identified that high TPS was the only independent risk factor for grade B or higher POPF and grade II or higher postoperative intra-abdominal infectious complications.
Conclusions
The TPS is a specific predictive factor for POPF and postoperative intra-abdominal infectious complications in patients undergoing laparoscopic or robotic gastrectomy. Careful pancreatic manipulation during suprapancreatic lymphadenectomy is necessary for patients with increased TPS (> 11.8 mm) to avoid postoperative complications.



Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Morgan E, Arnold M, Camargo MC, Gini A, Kunzmann AT, Matsuda T, Meheus F, Verhoeven RHA, Vignat J, Laversanne M, Ferlay J, Soerjomataram I (2022) The current and future incidence and mortality of gastric cancer in 185 countries, 2020–40: a population-based modelling study. EClinicalMedicine 47:101404
Hiki N, Honda M, Etoh T, Yoshida K, Kodera Y, Kakeji Y, Kumamaru H, Miyata H, Yamashita Y, Inomata M, Konno H, Seto Y, Kitano S (2018) Higher incidence of pancreatic fistula in laparoscopic gastrectomy. Real-world evidence from a nationwide prospective cohort study. Gastric Cancer 21:162–170
Guerra F, Giuliani G, Iacobone M, Bianchi PP, Coratti A (2017) Pancreas-related complications following gastrectomy: systematic review and meta-analysis of open versus minimally invasive surgery. Surg Endosc 31:4346–4356
Jiang X, Hiki N, Nunobe S, Kumagai K, Nohara K, Sano T, Yamaguchi T (2012) Postoperative pancreatic fistula and the risk factors of laparoscopy-assisted distal gastrectomy for early gastric cancer. Ann Surg Oncol 19:115–121
Tsujinaka T, Sasako M, Yamamoto S, Sano T, Kurokawa Y, Nashimoto A, Kurita A, Katai H, Shimizu T, Furukawa H, Inoue S, Hiratsuka M, Kinoshita T, Arai K, Yamamura Y, Gastric Cancer Surgery Study Group of Japan Clinical Oncology Group (2007) Influence of overweight on surgical complications for gastric cancer: results from a randomized control trial comparing D2 and extended para-aortic D3 lymphadenectomy (JCOG9501). Ann Surg Oncol 14:355–361
Wu J, Tang Z, Zhao G, Zang L, Li Z, Zang W, Li Z, Qu J, Yan S, Zheng C, Ji G, Zhu L, Zhao Y, Zhang J, Huang H, Hao Y, Fan L, Xu H, Li Y, Yang L, Song W, Zhu J, Zhang W, Li M, Qin X, Liu F (2022) Incidence and risk factors for postoperative pancreatic fistula in 2089 patients treated by radical gastrectomy: a prospective multicenter cohort study in China. Int J Surg 98:106219
Ojima T, Iwahashi M, Nakamori M, Nakamura M, Naka T, Ishida K, Ueda K, Katsuda M, Iida T, Tsuji T, Yamaue H (2009) Influence of overweight on patients with gastric cancer after undergoing curative gastrectomy: an analysis of 689 consecutive cases managed by a single center. Arch Surg 144:351–358
Yu HW, Jung DH, Son SY, Lee CM, Lee JH, Ahn SH, Park DJ, Kim HH (2013) Risk factors of postoperative pancreatic fistula in curative gastric cancer surgery. J Gastric Cancer 13:179–184
Kinoshita J, Yamaguchi T, Saito H, Moriyama H, Shimada M, Terai S, Okamoto K, Nakanuma S, Makino I, Nakamura K, Tajima H, Ninomiya I, Fushida S (2020) Comparison of prognostic impact of anatomic location of the pancreas on postoperative pancreatic fistula in laparoscopic and open gastrectomy. BMC Gastroenterol 20:325
Kobayashi N, Shinohara H, Haruta S, Ohkura Y, Mizuno A, Ueno M, Udagawa H, Sakai Y (2016) Process of pancreas head as a risk factor for postoperative pancreatic fistula in laparoscopic gastric cancer surgery. World J Surg 40:2194–2201
Kumagai K, Hiki N, Nunobe S, Kamiya S, Tsujiura M, Ida S, Ohashi M, Yamaguchi T, Sano T (2018) Impact of anatomical position of the pancreas on postoperative complications and drain amylase concentrations after laparoscopic distal gastrectomy for gastric cancer. Surg Endosc 32:3846–3854
Migita K, Matsumoto S, Wakatsuki K, Ito M, Kunishige T, Nakade H, Nakatani M, Kitano M, Nakajima Y (2016) The anatomical location of the pancreas is associated with the incidence of pancreatic fistula after laparoscopic gastrectomy. Surg Endosc 30:5481–5489
Ida S, Hiki N, Ishizawa T, Kuriki Y, Kamiya M, Urano Y, Nakamura T, Tsuda Y, Kano Y, Kumagai K, Nunobe S, Ohashi M, Sano T (2018) Pancreatic compression during lymph node dissection in laparoscopic gastrectomy: possible cause of pancreatic leakage. J Gastric Cancer 18:134–141
Japanese Gastric Cancer Association (2021) Japanese gastric cancer treatment guidelines. Gastric Cancer 24:1–21
De Sol A, Cirocchi R, Di Patrizi MS, Boccolini A, Barillaro I, Cacurri A, Grassi V, Corsi A, Renzi C, Giuliani D, Coccetta M, Avenia N (2015) The measurement of amylase in drain fluid for the detection of pancreatic fistula after gastric cancer surgery: an interim analysis. World J Surg Oncol 13:65
Iwata N, Kodera Y, Eguchi T, Ohashi N, Nakayama G, Koike M, Fujiwara M, Nakao A (2010) Amylase concentration of the drainage fluid as a risk factor for intra-abdominal abscess following gastrectomy for gastric cancer. World J Surg 34:1534–1539
Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, Allen P, Andersson R, Asbun HJ, Besselink MG, Conlon K, Del Chiaro M, Falconi M, Fernandez-Cruz L, Fernandez-Del Castillo C, Fingerhut A, Friess H, Gouma DJ, Hackert T, Izbicki J, Lillemoe KD, Neoptolemos JP, Olah A, Schulick R, Shrikhande SV, Takada T, Takaori K, Traverso W, Vollmer CR, Wolfgang CL, Yeo CJ, Salvia R, Buchler M, International Study Group on Pancreatic Surgery (ISGPS) (2017) The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery 161:584–591
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Cepeda MS, Boston R, Farrar JT, Strom BL (2003) Comparison of logistic regression versus propensity score when the number of events is low and there are multiple confounders. Am J Epidemiol 158:280–287
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48:452–458
Obama K, Okabe H, Hosogi H, Tanaka E, Itami A, Sakai Y (2011) Feasibility of laparoscopic gastrectomy with radical lymph node dissection for gastric cancer: from a viewpoint of pancreas-related complications. Surgery 149:15–21
Katai H, Sasako M, Fukuda H, Nakamura K, Hiki N, Saka M, Yamaue H, Yoshikawa T, Kojima K, JCOG Gastric Cancer Surgical Study Group (2010) Safety and feasibility of laparoscopy-assisted distal gastrectomy with suprapancreatic nodal dissection for clinical stage I gastric cancer: a multicenter phase II trial (JCOG 0703). Gastric Cancer 13:238–244
Inaki N, Etoh T, Ohyama T, Uchiyama K, Katada N, Koeda K, Yoshida K, Takagane A, Kojima K, Sakuramoto S, Shiraishi N, Kitano S (2015) A multi-institutional, prospective, phase II feasibility study of laparoscopy-assisted distal gastrectomy with D2 lymph node dissection for locally advanced gastric cancer (JLSSG0901). World J Surg 39:2734–2741
Miyai H, Hara M, Hayakawa T, Takeyama H (2013) Establishment of a simple predictive scoring system for pancreatic fistula after laparoscopy-assisted gastrectomy. Dig Endosc 25:585–592
Washio M, Yamashita K, Niihara M, Hosoda K, Hiki N (2020) Postoperative pancreatic fistula after gastrectomy for gastric cancer. Ann Gastroenterol Surg 4:618–627
Fujita T, Ohta M, Ozaki Y, Takahashi Y, Miyazaki S, Harada T, Iino I, Kikuchi H, Hiramatsu Y, Kamiya K, Konno H (2015) Collateral thermal damage to the pancreas by ultrasonic instruments during lymph node dissection in laparoscopic gastrectomy. Asian J Endosc Surg 8:281–288
Itamoto K, Hikage M, Fujiya K, Kamiya S, Tanizawa Y, Bando E, Terashima M (2021) The impact of pancreas compression time during minimally invasive gastrectomy on the postoperative complications in gastric cancer. Ann Gastroenterol Surg 5:785–793
Tsujiura M, Hiki N, Ohashi M, Nunobe S, Kumagai K, Ida S, Okumura Y, Sano T, Yamaguchi T (2017) “Pancreas-compressionless gastrectomy”: a novel laparoscopic approach for suprapancreatic lymph node dissection. Ann Surg Oncol 24:3331–3337
Brinkman DJ, Troquay S, de Jonge WJ, Irwin ED, Vervoordeldonk MJ, Luyer MDP, Nederend J (2021) Morphometric analysis of the splenic artery using contrast-enhanced computed tomography (CT). Surg Radiol Anat 43:377–384
Moraes DMV, Gutierres A, Colleoni Neto R, Lindemann IL, Rottenfusser R, Carlotto JRM (2022) Anatomy of the splenic artery: what does the surgeon need to know? Rev Col Bras Cir 49:e20223294
Kou K, Saisho Y, Jinzaki M, Itoh H (2014) Relationship between body mass index and pancreas volume in Japanese people. JOP 15:626–627
Kanaya S, Haruta S, Kawamura Y, Yoshimura F, Inaba K, Hiramatsu Y, Ishida Y, Taniguchi K, Isogaki J, Uyama I (2011) Video: laparoscopy distinctive technique for suprapancreatic lymph node dissection: medial approach for laparoscopic gastric cancer surgery. Surg Endosc 25:3928–3929
Suda K, Nakauchi M, Inaba K, Ishida Y, Uyama I (2016) Minimally invasive surgery for upper gastrointestinal cancer: our experience and review of the literature. World J Gastroenterol 22:4626–4637
Uyama I, Kanaya S, Ishida Y, Inaba K, Suda K, Satoh S (2012) Novel integrated robotic approach for suprapancreatic D2 nodal dissection for treating gastric cancer: technique and initial experience. World J Surg 36:331–337
Haverkamp L, Weijs TJ, van der Sluis PC, van der Tweel I, Ruurda JP, van Hillegersberg R (2013) Laparoscopic total gastrectomy versus open total gastrectomy for cancer: a systematic review and meta-analysis. Surg Endosc 27:1509–1520
Jeong O, Ryu SY, Zhao XF, Jung MR, Kim KY, Park YK (2012) Short-term surgical outcomes and operative risks of laparoscopic total gastrectomy (LTG) for gastric carcinoma: experience at a large-volume center. Surg Endosc 26:3418–3425
Ojima T, Nakamura M, Hayata K, Kitadani J, Katsuda M, Takeuchi A, Tominaga S, Nakai T, Nakamori M, Ohi M, Kusunoki M, Yamaue H (2021) Short-term outcomes of robotic gastrectomy vs laparoscopic gastrectomy for patients with gastric cancer: a randomized clinical trial. JAMA Surg 156:954–963
Kim YW, Reim D, Park JY, Eom BW, Kook MC, Ryu KW, Yoon HM (2016) Role of robot-assisted distal gastrectomy compared to laparoscopy-assisted distal gastrectomy in suprapancreatic nodal dissection for gastric cancer. Surg Endosc 30:1547–1552
Suda K, Nakauchi M, Inaba K, Ishida Y, Uyama I (2016) Robotic surgery for upper gastrointestinal cancer: current status and future perspectives. Dig Endosc 28:701–713
Ushiku H, Sakuraya M, Washio M, Hosoda K, Niihara M, Harada H, Miura H, Sato T, Nishizawa N, Tajima H, Kaizu T, Kato H, Sengoku N, Tanaka K, Naitoh T, Kumamoto Y, Sangai T, Yamashita K, Hiki N (2022) Pancreas-contactless gastrectomy for gastric cancer prevents postoperative inflammation. Surg Endosc 36:5644–5651
Acknowledgements
We would like to thank Editage for the English language editing.
Funding
This research received no specific grant from any funding agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Kengo Hayashi, Noriyuki Inaki, Yusuke Sakimura, Takahisa Yamaguchi, Yoshinao Obatake, Shiro Terai, Hirotaka Kitamura, Shinichi Kadoya, and Hiroyuki Bando have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hayashi, K., Inaki, N., Sakimura, Y. et al. Pancreatic thickness as a predictor of postoperative pancreatic fistula after laparoscopic or robotic gastrectomy. Surg Endosc 37, 5358–5367 (2023). https://doi.org/10.1007/s00464-023-10021-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-023-10021-0