Abstract
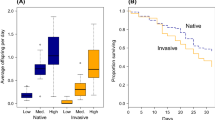
Invasive species can have large effects on native communities. When native and invasive species share parasites, an epidemic in a native species could facilitate or inhibit the invasion. We sought to understand how the incidence and timing of epidemics in native species caused by a generalist parasite influenced the success and impact of an invasive species. We focused on North American native and invasive species of zooplankton (Daphnia dentifera and Daphnia lumholtzi, respectively), that can both become infected with a fungal parasite (Metschnikowia bicuspidata). In a laboratory microcosm experiment, we exposed the native species to varying parasite inocula (none, low, high) and two invasive species introduction times (before or during an epidemic in the native species). We found that the invasive species density in treatments with the parasite was higher compared to uninfected treatments, though only the early invasion, low-parasite and uninfected treatments exhibited significant pairwise differences. However, invasive resting eggs were only found in the uninfected treatments. The density of the native species was lowest with a combination of the parasite present, and the invasive species introduced during the epidemic. Native infection prevalence in these treatments (late invasion, parasite present) was also higher than prevalence in treatments where the invasive species was introduced before the epidemic. Therefore, the timing of an invasion relative to an epidemic can affect both the native and invasive species. Our results suggest that the occurrence and timing of epidemics in native species can influence the impacts of a species invasion.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available on the repository osf.io (https://osf.io/n69r7).
Code availability
The code developed during this study is available on the repository osf.io (https://osf.io/n69r7).
References
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Statistical Software. https://doi.org/10.18637/jss.v067.i01
Benzie JAH (1988) The systematics of Australian daphnia (Cladocera: daphniidae). Multivariate Morphometrics Hydrobiologia 166:163–182. https://doi.org/10.1007/BF00028633
Brooks ME, Kristensen K, van Benthem KJ et al (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal 9:378–400
Byers JE (2002) Impact of non-indigenousspecies on natives enhanced by anthropogenic alteration regimes of selection anthropogenic. 449–458
Cáceres CE (1998) Interspecific variation in the abundance, production, and emergence of Daphnia diapausing eggs. Ecology 79:1699–1710. https://doi.org/10.2307/176789
Callaway RM, Ridenour WM (2004) Novel weapons: Invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436–443. https://doi.org/10.1890/1540-9295(2004)002[0436:NWISAT]2.0.CO;2
Civitello DJ, Penczykowski RM, Smith AN et al (2015) Resources, key traits and the size of fungal epidemics in Daphnia populations. J Anim Ecol 84:1010–1017. https://doi.org/10.1111/1365-2656.12363
Colautti RI, Ricciardi A, Grigorovich IA, MacIsaac HJ (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733. https://doi.org/10.1111/j.1461-0248.2004.00616.x
Duffy MA, Hall SR (2008) Selective predation and rapid evolution can jointly dampen effects of virulent parasites on Daphnia populations. Am Nat 171:499–510. https://doi.org/10.1086/528998
Dunn AM, Hatcher MJ (2015) Parasites and biological invasions: Parallels, interactions, and control. Trends Parasitol 31:189–199. https://doi.org/10.1016/j.pt.2014.12.003
Dunn AM, Torchin ME, Hatcher MJ et al (2012) Indirect effects of parasites in invasions. Funct Ecol 26:1262–1274. https://doi.org/10.1111/j.1365-2435.2012.02041.x
Ebert D, Lipsitch M, Mangin KL (2000) The effect of parasites on host population density and extinction: experimental epidemiology with Daphnia and six microparasites. Am Nat 156:459–477. https://doi.org/10.1086/303404
Elton C (1958) The ecology of invasions by animals and plants. London: Methuen. Prog Phys Geogr 31:659–666. https://doi.org/10.1177/0309133307087089
Faillace CA, Lorusso NS, Duffy S (2017) Overlooking the smallest matter: viruses impact biological invasions. Ecol Lett 20:524–538. https://doi.org/10.1111/ele.12742
Gill NS, Mahood AL, Meier CL et al (2021) Six central questions about biological invasions to which NEON data science is poised to contribute. Ecosphere. https://doi.org/10.1002/ecs2.3728
Guo Q, Songlin F, Dukes J et al (2015) A unified approach for quantifying invasibility and degree of invasion. Ecology 96:2613–2621
Hall SR, Becker CR, Duffy MA, Cáceres CE (2011) Epidemic size determines population-level effects of fungal parasites on Daphnia hosts. Oecologia 166:833–842. https://doi.org/10.1007/s00442-011-1905-4
Hartig F (2022) DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models
Havel JE, Hebert PDN (1993) Daphnia lumholtzi in North America: another exotic zooplankter. Limnol Oceanogr 38:1823–1827. https://doi.org/10.4319/lo.1993.38.8.1823
Havel JE, Kovalenko KE, Thomaz SM et al (2015) Aquatic invasive species: challenges for the future. Hydrobiologia 750:147–170. https://doi.org/10.1007/s10750-014-2166-0
Hebert PDN (1995) The Daphnia of North America: an illustrated faun. University of Geulph, CyberNatural Software
Hudson PJ, Dobson AP (1989) Population biology of Trichostrongy1us tenuis, a parasite of economic importance for red grouse management. Parasitol Today 5:283–291. https://doi.org/10.1016/0169-4758(89)90019-7
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170. https://doi.org/10.1016/S0169-5347(02)02499-0
Knevel IC, Lans T, Menting FBJ et al (2004) Release from native root herbivores and biotic resistance by soil pathogens in a new habitat both affect the alien Ammophila arenaria in South Africa. Oecologia 141:502–510. https://doi.org/10.1007/s00442-004-1662-8
Kolar CS, Boase JC, Clapp DF, Wahl DH (1997) Potential effect of invasion by an exotic zooplankter, daphnia iumholtzi. J Freshw Ecol 12:521–530. https://doi.org/10.1080/02705060.1997.9663566
Lenth RV (2022) emmeans: Estimated Marginal Means, aka Least-Squares Means
McKenzie VJ, Townsend AR (2007) Parasitic and infectious disease responses to changing global nutrient cycles. EcoHealth 4:384–396. https://doi.org/10.1007/s10393-007-0131-3
Novak SJ (2007) The role of evolution in the invasion process. Proc Natl Acad Sci USA 104:3671–3672. https://doi.org/10.1073/pnas.0700224104
Panov VE, Krylov PI, Riccardi N (2004) Role of diapause in dispersal and invasion success by aquatic invertebrates. J Limnol 63:56–69. https://doi.org/10.4081/jlimnol.2004.s1.56
Pimentel D, Zuniga R, Morrison D (2005) Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol Econ 52:273–288. https://doi.org/10.1016/j.ecolecon.2004.10.002
Prenter J, MacNeil C, Dick JTA, Dunn AM (2004) Roles of parasites in animal invasions. Trends Ecol Evol 19:385–390. https://doi.org/10.1016/j.tree.2004.05.002
Price P, Westoby M, Rice B (1988) Parasite-Mediated Competition : Some Predictions and Tests Author ( s ): Peter W . Price , Mark Westoby , Barbara Rice Published by : The University of Chicago Press for The American Society of Naturalists Stable URL : http://www.jstor.org/stable/2461743. American Society of Naturalists 131:544–555
R Core Team (2022) R: A language and environment for statistical computing
Searle CL, Cortez MH, Hunsberger KK et al (2016) Population density, not host competence, drives patterns of disease in an invaded community. Am Nat 188:554–566. https://doi.org/10.1086/688402
Searle CL, Hochstedler BR, Merrick AM et al (2018) High resources and infectious disease facilitate invasion by a freshwater crustacean. Oecologia 188:571–581. https://doi.org/10.1007/s00442-018-4237-9
Settle W, Wilson L (1990) Invasion by the Variegated Leafhopper and Biotic Interactions : Parasitism , Competition , and Apparent Competition Author ( s ): W . H . Settle and L . T . Wilson Published by : Wiley on behalf of the Ecological Society of America Stable URL : https://ww. Ecological Society of America 71:1461–1470
Smith AS, Acharya K, Jack J (2009) Overcrowding, food and phosphorus limitation effects on ephipphia production and population dynamics in the invasive species Daphnia lumholtzi. Hydrobiologia 618:47–56. https://doi.org/10.1007/s10750-008-9546-2
Tuttle LJ, Sikkel PC, Cure K, Hixon MA (2017) Parasite-mediated enemy release and low biotic resistance may facilitate invasion of Atlantic coral reefs by Pacific red lionfish (Pterois volitans). Biol Invasions 19:563–575. https://doi.org/10.1007/s10530-016-1342-8
Walsman JC, Strauss AT, Hall SR (2022) Parasite-driven cascades or hydra effects: Susceptibility and foraging depression shape parasite–host–resource interactions. Funct Ecol 36:1268–1278. https://doi.org/10.1111/1365-2435.14030
Acknowledgements
We would like to thank M. Scherer, J. Davis, and M. Sparks for help with data collection and analysis. We would like to thank B. Berggren for help with manuscript editing.
Funding
This study was funded by the National Science Foundation under the Graduate Research Fellowship under grant number DGE-1842166 (PEB) and under NSF grant number DEB-1856710 (CLS).
Author information
Authors and Affiliations
Contributions
CLS and KLJ conceived and designed the experiments. KLJ, PEB, BNH performed the experiments. BDH analyzed the data. PEB wrote the manuscript; all other authors provided editorial advice.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethics approval was not required for this study.
Consent to participate
Not applicable.
Consent to publication
All the authors approve this manuscript for submission to Oecologia.
Additional information
Communicated by Tara Merrill and Jason Hoverman.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Blackwood, P.E., Jonasen, K.L., Hoenig, B.D. et al. Epidemics in native species influence the outcome of a species invasion. Oecologia 204, 327–337 (2024). https://doi.org/10.1007/s00442-023-05444-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-023-05444-4