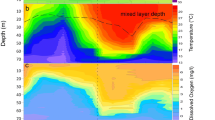
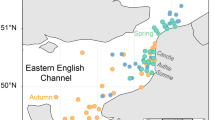
Abstract
Marine food webs are strongly size-structured and size-based analysis of communities is a useful approach to evaluate food webs in a way that can be compared across systems. Fatty acid analysis is commonly used to identify diet sources of species, offering a powerful complement to stable isotopes, but is rarely applied to size-structured communities. In this study, we used fatty acids and stable isotopes to characterize size-based variation in prey resources and trophic pathways over a nine-month temperate coastal ocean time series of seven plankton size classes, from > 0.7-μm particulate organic matter through > 2000-μm zooplankton. Zooplankton size classes were generally distinguishable by their dietary fatty acids, while stable isotopes revealed more seasonal variability. Fatty acids of zooplankton were correlated with those of their prey (particulate organic matter and smaller zooplankton) and identified trophic pathways, including widespread ties to the microbial food web. Diatom fatty acids also contributed to zooplankton but fall blooms were more important than spring. Concurrent isotope-based trophic position estimates and fatty acid markers of carnivory showed that some indicators (18:1ω9/18:1ω7) are not consistent across size classes, while others (DHA:EPA) are relatively reliable. Both analysis methods provided distinct information to build a more robust understanding of resource use. For example, fatty acid markers showed that trophic position was likely underestimated in 250-μm zooplankton, probably due to their consumption of protists with low isotopic fractionation factors. Applying fatty acid analysis to a size-structured framework provides more insight into trophic pathways than isotopes alone.





Similar content being viewed by others
Data and code availability
Data and code are provided at https://github.com/Pelagic-Ecosystems/QU39zoops2015.
References
Alfaro AC, Thomas F, Sergent L, Duxbury M (2006) Identification of trophic interactions within an estuarine food web (northern New Zealand) using fatty acid biomarkers and stable isotopes. Estuar Coast Shelf Sci 70:271–286. https://doi.org/10.1016/j.ecss.2006.06.017
Barnes C, Maxwell D, Reuman DC, Jennings S (2010) Global patterns in predator–prey size relationships reveal size dependency of trophic transfer efficiency. Ecology 91:222–232
Bell MV, Tocher DR (2009) Biosynthesis of polyunsaturated fatty acids in aquatic ecosystems: general pathways and new directions. Lipids in aquatic ecosystems. Springer, New York, pp 211–236
Budge SM, Parrish CC (1998) Lipid biogeochemistry of plankton, settling matter and sediments in Trinity Bay, Newfoundland II. Fatty Acids. Org Geochem 29:1547–1559. https://doi.org/10.1016/S0146-6380(98)00177-6
Budge SM, Iverson SJ, Bowen WD, Ackman RG (2002) Among- and within-species variability in fatty acid signatures of marine fish and invertebrates on the Scotian Shelf, Georges Bank, and southern Gulf of St. Lawrence. Can J Fish Aquat Sci 59:886–898
Cabrol J, Trombetta T, Amaudrut S et al (2019) Trophic niche partitioning of dominant North-Atlantic krill species, Meganyctiphanes norvegica, Thysanoessa inermis, and T. raschii. Limnol Oceanogr 64:165–181. https://doi.org/10.1002/lno.11027
Colombo SM, Wacker A, Parrish CC et al (2017) A fundamental dichotomy in long-chain polyunsaturated fatty acid abundance between and within marine and terrestrial ecosystems. Environ Rev 25:163–174. https://doi.org/10.1139/er-2016-0062
Costalago D, Forster I, Nemcek N et al (2020) Seasonal and spatial dynamics of the planktonic trophic biomarkers in the Strait of Georgia (northeast Pacific) and implications for fish. Sci Rep 10:8517. https://doi.org/10.1038/s41598-020-65557-1
Dalsgaard J, St. John M, Kattner G et al (2003) Fatty acid trophic markers in the pelagic marine environment. Advances in marine biology. Academic Press, London, pp 225–340
Décima M (2022) Zooplankton trophic structure and ecosystem productivity. Mar Ecol Prog Ser 692:23–42. https://doi.org/10.3354/meps14077
Décima M, Landry MR, Bradley CJ, Fogel ML (2017) Alanine δ15N trophic fractionation in heterotrophic protists. Limnol Oceanogr 62:2308–2322. https://doi.org/10.1002/lno.10567
Del Bel Belluz BJ, Peña MA, Jackson JM, Nemcek N (2021) Phytoplankton composition and environmental drivers in the Northern Strait of Georgia (Salish Sea), British Columbia, Canada. Estuar Coasts 44:1419–1439. https://doi.org/10.1007/s12237-020-00858-2
El-Sabaawi R, Dower JF, Kainz M, Mazumder A (2009a) Characterizing dietary variability and trophic positions of coastal calanoid copepods: insight from stable isotopes and fatty acids. Mar Biol 156:225–237. https://doi.org/10.1007/s00227-008-1073-1
El-Sabaawi R, Dower J, Kainz M, Mazumder A (2009b) Interannual variability in fatty acid composition of the copepod Neocalanus plumchrus in the Strait of Georgia, British Columbia. Mar Ecol Prog Ser 382:151–161. https://doi.org/10.3354/meps07915
El-Sabaawi RW, Sastri AR, Dower JF, Mazumder A (2010) Deciphering the seasonal cycle of copepod trophic dynamics in the Strait of Georgia, Canada, using stable isotopes and fatty acids. Estuar Coasts 33:738–752. https://doi.org/10.1007/s12237-009-9263-8
Falk-Petersen S, Hagen W, Kattner G et al (2000) Lipids, trophic relationships, and biodiversity in Arctic and Antarctic krill. Can J Fish Aquat Sci 57:178–191. https://doi.org/10.1139/f00-194
Fortier L, Le Fèvre J, Legendre L (1994) Export of biogenic carbon to fish and to the deep ocean: the role of large planktonic microphages. J Plankton Res 16:809–839. https://doi.org/10.1093/plankt/16.7.809
Fraser AJ, Sargent JR, Gamble JC, Seaton DD (1989) Formation and transfer of fatty acids in an enclosed marine food chain comprising phytoplankton, zooplankton and herring (Clupea harengus L.) larvae. Mar Chem 27:1–18. https://doi.org/10.1016/0304-4203(89)90024-8
Galloway AWE, Lowe AT, Sosik EA et al (2013) Fatty acid and stable isotope biomarkers suggest microbe-induced differences in benthic food webs between depths. Limnol Oceanogr 58:1451–1462. https://doi.org/10.4319/lo.2013.58.4.1451
Galloway AWE, Winder M (2015) Partitioning the relative importance of phylogeny and environmental conditions on phytoplankton fatty acids. PLoS ONE 10:e0130053
Gladyshev MI, Sushchik NN, Makhutova ON et al (2010) Correlations between fatty acid composition of seston and zooplankton and effects of environmental parameters in a eutrophic Siberian reservoir. Limnologica 40:343–357. https://doi.org/10.1016/j.limno.2009.12.004
Graeve M, Kattner G (1992) Species-specific differences in intact wax esters of Calanus hyperboreus and C. finmarchicus from Fram Strait—Greenland Sea. Mar Chem 39:269–281. https://doi.org/10.1016/0304-4203(92)90013-Z
Graeve M, Kattner G, Hagen W (1994) Diet-induced changes in the fatty acid composition of Arctic herbivorous copepods: experimental evidence of trophic markers. J Exp Mar Biol Ecol 182:97–110
Graeve M, Albers C, Kattner G (2005) Assimilation and biosynthesis of lipids in Arctic Calanus species based on feeding experiments with a 13C labelled diatom. J Exp Mar Biol Ecol 317:109–125. https://doi.org/10.1016/j.jembe.2004.11.016
Heneghan RF, Everett JD, Sykes P et al (2020) A functional size-spectrum model of the global marine ecosystem that resolves zooplankton composition. Ecol Model 435:109265. https://doi.org/10.1016/j.ecolmodel.2020.109265
Henschke N, Everett JD, Suthers IM et al (2015) Zooplankton trophic niches respond to different water types of the western Tasman Sea: a stable isotope analysis. Deep Sea Res Part I Oceanogr Res Pap 104:1–8. https://doi.org/10.1016/j.dsr.2015.06.010
Hiltunen M, Strandberg U, Brett MT et al (2022) Taxonomic, temporal, and spatial variations in zooplankton fatty acid composition in Puget Sound, WA, USA. Estuar Coasts 45:567–581. https://doi.org/10.1007/s12237-021-00973-8
Hobson KA (1999) Tracing the origins and migration of wildlife using stable isotopes: a review. Oecologia 120:314–326
Holm-Hansen O, Lorenzen CJ, Holmes RW, Strickland JDH (1965) Fluorometric determination of chlorophyll. ICES J Mar Sci 30:3–15. https://doi.org/10.1093/icesjms/30.1.3
Im D-H, Suh H-L (2016) Ontogenetic feeding migration of the euphausiid Euphausia pacifica in the East Sea (Japan Sea) in autumn: a stable isotope approach. J Plankton Res 38:904–914. https://doi.org/10.1093/plankt/fbw041
Irigoien X, Flynn KJ, Harris RP (2005) Phytoplankton blooms: a ‘loophole’ in microzooplankton grazing impact? J Plankton Res 27:313–321. https://doi.org/10.1093/plankt/fbi011
Jónasdóttir SH (2019) Fatty acid profiles and production in marine phytoplankton. Mar Drugs 17:151. https://doi.org/10.3390/md17030151
Kainz M, Arts MT, Mazumder A (2004) Essential fatty acids in the planktonic food web and their ecological role for higher trophic levels. Limnol Oceanogr 49:1784–1793. https://doi.org/10.4319/lo.2004.49.5.1784
Kaneda T (1991) Iso- and anteiso-fatty acids in bacteria: biosynthesis, function, and taxonomic significance. Microbiol Rev 55:288–302. https://doi.org/10.1128/mr.55.2.288-302.1991
Kenitz KM, Visser AW, Ohman MD et al (2019) Community trait distribution across environmental gradients. Ecosystems 22:968–980. https://doi.org/10.1007/s10021-018-0314-5
Klein Breteler WCM, Schogt N, Baas M et al (1999) Trophic upgrading of food quality by protozoans enhancing copepod growth: role of essential lipids. Mar Biol 135:191–198. https://doi.org/10.1007/s002270050616
Lee RF, Hagen W, Kattner G (2006) Lipid storage in marine zooplankton. Mar Ecol Prog Ser 307:273–306. https://doi.org/10.3354/meps307273
Mahara N, Pakhomov EA, Jackson JM, Hunt BP (2019) Seasonal zooplankton development in a temperate semi-enclosed basin: two years with different spring bloom timing. J Plankton Res 41:309–328. https://doi.org/10.1093/plankt/fbz016
McLaskey AK, Forster I, Del Bel BJ, Hunt BPV (2022) A high-resolution time series of particulate matter fatty acids reveals temporal dynamics of the composition and quality available to zooplankton in a temperate coastal ocean. Prog Oceanogr 206:102843. https://doi.org/10.1016/j.pocean.2022.102843
Miller JA, Peterson WT, Copeman LA et al (2017) Temporal variation in the biochemical ecology of lower trophic levels in the Northern California Current. Prog Oceanogr 155:1–12. https://doi.org/10.1016/j.pocean.2017.05.003
Minagawa M, Wada E (1984) Stepwise enrichment of 15N along food chains: further evidence and the relation between δ15N and animal age. Geochim Cosmochim Acta 48:1135–1140. https://doi.org/10.1016/0016-7037(84)90204-7
Mitra A, Castellani C, Gentleman WC et al (2014) Bridging the gap between marine biogeochemical and fisheries sciences; configuring the zooplankton link. Prog Oceanogr 129:176–199. https://doi.org/10.1016/j.pocean.2014.04.025
Nyssen F, Brey T, Dauby P, Graeve M (2005) Trophic position of Antarctic amphipods—enhanced analysis by a 2-dimensional biomarker assay. Mar Ecol Prog Ser 300:135–145
Olson EM, Allen SE, Do V et al (2020) Assessment of nutrient supply by a tidal jet in the northern strait of Georgia based on a biogeochemical model. J Geophys Res Oceans 125:8
O’Reilly CM, Hecky RE, Cohen AS, Plisnier P-D (2002) Interpreting stable isotopes in food webs: Recognizing the role of time averaging at different trophic levels. Limnol Oceanogr 47:306–309. https://doi.org/10.4319/lo.2002.47.1.0306
Park JY, Jung J-H, Kwak JH et al (2021) Trophic enrichment factors of carbon and nitrogen isotopic ratios (Δ13C and Δ15N) in four marine ciliates. Front Microbiol 12:721157
Parrish CC, French VM, Whiticar MJ (2012) Lipid class and fatty acid composition of copepods (Calanus finmarchicus, C. glacialis, Pseudocalanus sp., Tisbe furcata and Nitokra lacustris) fed various combinations of autotrophic and heterotrophic protists. J Plankton Res 34:356–375. https://doi.org/10.1093/plankt/fbs003
Peña MA, Masson D, Callendar W (2016) Annual plankton dynamics in a coupled physical–biological model of the Strait of Georgia, British Columbia. Prog Oceanogr 146:58–74. https://doi.org/10.1016/j.pocean.2016.06.002
Post DM (2002) Using stable isotopes to estimate trophic postition: models, methods, and assumptions. Ecology 83:703–718
Preikshot D, Beamish RJ, Neville CM (2013) A dynamic model describing ecosystem-level changes in the Strait of Georgia from 1960 to 2010. Prog Oceanogr 115:28–40
Reuss N, Poulsen L (2002) Evaluation of fatty acids as biomarkers for a natural plankton community. A field study of a spring bloom and a post-bloom period off West Greenland. Mar Biol 141:423–434. https://doi.org/10.1007/s00227-002-0841-6
Rolff C (2000) Seasonal variation in δ13C and δ15N of size-fractionated plankton at a coastal station in the northern Baltic proper. Mar Ecol Prog Ser 203:47–65
Schoo KL, Boersma M, Malzahn AM et al (2018) Dietary and seasonal variability in trophic relations at the base of the North Sea pelagic food web revealed by stable isotope and fatty acid analysis. J Sea Res 141:61–70. https://doi.org/10.1016/j.seares.2018.08.004
Stanley-Samuelson DW (1994) The biological significance of prostaglandins and related eicosanoids in invertebrates. Am Zool 34:589–598. https://doi.org/10.1093/icb/34.6.589
Stevens C, Sahota R, Galbraith M et al (2022) Total lipid and fatty acid composition of mesozooplankton functional group members in the NE Pacific over a range of productivity regimes. Mar Ecol Prog Ser 687:43–64. https://doi.org/10.3354/meps14004
Stevens CJ, Deibel D, Parrish CC (2004) Incorporation of bacterial fatty acids and changes in a wax ester-based omnivory index during a long-term incubation experiment with Calanus glacialis Jaschnov. J Exp Mar Biol Ecol 303:135–156
Suchy KD, Young K, Galbraith M et al (2022) Match/mismatch between phytoplankton and crustacean zooplankton phenology in the Strait of Georgia, Canada. Front Mar Sci 9:832684
Taipale S, Kankaala P, Hämäläinen H, Jones RI (2009) Seasonal shifts in the diet of lake zooplankton revealed by phospholipid fatty acid analysis. Freshw Biol 54:90–104
Taipale SJ, Kahilainen KK, Holtgrieve GW, Peltomaa ET (2018) Simulated eutrophication and browning alters zooplankton nutritional quality and determines juvenile fish growth and survival. Ecol Evol 8:2671–2687. https://doi.org/10.1002/ece3.3832
Acknowledgements
We thank the Hakai Institute’s Quadra Island field station for their time and energy in the field collection and curation of oceanographic data, and the staff at the Pacific Science Enterprise Center for fatty acid analyses.
Funding
A. McLaskey was funded by the Tula-Mitacs Canada Grant IT13677. Funding for this research was provided by the Tula Foundation and B. Hunt’s Natural Sciences and Engineering Research Council (NSERC) Grant No. RGPIN-2017-04499.
Author information
Authors and Affiliations
Contributions
AKM and BPVH conceptualized the study; IF and BPVH acquired funding and provided resources; AKM curated the data, did the analysis and visualization, and wrote the original draft. AKM, IF, and BPVH reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Maarten Boersma.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McLaskey, A.K., Forster, I. & Hunt, B.P.V. Distinct trophic ecologies of zooplankton size classes are maintained throughout the seasonal cycle. Oecologia 204, 227–239 (2024). https://doi.org/10.1007/s00442-023-05501-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-023-05501-y