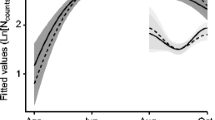
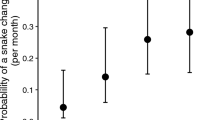
Abstract
Understanding the interactions between parasites, hosts, and their shared environment is central to ecology. Variation in infestation prevalence may be the result of varying environmental and population characteristics; however, variations in parasitism may also depend on individual characteristics that influence both the exposure and susceptibility to parasites. Using 12 years of data from a population of wild eastern chipmunks relying on pulsed food resources, we investigated the determinants of bot fly parasitism at both the population and individual level. We assessed the relationship between infestation prevalence and weather conditions, population size and food abundance. Then, we assessed the relationship between infestation intensity and chipmunk behavior, sex, age, body mass and food abundance. Precipitation, temperature and population size were positively related to infestation prevalence, while beech masts were negatively related to infestation prevalence, highlighting the importance of local environmental conditions on hosts and parasites. We also found that the influence of activity and exploration on infestation intensity varied according to sex in adults. More active and faster exploring males had more parasites compared to females, suggesting that reproductive behaviors may influence parasite exposure. For juveniles, infestation intensity was greater when juveniles emerged in the spring as opposed to fall, possibly because spring emergence is synchronized with the peak of bot fly eggs in the environment, low food availability and longer activity period. Our results suggest that the environmental, population and host characteristics that are advantageous for reproduction and resource acquisition may come at the cost of increasing parasitism.



Similar content being viewed by others
References
Adamo SA, Jensen M, Younger M (2001) Changes in lifetime immunocompetence in male and female Gryllus texensis (formerly G. integer)*: trade-offs between immunity and reproduction. Anim Behav 62:417–425. https://doi.org/10.1006/anbe.2001.1786
Anderson RM, May RM (1978) Regulation and stability of host-parasite population interactions: I. Regulatory processes. J Anim Ecol 47:219–247. https://doi.org/10.2307/3933
Anderson RM, May RM (1981) The population dynamics of microparasites and their invertebrate hosts. Philos Trans R Soc B Biol Sci 291:451–524. https://doi.org/10.1098/rstb.1981.0005
Arneberg P, Skorping A, Grenfell B, Read AF (1998) Host densities as determinants of abundance in parasite communities. Proc R Soc B 265:1283–1289. https://doi.org/10.1098/rspb.1998.0431
Baird CR (1975) Larval development of the rodent bot fly, Cuterebra tenebrosa, in bushy-tailed wood rats and its relationship to pupal diapause. Can J Zool 53:1788–1798. https://doi.org/10.1139/z75-212
Baird CR (1997) Bionomics of Cuterebra austeni (Diptera: Cuterebridae) and its association with Neotoma albigula (Rodentia: Cricetidae) in the southwestern United States. J Med Entomol 34:690–695. https://doi.org/10.1093/jmedent/34.6.690
Barber I, Dingemanse NJ (2010) Parasitism and the evolutionary ecology of animal personality. Philos Trans R Soc B Biol Sci 365:4077–4088. https://doi.org/10.1098/rstb.2010.0182
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bergeron P, Réale D, Humphries MM, Garant D (2011) Anticipation and tracking of pulsed resources drive population dynamics in eastern chipmunks. Ecology 92:2027–2034. https://doi.org/10.1890/11-0766.1
Bohn SJ, Webber QMR, Florko KRN et al (2017) Personality predicts ectoparasite abundance in an asocial sciurid. Ethology 123:761–771. https://doi.org/10.1111/eth.12651
Boonstra R, Krebs C, Beacham T (1980) Impact of botfly parasitism on Microtus townsendii populations. Can J Zool 58:1683–1692. https://doi.org/10.1139/z80-230
Bordes F, Morand S, Kelt DA, Van Vuren DH (2009) Home range and parasite diversity in mammals. Am Nat 173:467–474. https://doi.org/10.1086/597227
Bowman DD, Hendrix CM, Lindsay DS, Barr SC (2002) Feline clinical parasitology. Iowa State University Press, Armes
Boyer N, Réale D, Marmet J et al (2010) Personality, space use and tick load in an introduced population of Siberian chipmunks Tamias sibiricus. J Anim Ecol 79:538–547. https://doi.org/10.1111/j.1365-2656.2010.01659.x
Brunner JL, Ostfeld RS (2008) Multiple causes of variable tick burdens on small-mammal hosts. Ecology 89:2259–2272. https://doi.org/10.1890/07-0665.1
Burns CE, Goodwin BJ, Ostfeld RS (2005) A prescription for longer life? Bot fly parasitism of the white-footed mouse. Ecology 86:753–761. https://doi.org/10.1890/03-0735
Calenge C (2006) The package adehabitat for the R software: a tool for the analysis of space and habitat use by animals. Ecol Modell 197:516–519. https://doi.org/10.1016/j.ecolmodel.2006.03.017
Careau V, Thomas DW, Humphries MM (2010) Energetic cost of bot fly parasitism in free-ranging eastern chipmunks. Oecologia 162:303–312. https://doi.org/10.1007/s00442-009-1466-y
Catts EP (1982) Biology of new world bot flies: Cuterebridae. Annu Rev Entomol 27:313–338. https://doi.org/10.1146/annurev.en.27.010182.001525
Christe P, Arlettaz R, Vogel P (2000) Variation in intensity of a parasitic mite (Spinturnix myoti) in relation to the reproductive cycle and immunocompetence of its bat host (Myotis myotis). Ecol Lett 3:207–212. https://doi.org/10.1046/j.1461-0248.2000.00142.x
Christe P, Giorgi MS, Vogel P, Arlettaz R (2003) Differential species-specific ectoparasitic mite intensities in two intimately coexisting sibling bat species: resource-mediated host attractiveness or parasite specialization? J Anim Ecol 72:866–872. https://doi.org/10.1046/j.1365-2656.2003.00759.x
Christopher D (1972) Summer activity of eastern chipmunks. J Mamm 53:176–180. https://doi.org/10.2307/1378838
Cramer MJ, Cameron GN (2006) Effects of bot fly (Cuterebra fontinella) parasitism on a population of white-footed mice (Peromyscus leucopus). J Mammal 87:1103–1111. https://doi.org/10.1644/05-MAMM-A-388R1.1
Dingemanse NJ, Moiron M, Araya-Ajoy YG et al (2019) Individual variation in age-dependent reproduction: fast explorers live fast but senesce young? J Anim Ecol 00:1–13. https://doi.org/10.1111/1365-2656.13122
Forbes KM, Mappes T, Sironen T et al (2016) Food limitation constrains host immune responses to nematode infections. Biol Lett 12:20160471. https://doi.org/10.1098/rsbl.2016.0471
Froeschke G, Harf R, Sommer S, Matthee S (2010) Effects of precipitation on parasite burden along a natural climatic gradient in southern Africa—implications for possible shifts in infestation patterns due to global changes. Oikos 119:1029–1039. https://doi.org/10.1111/j.1600-0706.2009.18124.x
Gardner ID (1980) The effect of aging on susceptibility to infection. Rev Infect Dis 2:801–810. https://doi.org/10.1093/clinids/2.5.801
Gelman A, Su Y (2018) arm: Data analysis using regression and multilevel hierarchical models. R package version 9.01. https://cran.r-project.org/package=arm. Accessed Mar 2019
Giraldeau L-A, Kramer DL, Deslandes I, Lair H (1994) The effect of competitors and distance on central place foraging eastern chipmunks, Tamias striatus. Anim Behav 47:621–632. https://doi.org/10.1006/anbe.1994.1085
Gyuris E, Hankó JF, Feró O, Barta Z (2016) Personality and ectoparasitic mites (Hemipteroseius adleri) in firebugs (Pyrrhocoris apterus). Behav Processes 122:67–74. https://doi.org/10.1016/j.beproc.2015.11.011
Hadfield JD, Wilson AJ, Garant D et al (2010) The misuse of BLUP in ecology and evolution. Am Nat 175:116–125. https://doi.org/10.1086/648604
Hawlena H, Abramsky Z, Krasnov BR (2005) Age-biased parasitism and density-dependent distribution of fleas (Siphonaptera) on a desert rodent. Oecologia 146:200–208. https://doi.org/10.1007/s00442-005-0187-0
Houslay TM, Wilson AJ (2017) Avoiding the misuse of BLUP in behavioural ecology. Behav Ecol 28:948–952. https://doi.org/10.1093/beheco/arx023
Houston AI, McNamara JM, Barta Z, Klasing KC (2007) The effect of energy reserves and food availability on optimal immune defence. Proc R Soc B Biol Sci 274:2835–2842. https://doi.org/10.1098/rspb.2007.0934
Hudson PJ, Dobson AP, Newborn D (1998) Prevention of population cycles by parasite removal. Science (80-) 282:2256–2258. https://doi.org/10.1126/science.282.5397.2256
Humphries MM, Thomas DW, Kramer DL (2003) The role of energy availability in mammalian hibernation: a cost-benefit approach. Physiol Biochem Zool 76:180–186. https://doi.org/10.1086/367950
Hunter DM, Webster JM (1973) Aggregation behavior of adult Cuterebra grisea and C. tenebrosa (Diptera: Cuterebridae). Can Entomol 105:1301–1307. https://doi.org/10.4039/Ent1051301-10
Hunter DM, Sadleir RMFS, Webster JM (1972) Studies on the ecology of cuterebrid parasitism in deermice. Can J Zool 50:25–29. https://doi.org/10.1139/z72-005
Jaffe G, Zegers DA, Steele MA, Merritt JF (2005) Long-term patterns of botfly parasitism in Peromyscus maniculatus, P. leucopus, and Tamias striatus. J Mammal 86:39–45. https://doi.org/10.1644/1545-1542(2005)086%3c0039:LPOBPI%3e2.0.CO;2
Kramer DL, Weary DM (1991) Exploration versus exploitation: a field study of time allocation to environmental tracking by foraging chipmunks. Anim Behav 41:443–449. https://doi.org/10.1016/S0003-3472(05)80846-2
Lafferty KD, Kuris AM (1999) How environmental stress affects the impacts of parasites. Limnol Oceanogr 44:925–931. https://doi.org/10.4319/lo.1999.44.3_part_2.0925
Landry-Cuerrier M, Munro D, Thomas DW, Humphries MM (2008) Climate and resource determinants of fundamental and realized metabolic niches of hibernating chipmunks. Ecology 89:3306–3316. https://doi.org/10.1890/08-0121.1
Lefcheck JS (2018) piecewiseSEM: piecewise structural equation modeling in R. Methods Ecol Evol 7:1–23. https://doi.org/10.1111/2041-210X.12512
Lindenfors P, Nunn CL, Jones KE et al (2007) Parasite species richness in carnivores: effects of host body mass, latitude, geographical range and population density. Glob Ecol Biogeogr 16:496–509. https://doi.org/10.1111/j.1466-8238.2006.00301.x
Llyod S (1995) Environmental influences on host immunity. In: Grenfell BT, Dobson AP (eds) Ecology of infectious diseases in natural populations. Cambridge University Press, Cambridge, pp 327–361
Lynsdale CL, Mumby HS, Hayward AD et al (2017) Parasite-associated mortality in a long-lived mammal: variation with host age, sex, and reproduction. Ecol Evol 7:10904–10915. https://doi.org/10.1002/ece3.3559
Mares MA, Willig MR, Bitar NA (1980) Home range size in eastern chipmunks, Tamias striatus, as a function of number of captures: statistical biases of inadequate sampling. J Mamm 61:661–669. https://doi.org/10.2307/1380311
Martin JGA, Réale D (2008) Temperament, risk assessment and habituation to novelty in eastern chipmunks, Tamias striatus. Anim Behav 75:309–318. https://doi.org/10.1016/j.anbehav.2007.05.026
McLean JA, Speakman JR (1997) Non-nutritional maternal support in the brown long-eared bat. Anim Behav 54:1193–1204. https://doi.org/10.1006/anbe.1997.0498
Messier GD, Garant D, Bergeron P, Réale D (2012) Environmental conditions affect spatial genetic structures and dispersal patterns in a solitary rodent. Mol Ecol 21:5363–5373. https://doi.org/10.1111/mec.12022
Moilliet TK (1949) Some preliminary observations on the life-history of Cuterebra tenebrosa Coquillet. Entomol Soc Br Columbia 46:1–3
Montiglio P-O, Garant D, Thomas D, Réale D (2010) Individual variation in temporal activity patterns in open-field tests. Anim Behav 80:905–912. https://doi.org/10.1016/j.anbehav.2010.08.014
Montiglio P-O, Garant D, Pelletier F, Réale D (2012) Personality differences are related to long-term stress reactivity in a population of wild eastern chipmunks, Tamias striatus. Anim Behav 84:1071–1079. https://doi.org/10.1016/j.anbehav.2012.08.010
Montiglio P-O, Garant D, Bergeron P et al (2014) Pulsed resources and the coupling between life-history strategies and exploration patterns in eastern chipmunks (Tamias striatus). J Anim Ecol 83:720–728. https://doi.org/10.1111/1365-2656.12174
Munro D, Thomas DW, Humphries MM (2008) Extreme suppression of aboveground activity by a food-storing hibernator, the eastern chipmunk (Tamias striatus). Can J Zool 86:364–370. https://doi.org/10.1139/Z08-008
Nunn CL, Altizer S, Jones KE, Sechrest W (2003) Comparative tests of parasite species richness in primates. Am Nat 162:597–614. https://doi.org/10.1086/378721
Penczykowski RM, Laine AL, Koskella B (2016) Understanding the ecology and evolution of host-parasite interactions across scales. Evol Appl 9:37–52. https://doi.org/10.1111/eva.12294
Price PW (1980) Evolutionary biology of parasites. Princeton University Press, Princeton
R Core Team (2019) R: a language and environment for statistical computing. Foundations for Statistical Computing, Vienna, Austria. https://www.r-project.org/. Accessed May 2018
Sabrosky CW (1986) North American Species of Cuterebra, the rabbit and rodent bot flies (Diptera: Cuterebridae). Entomological Society of America, Maryland
Schalk G, Forbes MR (1997) Male biases in parasitism of mammals: effects of study type, host age, and parasite taxon. Oikos 78:67–74. https://doi.org/10.2307/3545801
Sheldon BC, Verhulst S (1996) Ecological immunology: costly parasite defenses and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321. https://doi.org/10.1016/0169-5347(96)10039-2
St-Hilaire É, Réale D, Garant D (2017) Determinants, selection and heritability of docility in wild eastern chipmunks (Tamias striatus). Behav Ecol Sociobiol 71:88. https://doi.org/10.1007/s00265-017-2320-6
Villegas-Ríos D, Réale D, Freitas C et al (2018) Personalities influence spatial responses to environmental fluctuations in wild fish. J Anim Ecol 87:1309–1319. https://doi.org/10.1111/1365-2656.12872
Wilson K, Bjørnstad ON, Dobson AP et al (2001) Heterogeneities in macroparasite infections: patterns and processes. In: Hudson PJ, Rizzoli A, Grenfell BT et al (eds) The ecology of wildlife diseases. Oxford University Press, Oxford, pp 6–44
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70:164–168. https://doi.org/10.2307/1938423
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1024. https://doi.org/10.1016/S0020-7519(96)00086-0
Acknowledgements
We thank two anonymous reviewers for their helpful comments on an earlier version of this manuscript. We are grateful for all graduate students and field assistants who have contributed to the Chipmunk Project since 2005 and the Réserve Naturelle des Montagnes-Vertes (Québec, Canada) for allowing us to conduct this research within their boundaries.
Funding
This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery grants of DR, JS, DG, PB and by the Fonds de Recherche du Québec—Nature et technologies (FRQNT) team grant to DG, DR, and PB. CP was supported by FRQNT and NSERC scholarships.
Author information
Authors and Affiliations
Contributions
CP wrote the manuscript with support from DG, DR, PB and JS; CP performed analyses with the help of DG, PB and DR; DG, PB and JS supervised the project.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
All applicable institutional and/or national guidelines for the care and use of animals were followed. Animal handling protocols have been approved by the Ministère des Ressources Naturelles et de la Faune du Québec (no. 2018-04-20-103-05-S-F) and the Canadian Council on Animal Care (no. DG2011–01-Université de Sherbrooke).
Additional information
Communicated by Thomas Lilley.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paquette, C., Garant, D., Savage, J. et al. Individual and environmental determinants of Cuterebra bot fly parasitism in the eastern chipmunk (Tamias striatus). Oecologia 193, 359–370 (2020). https://doi.org/10.1007/s00442-020-04685-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04685-x