Abstract
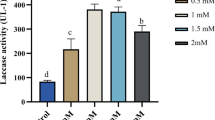
Chitosan multiple addition strategy was developed to improve laccase production from Trametes versicolor cultures. The optimized multiple addition strategy was carried out by two-time addition of 0.1 g L−1 chitosan to a 2-day-old culture media, with 24-h interval between the treatments. Under these conditions, laccase activity of 644.9 U l−1 was achieved on the seventh day and laccase production was improved by 93.5 % higher than the control. Chitosan treatment increased reactive oxygen species generation and extracellular protein concentration in the treated mycelia. In contrast, the inducer inhibited the mycelia growth. The result of the quantitative reverse transcription polymerase chain reaction showed that the copy number of the laccase gene transcript increased by 16.7-fold in the treated mycelia relative to the control. This study provides insight into some of the intrinsic metabolic processes involved in the upregulation of laccase production in the presence of chitosan inducer in fungal culture.






Similar content being viewed by others
References
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31(7):603–632
Yin H, Zhao X, Du Y (2010) Oligochitosan: a plant diseases vaccine—a review. Carbohyd Polym 82:1–8
No HK, Meyers SP, Prinyawiwatkul W, Xu Z (2007) Applications of chitosan for improvement of quality and shelf life of foods: a review. J Food Sci 72:87–100
Hadwiger LA (2013) Multiple effects of chitosan on plant systems: solid science or hype. Plant Sci 208:42–49
Kumar MNVR (2000) A review of chitin and chitosan applications. React Funct Polym 46:1–27
Maeda Y, Kimura Y (2004) Antitumor effects of various low-molecular-weight chitosans are due to increased natural killer activity of intestinal intraepithelial lymphocytes in sarcoma 180—bearing mice. J Nutr 134:945–950
Ragelle H, Vandermeulen G, Preat V (2013) Chitosan-based siRNA delivery systems. J Controll Releas 172:207–218
Raftery R, O’Brien FJ, Cryan S (2013) Chitosan for gene delivery and orthopedic tissue engineering applications. Molecules 18:5611–5647
Toivonen MS, Kurki-Suonio S, Schacher FH, Hietala S, Rojas OJ, Ikkala O (2015) Biomacromolecules. doi:10.1021/acs.biomac.5b00145
El Ghaouth A, Arul J, Asselin A, Benhamou N (1992) Antifungal activity of chitosan on post-harvest pathogens: induction of morphological and cytological alterations in Rhizopus stolonifer. Mycol Res 96:769–772
Rast DM, Baumgartner D, Mayer C, Hollenstein GO (2003) Cell wall-associated enzymes in fungi. Phytochem 64:339–366
Baldrian P (2006) Fungal laccases: occurrence and properties. FEMS Microbiol Rev 30:215–242
Rivera-Hoyos CM, Morales-Alvarez ED, Poutou-Pinales RA, Pedroza-Rodriguez AM, Rodriguez-Vazquez R, Delgado-Boada JM (2013) Fungal laccases. Fungal Biol Rev 27:67–82
Sharma P, Goel R, Capalash N (2007) Bacterial laccases. World J Microbiol Biotechnol 23:823–832
Rodríguez-Couto S, Herrera JLT (2006) Industrial and biotechnological applications of laccases: a review. Biotechnol Adv 24:500–513
Dwivedi UN, Singh P, Pandey VP, Kumar A (2011) Structure–function relationship among bacterial, fungal and plant laccases. J Molecul Catal B Enz 68:117–128
Wang F, Ma A, Guo C, Zhuang G, Liu C (2013) Ultrasound-intensified laccase production from Trametes versicolor. Ultrason Sonochem 20:118–124
Galhaup C, Wagner H, Hinterstoisser B, Haltrich D (2002) Increased production of laccase by the wood-degrading basidiomycete Trametes pubescens. Enzyme Microb Technol 30:529–536
De-Souza DGM, Tychanowicz GT, De-Souza DF, Peralta RM (2004) Production of laccase isoforms by Pleurotus pulmonarius in response to presence of phenolic and aromatic compounds. J Basic Microbiol 44(2):129–136
Wang F, Guo C, Wei T, Zhang T, Liu C (2012) Heat shock treatment improves Trametes versicolor laccase Production. Appl Biochem Biotechnol 168:256–265
Ranieri D, Colao MC, Ruzzi M, Romagnoli G, Bianchi MM (2009) Optimization of recombinant fungal laccase production with strains of the yeast Kluyveromyces lactis from the pyruvate decarboxylase promoter. FEMS Yeast Res 9:892–902
Wang F, Hu J, Guo C, Liu C (2014) Enhanced laccase production by Trametes versicolor using corn steep liquor as both nitrogen source and inducer. Biores Technol 166:602–605
Osma JF, Toca-Herrera JL, Rodrıguez-Couto S (2011) Cost analysis in laccase production. J Environ Management 92:2907–2912
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem 72:248–254
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Brennan T, Frenkel C (1977) Involvement of hydrogen peroxide in the regulation of senescence in pear. Plant Physiol 59:411–416
Gomez-Toribio V, Garcıa-Martın AB, Martınez MJ, Martınez AT, Guillen F (2009) Induction of extracellular hydroxyl radical production by white-rot fungi through quinone redox cycling. Appl Environ Microbiol 75:3944–3953
Elstner EF, Heupel A (1976) Inhibition of nitrite formation from hydroxylammonium chloride: a simple assay for superoxide dismutase. Anal Biochem 70:616–620
Palma-Guerrero J, Jansson HB, Salinas J, Lopez-Llorca LV (2008) Effect of chitosan on hyphal growth and spore germination of plant pathogenic and biocontrol fungi. J Applied Microbiol 104:541–553
Palma-Guerrero J, Lopez-Jimenez JA, Perez-Berna AJ, Huang IC, Jansson HB, Salinas J, Villalaín J, Read ND, Lopez-Llorca LV (2010) Membrane fluidity determines sensitivity of filamentous fungi to chitosan. Mol Microbiol 75(4):1021–1032
Bautista-Banos S, Hernandez-Lauzardo AN, Vela zquez-del Valle MG, Hernandez-Lopez M, Barka EA, Bosquez-Molina E, Wilson CL (2006) Chitosan as a potential natural compound to control pre and postharvest diseases of horticultural commodities. Crop Protection 25:108–118
El Ghaouth A, Arul J, Grenier J, Asselin A (1992) Effect of chitosan and other polyions on chitin deacetylase in Rhizopus stolonifer. Expt Mycol 16:173–177
Bocking SP, Wiebe MG, Robson GD, Hansen K, Christiansen LH, Trinci APJ (1999) Effect of branch frequency in Aspergillus oryzae on protein secretion and culture viscosity. Biotechnol Bioeng 65(6):638–848
El Ghaouth A, Arul J, Grenier J, Asselin A (1992) Antifungal activity of chitosan on two postharvest pathogens of strawberry fruits. Phytopathology 82(4):398–402
Prescott LM, Harley PJ, Klein DA (2002) Microbial growth. In: Prescott LM, Harley PJ, Klein DA (eds) Microbiology vol 5. The McGraw-Hill Companies, US, pp 125–128
Calvo AM, Wilson RA, Bok JW, Keller NP (2002) Relationship between secondary metabolism and fungal development. Microbiol Molecul Boiol Rev 66(3):447–459
Jaszek M, Grzywnowicz K, Malarczyk E, Leonowicz A (2006) Enhanced extracellular laccase activity as a part of the response system of white rot fungi Trametes versicolor and Abortiporus biennis to paraquat-caused oxidative stress conditions. Pestic Biochem Physiol 85:147–154
Taillandier P, Joannis-Cassan C, Jentzer JB, Gautier S, Sieczkowski N, Granes D, Brandam C (2014) Effect of a fungal chitosan preparation on Brettanomyces bruxellensis, a wine contaminant. J App Microbiol 118:123–131
Basto C, Silva CJ, Gubitz G, Cavaco-Paulo A (2007) Stability and decolourization ability of Trametes villosa laccase in liquid ultrasonic fields. Ultrason Sonochem 14:355–362
Nützmann H, Reyes-Dominguez Y, Scherlach K, Schroeckh K, Horn F, Gacek A, Schümann H, Hertweck C, Strauss J, Brakhage AA (2011) Bacteria-induced natural product formation in the fungus Aspergillus nidulans requires Saga/Ada-mediated histone acetylation. Proc Natl Acad Sci 108(34):14282–14287
Cap M, Vachov L, Palkova Z (2012) Reactive oxygen species in the signaling and adaptation of multicellular microbial communities. Oxid Med Cell Longev. doi:10.1155/2012/976753
Eyre C, Muftah W, Hiscox J, Hunt J, Kille P, Boddy L, Rogers HJ (2010) Microarray analysis of differential gene expression elicited in Trametes versicolor during interspecific mycelia interactions. Fungal boil 114:646–660
Coman C, Mot AC, Gal E, Parvu M, Silaghi-Dumitrescu R (2013) Laccase is upregulated via stress pathways in the phytopathogenic fungus Sclerotinia sclerotiorum. Fungal boil 117:528–539
Viswanath B, Rajesh B, Janardhan A, Kumar AP, Narasimha G (2014) Fungal laccases and their applications in bioremediation. Enzyme Res. doi:10.1155/2014/163242
Yaver DS, Xu F, Golightly EJ, Brown KM, Brown SH, Rey MW, Schneider P, Halkier T, Mondorf K, Dalboge H (1996) The purification, characterization, molecular cloning and expression of two laccase genes from the white-rot basidiomycete Trametes villosa. Appl Environ Microbiol 62:834–841
Soden DM, Dobson ADW (2001) Differential regulation of laccase gene expression in Pleurotus sajor-caju. Microbiology 147:1755–1763
Acknowledgments
This work was financially supported by the National Basic Research Program (973 Program) of China (No. 2013CB733600), the National Natural Science Foundation of China (no. 21206177), the National Key Technology Research and Development Program of China (No. 2012BAK25B01), the National High Technology Research and Development Program (863 Program) of China (No. 2012AA101803), and the National Key Laboratory of Biochemical Engineering Grant for Young Scientists.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adekunle, A.E., Wang, F., Hu, J. et al. Chitosan multiple addition enhances laccase production from Trametes versicolor . Bioprocess Biosyst Eng 38, 1973–1981 (2015). https://doi.org/10.1007/s00449-015-1438-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-015-1438-z