Abstract
Objective
Thymectomy has been utilized as a treatment for Myasthenia Gravis (MG) for many decades, with both open and minimally invasive surgical (MIS) techniques currently used. Although MIS has shown improved short-term results, long-term effects remain uncertain. This systematic review and meta-analysis aim to compare the post-operative and long-term outcomes of MIS versus open thymectomy in MG patients.
Methods
MEDLINE, EMBASE and CENTRAL databases were searched from inception till January 2022 for keywords related to MG and open or MIS thymectomy. Primary outcome was complete stable remission (CSR), and secondary outcomes were clinical improvement, complications, length of stay, operation time, and blood loss. Grading of recommendations, assessment, development, and evaluation was used to assess the certainty of evidence.
Results
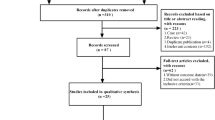
26 studies with 3588 patients were included in the analysis. At 1, 3 and 5 years, there was no statistical difference noted in CSR between open versus MIS thymectomy. However, CSR was improved at 1 year for MIS thymectomy in non-thymomatous MG (P = 0.03). There was no significant difference in rates of partial clinical improvement between techniques at 1-year. Although analyses on length of hospital stay and blood loss showed improvement following MIS thymectomy, operative time was shorter for open thymectomy.
Conclusion
This is the first systematic review and meta-analysis assessing long-term effects of MIS versus open thymectomy in MG patients. Given the lack of significant differences noted, either MIS or open thymectomy can be performed, based on surgeon preference. Further high-level, long-term research should be conducted to determine the benefit of each technique.




Similar content being viewed by others
References
Gilhus NE, Skeie GO, Romi F, Lazaridis K, Zisimopoulou P, Tzartos S (2016) Myasthenia gravis—autoantibody characteristics and their implications for therapy. Nat Rev Neurol 12(5):259–268. https://doi.org/10.1038/nrneurol.2016.44
Cea G, Benatar M, Rj V, Ra S (2013) Thymectomy for non-thymomatous myasthenia gravis. Cochrane Database Systemat Rev. https://doi.org/10.1002/14651858.CD008111.pub2
Wolfe GI, Kaminski HJ, Aban IB et al (2016) Randomized trial of thymectomy in myasthenia gravis. N Engl J Med 375(6):511–522. https://doi.org/10.1056/NEJMOA1602489
Gronseth GS, Barohn RJ (2000) Practice parameter: thymectomy for autoimmune myasthenia gravis (an evidence-based review): report of the quality standards subcommittee of the American academy of neurology. Neurology 55(1):7–15. https://doi.org/10.1212/WNL.55.1.7
Narayanaswami P, Sanders DB, Wolfe G et al (2021) International consensus guidance for management of myasthenia gravis: 2020 update. Neurology 96(3):114–122. https://doi.org/10.1212/WNL.0000000000011124
Lo CM, Lu HI, Hsieh MJ, Lee SS, Chang JP (2014) Thymectomy for myasthenia gravis: video-assisted versus transsternal. J Formos Med Assoc 113(10):722–726. https://doi.org/10.1016/j.jfma.2014.05.010
Landreneau RJ, Dowling RD, Castillo WM, Ferson PF (1992) Thoracoscopic resection of an anterior mediastinal tumor. Ann Thorac Surg 54(1):142–144. https://doi.org/10.1016/0003-4975(92)91162-3
Qi K, Wang B, Wang B, Bin ZL, Chu XY (2016) Video-assisted thoracoscopic surgery thymectomy versus open thymectomy in patients with myasthenia gravis: a meta-analysis. Acta Chir Belg 116(5):282–288. https://doi.org/10.1080/00015458.2016.1176419
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. https://doi.org/10.1136/bmj.b2700
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205. https://doi.org/10.1097/01.SLA.0000133083.54934.AE
Osserman KE, Genkins G (1971) Studies in myasthenia gravis: review of a twenty-year experience in over 1200 patients. Mt Sinai J Med 38(6):497–537
Jaretzki A, Barohn RJ, Ernstoff RM et al (2000) Myasthenia gravis: recommendations for clinical research standards. Task force of the medical scientific advisory board of the myasthenia gravis foundation of America. Ann Thorac Surg 70(1):327–334. https://doi.org/10.1016/S0003-4975(00)01595-2
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14(1):1–13. https://doi.org/10.1186/1471-2288-14-135
Higgins JPTGS (eds) Cochrane handbook for systematic reviews of interventions version 5.1.0. The Cochrane Collaboration
Lau J, Ioannidis JPA, Terrin N, Schmid CH, Olkin I (2006) The case of the misleading funnel plot. Br Med J. https://doi.org/10.1136/bmj.333.7568.597
Bachmann K, Burkhardt D, Schreiter I et al (2008) Long-term outcome and quality of life after open and thoracoscopic thymectomy for myasthenia gravis: analysis of 131 patients. Surg Endosc Other Interv Tech 22(11):2470–2477. https://doi.org/10.1007/s00464-008-9794-2
Bagheri R, Boonstani R, Sadrizadeh A et al (2018) Thymectomy for nonthymomatous myasthenia gravis: comparison of video-assisted thoracoscopic and transsternal thymectomy. Innov Technol Tech Cardiothorac Vasc Surg 13(2):77–80. https://doi.org/10.1097/IMI.0000000000000490
Brenna G, Antozzi C, Montomoli C, Baggi F, Mantegazza R (2017) A propensity score analysis for comparison of T-3b and VATET in myasthenia gravis. Neurology 89(2):189–195. https://doi.org/10.1212/WNL.0000000000004082
Cakar F, Werner P, Augustin F et al (2007) A comparison of outcomes after robotic open extended thymectomy for myasthenia gravis. Eur J Cardio-thoracic Surg 31(3):501–505. https://doi.org/10.1016/j.ejcts.2006.12.016
Chang PC, Chou SH, Kao EL et al (2005) Bilateral video-assisted thoracoscopic thymectomy vs extended transsternal thymectomy in myasthenia gravis: a prospective study. Eur Surg Res 37(4):199–203. https://doi.org/10.1159/000087863
Chen Z, Zuo J, Zou J et al (2014) Cellular immunity following video-assisted thoracoscopic and open resection for non-thymomatous myasthenia gravis. Eur J Cardio-thoracic Surg 45(4):646–651. https://doi.org/10.1093/ejcts/ezt443
He Z, Zhu Q, Wen W, Chen L, Xu H, Li H (2013) Surgical approaches for stage I and II thymoma-associated myasthenia gravis: feasibility of complete video-assisted thoracoscopic surgery (VATS) thymectomy in comparison with trans-sternal resection. J Biomed Res 27(1):62–70. https://doi.org/10.7555/JBR.27.20120060
Huang CS, Cheng CY, Hsu HS et al (2011) Video-assisted thoracoscopic surgery versus sternotomy in treating myasthenia gravis: comparison by a case-matched study. Surg Today 41(3):338–345. https://doi.org/10.1007/s00595-010-4270-8
Kaufman AJ, Palatt J, Sivak M et al (2016) Thymectomy for myasthenia gravis: complete stable remission and associated prognostic factors in over 1000 cases. Semin Thorac Cardiovasc Surg 28(2):561–568. https://doi.org/10.1053/j.semtcvs.2016.04.002
Lin MW, Chang YL, Huang PM, Lee YC (2010) Thymectomy for non-thymomatous myasthenia gravis: a comparison of surgical methods and analysis of prognostic factors. Eur J Cardio-thoracic Surg 37(1):7–12. https://doi.org/10.1016/j.ejcts.2009.05.027
Liu CW, Luo M, Mei JD et al (2013) Perioperative and long-term outcome of thymectomy for myasthenia gravis: comparison of surgical approaches and prognostic analysis. Chin Med J (Engl) 126(1):34–40. https://doi.org/10.3760/cma.j.issn.0366-6999.20120874
Mantegazza R, Baggi F, Bernasconi P et al (2003) Video-assisted thoracoscopic extended thymectomy and extended transsternal thymectomy (T-3b) in non-thymomatous myasthenia gravis patients: remission after 6 years of follow-up. J Neurol Sci 212(1–2):31–36. https://doi.org/10.1016/S0022-510X(03)00087-X
Meyer DM, Herbert MA, Sobhani NC et al (2009) Comparative clinical outcomes of thymectomy for myasthenia gravis performed by extended transsternal and minimally invasive approaches. Ann Thorac Surg 87(2):385–391. https://doi.org/10.1016/j.athoracsur.2008.11.040
Muhammad MIA (2014) Thymectomy by video-assisted thoracoscopy versus open surgical techniques. Asian Cardiovasc Thorac Ann 22(4):442–447. https://doi.org/10.1177/0218492313479596
Orsini B, Santelmo N, Pages PB et al (2016) Comparative study for surgical management of thymectomy for non-thymomatous myasthenia gravis from the French national database EPITHOR. Eur J Cardio-thoracic Surg 50(3):418–422. https://doi.org/10.1093/ejcts/ezw064
Rückert JC, Walter M, Müller JM (2000) Pulmonary function after thoracoscopic thymectomy versus median sternotomy for myasthenia gravis. Ann Thorac Surg 70(5):1656–1661. https://doi.org/10.1016/S0003-4975(00)01972-X
Rückert JC, Sobel HK, Göhring S, Einhäupl KM, Müller JM (2003) Matched-pair comparison of three different approaches for thymectomy in myasthenia gravis. Surg Endosc Other Interv Tech 17(5):711–715. https://doi.org/10.1007/s00464-002-9162-6
Salim EF (2018) Role of VATS in thymectomy for non-thymomatous myasthenia gravis. J Egypt Soc Cardio-Thoracic Surg 26(3):205–211. https://doi.org/10.1016/j.jescts.2018.05.001
Shiono H, Kadota Y, Hayashi A, Okumura M (2009) Comparison of outcomes after extended thymectomy for myasthenia gravis: bilateral thoracoscopic approach versus sternotomy. Surg Laparosc Endosc Percutaneous Tech 19(6):424–427. https://doi.org/10.1097/SLE.0b013e3181c48242
Siwachat S, Tantraworasin A, Lapisatepun W, Ruengorn C, Taioli E, Saeteng S (2018) Comparative clinical outcomes after thymectomy for myasthenia gravis: thoracoscopic versus trans-sternal approach. Asian J Surg 41(1):77–85. https://doi.org/10.1016/j.asjsur.2016.09.006
Tian W, Li X, Tong H et al (2020) Surgical effect and prognostic factors of myasthenia gravis with thymomas. Thorac Cancer 11(5):1288–1296. https://doi.org/10.1111/1759-7714.13396
Toker A, Eroglu O, Ziyade S et al (2005) Comparison of early postoperative results of thymectomy: partial sternotomy vs videothoracoscopy. Thorac Cardiovasc Surg 53(2):110–113. https://doi.org/10.1055/s-2004-830424
Xie X, Gan X, Chen B et al (2016) Left- and right-sided video-assisted thoracoscopic thymectomy exhibit similar effects on myasthenia gravis. J Thorac Dis 8(1):124–132. https://doi.org/10.3978/j.issn.2072-1439.2016.01.40
Yim APC, Kay RLC, Ho JKS (1995) Video-assisted thoracoscopic thymectomy for myasthenia gravis. Chest 108(5):1440–1443. https://doi.org/10.1378/chest.108.5.1440
Zielinski M, Hauer L, Hauer J, Pankowski J, Nabialek T, Szlubowski A (2010) Comparison of complete remission rates after 5 year follow-up of three different techniques of thymectomy for myasthenia gravis. Eur J Cardio-thoracic Surg 37(5):1137–1143. https://doi.org/10.1016/j.ejcts.2009.11.029
Solis-Pazmino P, Baiu I, Lincango-Naranjo E et al (2021) Impact of the surgical approach to thymectomy upon complete stable remission rates in myasthenia gravis: a meta-analysis. Neurology 97(4):e357–e368. https://doi.org/10.1212/WNL.0000000000012153
Funding
No funding was received for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Yung Lee, Yasith Samarasinghe, Janhavi Patel, Adree Khondker, Tyler McKechnie, Nadeesha Samarasinghe, Christian Finley, Wael Hanna, Yaron Shargall, and John Agzarian have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
464_2022_9757_MOESM2_ESM.jpg
Supplementary Figure 1: Subgroup analysis of 5-year complete stable remission with non thymomatous only population. (JPG 164 KB)
464_2022_9757_MOESM3_ESM.jpg
Supplementary Figure 2: (A) Forest plot illustrating subgroup analysis of 1-year clinical improvement with non thymomatous only population (B) Forest plot illustrating unchanged or worsened disease status with non-thymomatous only population. (JPG 320 KB)
464_2022_9757_MOESM4_ESM.jpg
Supplementary Figure 3: Forest plot illustrating random effects meta-analysis of the mean difference in hospital length of stay (days) between minimally invasive surgery and open thymectomy. (JPG 410 KB)
464_2022_9757_MOESM5_ESM.jpg
Supplementary Figure 4: Forest plot illustrating random effects meta-analysis of the mean difference in blood loss (mL) between minimally invasive surgery and open thymectomy. (JPG 333 KB)
464_2022_9757_MOESM6_ESM.jpg
Supplementary Figure 5: Forest plot illustrating random effects meta-analysis of the mean difference in operation time (minutes) between minimally invasive surgery and open thymectomy. (JPG 460 KB)
464_2022_9757_MOESM7_ESM.jpg
Supplementary Figure 6: Forest plot illustrating subgroup analysis of the mean difference in hospital length of stay (days) between minimally invasive surgery and open thymectomy separated by thymomatous only, and non-thymomatous only population. (JPG 402 KB)
464_2022_9757_MOESM8_ESM.jpg
Supplementary Figure 7: Forest plot illustrating subgroup analysis of the mean difference in blood loss (mL) between minimally invasive surgery and open thymectomy separated by thymomatous only, and non-thymomatous only population. (JPG 390 KB)
464_2022_9757_MOESM9_ESM.jpg
Supplementary Figure 8: Forest plot illustrating subgroup analysis of the mean difference in operation time (minutes) between minimally invasive surgery and open thymectomy separated by thymomatous only, and non-thymomatous only population. (JPG 463 KB)
464_2022_9757_MOESM10_ESM.jpg
Supplementary Figure 9: Forest plot illustrating random effects meta-analysis comparing the incidence of operative or post-operative complications in minimally invasive surgery versus open thymectomy. (JPG 276 KB)
464_2022_9757_MOESM11_ESM.jpg
Supplementary Figure 10: Forest plot illustrating random effects meta-analysis comparing the incidence of operative or post-operative complications in minimally invasive surgery versus open thymectomy with non-thymomatous only population (JPG 186 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, Y., Samarasinghe, Y., Patel, J. et al. The short and long-term effects of open vs minimally invasive thymectomy in myasthenia gravis patients: a systematic review and meta-analysis. Surg Endosc 37, 3321–3339 (2023). https://doi.org/10.1007/s00464-022-09757-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-022-09757-y