Abstract
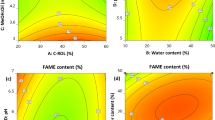
The synthesis of d-isoascorbyl stearate from d-isoascorbic acid and stearic acid with immobilized lipase (Novozym®435) as catalyst was studied. Response surface methodology and Box-Behnken design with six variables and three levels were employed to evaluate the effects of processing conditions on the conversion of d-isoascorbic acid. The results confirmed that the response surface method and statistical analysis were proved to be useful in developing optimal conditions for d-isoascorbyl stearate synthesis. The optimum conditions were predicted as follows: reaction temperature 48 °C, reaction time 17.7 h, immobilized lipase amount 50.0 % (w/w, of d-isoascorbic acid), substrate molar ratio 9:1 (stearic acid to d-isoascorbic acid), d-isoascorbic acid concentration 0.14 mol/L (based on solvent), 4A molecular sieve addition 200 g/L (based on solvent), and the optimal conversion was 90.6 %. Through the kinetics model fitting of the esterification, it was considered that the esterification conformed to a Ping–Pong bi–bi kinetic model with d-isoascorbic acid inhibition, and the obtained kinetic constants showed that the inhibition of d-isoascorbic acid and the enzyme affinity to substrate were abate with the increase of the reaction temperature.







Similar content being viewed by others
References
Andersen FA (2005) Final report of the safety assessment of l-ascorbic acid, calcium ascorbate, magnesium ascorbate, magnesium ascorbyl phosphate, sodium ascorbate, and sodium ascorbyl phosphate as used in cosmetics. Int J Toxicol 24:51–111
Chang SW, Yang CJ, Chen FY, Akoh CC, Shieh CJ (2009) Optimized synthesis of lipase-catalyzed l-ascorbyl laurate by Novozym®435. J Mol Catal B-Enzym 56:7–12
Liu XY, Guo FL, Wu LM, Liu YC, Liu ZL (1996) Remarkable enhancement of antioxidant activity of vitamin C in an artificial bilayer by making it lipo-soluble. Chem Phys Lipids 83:39–43
Zheng DG, Xiao ZP, Ye HD (2008) Synthesis of saturated alkanoyl-6-O-d-isoascorbates. Chin J Synth Chem 16:99–101
Zheng DG, Ye HD, Wu T, Liu F, Xiao ZP (2007) Synthesis of d-isoascorbyl myristate. Chem Res Appl 19:104–105
Zheng DG, Ye HD, Ye Q, Liu F, Wu T (2007) Synthesis of d-isoascorbyl caprate and its solubility test. Chem Reag 29:157–158
Sun WJ, Zhao HX, Cui FJ, Li YH, Yu SL, Zhou Q, Qian JY, Dong Y (2013) d-isoascorbyl palmitate: lipase-catalyzed synthesis, structural characterization and process optimization using response surface methodology. Chem Cent J 7:114–126
Zhang XL, Zhu XH, Zheng DG, Yu SL (2013) Lipase-catalyzed synthesis of d-isoascorbyl laurate. Chem World 54:293–296
Lerin LA, Richetti A, Dallago R, Treichel H, Mazutti MA, Oliveira JV, Antunes OAC, Oestreicher EG, De OD (2012) Enzymatic synthesis of ascorbyl palmitate in organic solvents: process optimization and kinetic evaluation. Food Bioprocess Tech 5:1068–1076
Burham H, Gafoor RA, Rasheed A, Noor NM, Badruddin S, Sidek H (2009) Enzymatic synthesis of palm-based ascorbyl esters. J Mol Catal B-Enzym 58:153–157
Xu FJ, Tan TW (2005) Biological synthesis of l-ascorbyl palmitate. Chin J Biotechnol 21:988–992
Zhang DH, Li C, Xie LL, Yuwen LX (2013) Enzymatic synthesis of l-ascorbyl laurate in DMSO-acetone mixed solvent. Ind Eng Chem Res 52:11875–11879
Gao J, Jiang YJ, Huang ZH, Zhou LY (2007) Evaluation of kinetic parameters for enzymatic interesterification synthesis of l-ascorbyl lactate by response surface methodology. Appl Biochem Biotech 136:153–164
Huang ZL, He BR, Mo C (2004) Study on the synthesis of l-ascorbyl benzoate and its property. Food Sci 25:87–90
Bezbradica D, Stojanović M, Veličković D, Dimitrijević A, Carević M, Mihailović M, Milosavić N (2013) Kinetic model of lipase-catalyzed conversion of ascorbic acid and oleic acid to liposoluble vitamin C ester. Biochem Eng J 71:89–96
Kuwabara K, Watanabe Y, Adachi S, Nakanishi K, Matsuno R (2003) Synthesis of 6-O-unsaturated acyl l-ascorbates by immobilized lipase in acetone in the presence of molecular sieve. Biochem Eng J 16:17–22
Song QX, Zhao Y, Xu WQ, Zhou WY, Wei DZ (2006) Enzymatic synthesis of l-ascorbyl linoleate in organic media. Bioprocess Biosyst Eng 28:211–215
Watanabe Y, Sawahara Y, Nosaka H, Yamanaka K, Adachi S (2008) Enzymatic synthesis of conjugated linoleoyl ascorbate in acetone. Biochem Eng J 40:368–372
Zhang DH, Li C, Zhi GY (2013) Kinetic and thermodynamic investigation of enzymatic l-ascorbyl acetate synthesis. J Biotechnol 168:416–420
Hans M, Keul H, Moeller M (2009) Ring-opening polymerization of DD-lactide catalyzed by novozyme 435. Macromol Biosci 9:239–247
Tamayo JJ, Ladero M, Santos VE, Garcia-Ochoa F (2012) Esterification of benzoic acid and glycerol to alpha-monobenzoate glycerol in solventless media using an industrial free Candida antarctica lipase B. Process Biochem 47:243–250
Gayot S, Santarelli X, Coulon D (2003) Modification of flavonoid using lipase in non-conventional media: effect of the water content. J Biotechnol 101:29–36
Srinivasan K, Devi KV (2010) Characterization of l-ascorbic acid single crystals grown from solution with different solvents. Cryst Res Technol 45:946–952
Acknowledgments
This research was financially supported by Technology Breakthrough Major Project in Henan Province (Grant No: 112101210200).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Z., Shen, Y., Li, W. et al. Process optimization and kinetic evaluation for biosynthesis of d-isoascorbyl stearate. Bioprocess Biosyst Eng 38, 833–839 (2015). https://doi.org/10.1007/s00449-014-1326-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-014-1326-y