Abstract
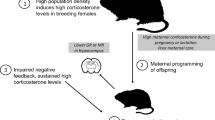
The literature reveals that stress in early life or adulthood can influence immune function. As most studies on this are from the laboratory, there is a need for replicated studies in wild animals. This study aims to examine the effects of density stress during the maternal period and adulthood on immune traits of root vole (Microtus oeconomus) individuals. Four replicated high- and low-density parental populations were established, from which we obtained offspring and assigned each into four enclosures, two for each of the two density treatments used in establishing parental populations. The F1 offspring fecal corticosterone metabolite response to acute immobilization stress, anti-keyhole limpet hemocyanin immunoglobulin G (anti-KLH IgG) level, phytohemagglutinin (PHA)-delayed hypersensitivity and hematology at the end of the first breeding season, and prevalence and intensity of coccidial infection throughout the two breeding seasons, were tested. Density-induced maternally stressed offspring had delayed responses to acute immobilization stress. Density-stressed offspring as adults had reduced anti-KLH IgG levels and PHA responses, and the effects further deteriorated in maternally stressed offspring, leading to higher coccidial infection in the first breeding season than in the second. No correlations were found between immune traits or coccidial infection and survival over winter. These findings indicated that the combined density stresses during the maternal period and adulthood exhibited negative synergistic effects on immune traits. The synergistic effects lead to higher coccidial infection; however, this consequently reduced the risk of subsequent infection. The increased coccidial infection mediated by the synergistic effects may have an adaptive value in the context of the environment.





Similar content being viewed by others
References
Bartolomucci A (2007) Social stress, immune functions and disease in rodents. Front Neuroendocrinol 28:28–49. doi:10.1016/j.yfrne.2007.02.001
Beldomenico PM, Telfer S, Gebert S, Lukomski L, Bennett M, Begon M (2008) The dynamics of health in wild field vole populations: a haematological perspective. J Anim Ecol 77:984–997. doi:10.1111/j.1365-2656.2008.01413.x
Bian JH, Wu Y, Getz LL, Cao YF, Chen F, Yang L (2011) Does maternal stress influence winter survival of offspring in root voles Microtus oeconomus? A field experiment. Oikos 120:47–56. doi:10.1111/j.1600-0706.2010.18165.x
Bian JH, Du SY, Wu Y, Cao YF, Nie XH, He H, You ZB (2015) Maternal effects and population regulation: maternal density-induced reproduction suppression impairs offspring capacity in response to immediate environment in root voles Microtus oeconomus. J Anim Ecol 84:326–336. doi:10.1111/1365-2656.12307
Boonstra R, Boag PT (1992) Spring declines in Microtus pennsylvanicus and the role of steroid hormones. J Anim Ecol 61:339–352. doi:10.2307/5326
Chen F, Du S, Bian J, You ZB, Wu Y (2012) Chronic hypoxia exposure during pregnancy is associated with a decreased active nursing activity in mother and an abnormal birth weight and postnatal growth in offspring of rats. Horm Behav 61:504–511. doi:10.1016/j.yhbeh.2012.01.009
Chung S, Son GH, Park SH, Park E, Lee KH, Geum D, Kim K (2005) Differential adaptive responses to chronic stress of maternally stressed male mice offspring. Endocrinology 146:3202–3210. doi:10.1210/en.2004-1458
Creel S, Dantzer B, Goymann W, Rubenstein DR (2013) The ecology of stress: effects of the social environment. Funct Ecol 27:66–80. doi:10.1111/j.1365-2435.2012.02029.x
Dantzer B, Newman AE, Boonstra R, Palme R, Boutin S, Humphries MM, McAdam AG (2013) Density triggers maternal hormones that increase adaptive offspring growth in a wild mammal. Science 340:1215–1217. doi:10.1126/science.1235765
Demas GE, Zysling DA, Beechler BR, Muehlenbein MP, French SS (2011) Beyond phytohaemagglutinin: assessing vertebrate immune function across ecological contexts. J Anim Ecol 80:710–730. doi:10.1111/j.1365-2656.2011.01813.x
Eberhardt AT, Costa SA, Marini MR, Racca A, Baldi CJ, Robles MR, Moreno PG, Beldomenico PM (2013) Parasitism and physiological trade-offs in stressed capybaras. PLoS One 8:e70382. doi:10.1371/journal.pone.0070382
French SS, Moore MC, Demas GE (2009) Ecological immunology: the organism in context. Integr Comp Biol 49:246–253. doi:10.1093/icb/icp032
Fuhrman MP, Charney P, Mueller CM (2004) Hepatic proteins and nutrition assessment. J Am Diet Assoc 104:1258–1264. doi:10.1016/j.jada.2004.05.213
Fuller CA, Blaustein AR (1996) Effects of the parasite Eimeria arizonensis on survival of deer mice (Peromyscus maniculatus). Ecology 77:2196–2202. doi:10.2307/2265712
Götz AA, Stefanski V (2007) Psychosocial maternal stress during pregnancy affects serum corticosterone, blood immune parameters and anxiety behaviour in adult male rat offspring. Physiol Behav 90:108–115. doi:10.1016/j.physbeh.2006.09.014
Goüy de Bellocq J, Krasnov BR, Khokhlova IS, Pinshow B (2006) Temporal dynamics of a T-cell mediated immune response in desert rodents. Comp Biochem Physiol A: Mol Integr Physiol 145:554–559. doi:10.1016/j.cbpa.2006.08.045
Hakkarainen H, Huhta E, Koskela E, Mappes T, Soveri T, Suorsa P (2007) Eimeria-parasites are associated with a lowered mother’s and offspring’s body condition in island and mainland populations of the bank vole. Parasitology 134:23–31. doi:10.1017/S0031182006001120
Harper JM, Austad SN (2004) Fecal corticosteroid levels in free-living populations of deer mice (Peromyscus maniculatus) and southern red-backed voles (Clethrionomys gapperi). Am Midl Nat 152:400–409. doi:10.1674/0003-0031(2004)152[0400:FCLIFP]2.0.CO;2
He H, Cao Y, Chen L, Du S, Nie X, Bian J (2013) The utility of detecting corticosterone levels in feces of root vole (Microtus oeconomus). Acta Theriol Sin 33:164–171 (in Chinese, English abstract)
Helle H, Koskela E, Mappes T (2012) Life in varying environments: experimental evidence for delayed effects of juvenile environment on adult life history. J Anim Ecol 81:573–582. doi:10.1111/j.1365-2656.2011.01937.x
Hudson PJ (1986) The effect of a parasitic nemade on the breeding production of red grouse. J Anim Ecol 55:85–92. doi:10.2307/4694
Jiang Y, Wei S, Wang Z, Zhen Y, Cui R (1991) Productivity investigation of the root vole (Microtus oeconomus) population in the Haibei alpine bushland (Potentilia fruticosa). I. Population dynamics. Acta Theirol Sin 11:270–278 (in Chinese, English abstract)
Kavaliers M, Colwell DD (1995) Decreased predator avoidance in parasitized mice: neuromodulatory correlates. Parasitology 111:257–263. doi:10.1017/S0031182000081816
Kay G, Tarcic N, Poltyrev T, Weinstock M (1998) Prenatal stress depresses immune function in rats. Physiol Behav 63:397–402. doi:10.1016/S0031-9384(97)00456-3
Klein SL, Rager DR (1995) Prenatal stress alters immune function in the offspring of rats. Dev Psychobiol 28:321–336. doi:10.1002/dev.420280603
Krebs CJ, Boonstra R, Boutin S, Sinclair ARE (2001) What drives the 10-year cycle of snowshoe hares? Bioscience 51:25–35. doi:10.1641/0006-3568(2001)051[0025:WDTYCO]2
Li Y, Zhao X, Cao G, Zhao L, Wang Q (2004) The climate of Haibei alpine meadow ecosystem research station, the analysis of background of vegetation productivity. Plat Meteorol 23:558–567 (in Chinese, English abstract)
Liang J, Zeng J, Wang Z, Han Y (1982) Studies on growth and development in the root vole (Microtus oeconomus). Acta Biol Plat Sin 1:195–208 (in Chinese, English abstract)
Llorente E, Brito ML, Machado P, Gonzalez MC (2002) Effect of prenatal stress on the hormonal response to acute and chronic stress and on immune parameters in the offspring. J Physiol Biochem 58:143–149. doi:10.1007/BF03179851
Lochmiller RL (1996) Immunocompetence and animal population regulation. Oikos 76:594–601. doi:10.2307/3546356
Lochmiller RL, Dabbert BC (1993) Immunocompetence, environmental stress and the regulation of animal populations. Trends Comp Biochem Physiol 1:823–855
Lochmiller RL, Deerenberg C (2000) Trade-offs in evolutionary immunology: just what is the cost of immunity? Oikos 88:87–98. doi:10.1034/j.1600-0706.2000.880110.x
Merlot E, Couret D, Otten W (2008) Prenatal stress, fetal imprinting and immunity. Brain Behav Immun 22:42–51. doi:10.1016/j.bbi.2007.05.007
Mills SC, Grapputo A, Jokinen I, Koskela E, Mappes T, Poikonen T (2010) Fitness trade-offs mediated by immunosuppression costs in a small mammal. Evolution 64:166–179. doi:10.1111/j.1558-5646.2009.00820.x
Møller AP, Erritzøe J, Saino N (2003) Seasonal changes in immune response and parasite impact on hosts. Am Nat 161:657–671. doi:10.1086/367879
Moshkin MP, Dobrotvorsky AK, Mak VV, Panov VV, Dobrotvorskaya EA (1998) Variability of immune response to heterologous erythrocytes during population cycles of red (Clethrionomys rutilus) and bank (Clethrionomys glareolus) voles. Oikos 82:131–138. doi:10.2307/3546923
Moynihan JA (2003) Mechanisms of stress-induced modulation of immunity. Brain Behav Immun 17:11–16. doi:10.1016/S0889-1591(02)00060-0
Nelson RJ, Demas GE, Klein SL, Kriegsfeld LJ (1995) Minireview: the influence of season, photoperiod, and pineal melatonin on immune function. J Pineal Res 19:149–165. doi:10.1111/j.1600-079X.1995.tb00184.x
Nelson RJ, Fine JB, Demas GE, Moffatt CA (1996) Photoperiod and population density interact to affect reproductive and immune function in male prairie voles. Am J Physiol Regul Integr Comp Physiol 270:R571–R577
Nie X, Cao Y, Du S, He H, Bian J (2014) Intestinal parasite infection rate of root vole (Microtus oeconomus): a field experiment. Acta Theriol Sin 34:172–180 (in Chinese, English abstract)
Padgett DA, Glaser R (2003) How stress influences the immune response. Trends Immunol 24:444–448. doi:10.1016/S1471-4906(03)00173-X
Palme R, Rettenbacher S, Touma C, El-Bahr SM, Möstle E (2005) Stress hormones in mammals and birds: comparative aspects regarding metabolism, excretion, and noninvasive measurement in fecal samples. Ann NY Acad Sci 1040:162–171. doi:10.1196/annals.1327.021
Pedersen AB, Greives TJ (2008) The interaction of parasites and resources cause crashes in a wild mouse population. J Anim Ecol 77:370–377. doi:10.1111/j.1365-2656.2007.01321.x
Pihl L, Hau J (2003) Fecal corticosterone and immunoglobulin A in young adult rats. Lab Anim 37:166–171. doi:10.1258/00236770360563822
Rifkin JL, Nunn CL, Garamszegi LZ (2012) Do animals living in larger groups experience greater parasitism? A meta-analysis. Am Nat 180:70–82. doi:10.1086/666081
Saino N, Canova L, Fasola M, Martinelli R (2000) Reproduction and population density affect humoral immunity in bank voles under field experimental conditions. Oecologia 124:358–366. doi:10.1007/s004420000395
Sheriff MJ (2015) The adaptive potential of maternal stress exposure in regulating population dynamics. J Anim Ecol 84:323–325. doi:10.1111/1365-2656.12334
Sheriff M, Krebs CJ, Boonstra R (2010) Assessing stress in animal populations: do fecal and plasma glucocorticoids tell the same story? Gen Comp Endocrinol 166:614–619. doi:10.1016/j.ygcen.2009.12.017
Sinclair JA, Lochmiller RL (2000) The winter immunoenhancement hypothesis: associations among immunity, density, and survival in prairie vole (Microtus ochrogaster) populations. Can J Zool 78:254–264. doi:10.1139/z99-203
Sun P, Zhao X, Xu S, Zhao T, Zhao W (2002) Changes after snow of the population characteristic of root vole (Microtus oeconomus) in Haibei alpine meadow. Acta Theriol Sin 22:318–320 (in Chinese, English abstract)
Temple SA (1987) Do predator always capture substandard individuals disproportionately from prey populations? Ecology 68:669–674. doi:10.2307/1938472
Tibary A, Fite C, Anouassi A, Sghiri A (2006) Infectious causes of reproductive loss in camelids. Theriogenology 66:633–647. doi:10.1016/j.theriogenology.2006.04.008
Tompkins DM, Dunn AM, Smith MJ, Telfer S (2011) Wildlife diseases: from individuals to ecosystems. J Anim Ecol 80:19–38. doi:10.1111/j.1365-2656.2010.01742.x
Tuchscherer M, Kanitz E, Otten W, Tuchscherer A (2002) Effects of prenatal stress on cellular and humoral immune responses in neonatal pigs. Vet Immunol Immunopathol 86:195–203. doi:10.1016/S0165-2427(02)00035-1
Wada H, Noguchi Y, Marino MW, Dunn AR, Old LJ (1997) T-cell functions in granulocyte/macrophage colony-stimulating factor deficient mice. Proc Natl Acad Sci 94:12557–12561
Wetzel R (1951) Verbesserte Mcmaster-Kammer zum Auszählen von Wurmeiern. Tierärztl Umsch 6:209–210
Xu DL, Wang DH (2010) Fasting suppresses T cell-mediated immunity in female Mongolian gerbils (Meriones unguiculatus). Comp Biochem Physiol A: Mol Integr Physiol 155:25–33. doi:10.1016/j.cbpa.2009.09.003
Xu D, Liu X, Wang D (2011) Impairment of cellular and humoral immunity in overweight Mongolian gerbils (Meriones unguiculatus). Integr Zool 6:352–365. doi:10.1111/j.1749-4877.2011.00259.x
Zysling DA, Demas GE (2007) Metabolic stress suppresses humoral immune function in long-day, but not short-day, Siberian hamsters (Phodopus sungorus). J Comp Physiol B 177:339–347. doi:10.1007/s00360-006-0133-4
Acknowledgments
We thank Hui He for her assistance in the fecal sample collection and fieldwork. This work was funded by the National Natural Science Foundation of China (grant no. 31170394) and the Natural Science Foundation of Qinghai Province (grant no. 2014-ZJ-913).
Author contribution statement
J. H. B. and Y. W. conceived and designed the experiments. S. Y. D., Y. F. C., and X. H. N. performed the experiments. J. H. B. and S. Y. D. analyzed the data. S. Y. D. wrote the manuscript. J. H. B. provided editorial advice.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by Michael Sheriff.
Immunity can influence individual fitness and regulate population dynamics in small mammals. As most studies are from the laboratory, there is a need for replicated studies in wild animals. Thus, exploring how immune function varies according to environmental variables that individuals experience may help to identify plasticity of life history traits and mechanisms driving population fluctuations of wild small mammals.
Rights and permissions
About this article
Cite this article
Du, SY., Cao, YF., Nie, XH. et al. The synergistic effect of density stress during the maternal period and adulthood on immune traits of root vole (Microtus oeconomus) individuals—a field experiment. Oecologia 181, 335–346 (2016). https://doi.org/10.1007/s00442-015-3445-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3445-9