Abstract
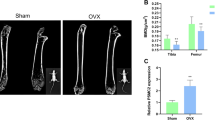
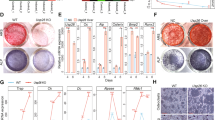
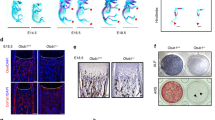
Estrogens play multiple roles in maintaining skeletal homeostasis by regulating many physiological processes in bone cells. Recently, cellular senescence in bone cells, especially in osteocytes, has been demonstrated to be a pivotal factor in bone loss. However, whether and how estrogen mediates cellular senescence in bone cells remains unknown. Here, we show that estrogen is negatively correlated with p53-related cellular senescence, primarily through the regulation of p53 protein levels, both in vivo and in vitro. Further study confirmed that estrogen attenuated the nuclear import of p53 and accelerated p53 degradation in osteocyte-like MLO-Y4 cells and osteoblastic MC3T3-E1 cells. A screen of p53-related ubiquitinating/deubiquitinating enzymes indicated that estrogen induced the degradation of p53 through the regulation of Usp10, a deubiquitinase that is directly linked to p53. Usp10 inhibition attenuated H2O2-induced senescence in MLO-Y4 cells, as indicated by p53/p21 quantification, a senescence-associated β-galactosidase (SA-β-gal) assay, and p53 localization visualization with a confocal microscope. Usp10 overexpression abolished the estrogen-mediated regulation of p53 and the downstream transcriptional gene p21. The injection of ovariectomized (OVX) mice with Spautin-1, a Usp10 inhibitor, inhibited the expression of p53 and the transcription of downstream senescence markers, as well as promoted bone mass recovery. Taken together, our study unveils the regulatory function of estrogen in the prevention of cellular senescence through the regulation of Usp10, thereby accelerating the degradation of senescent factor p53 and inhibiting its nuclear import.




Similar content being viewed by others
References
Arai YWS, Kimira M, Shimoi K, Mochizuki R, Kinae N (2000) Dietary intakes of flavonols, flavones and isoflavones by Japanese women and the inverse correlation between quercetin intake and plasma LDL cholesterol concentration. J Nutr 130(9):2243–2250
Chen JR, Lazarenko OP, Blackburn ML, Mercer KE, Badger TM, Ronis MJ (2015) p47phox-Nox2-dependent ROS signaling inhibits early bone development in mice but protects against skeletal aging. J Biol Chem 290(23):14692–14704. https://doi.org/10.1074/jbc.M114.633461
Chen JR, Lazarenko OP, Haley RL, Blackburn ML, Badger TM, Ronis MJ (2009) Ethanol impairs estrogen receptor signaling resulting in accelerated activation of senescence pathways, whereas estradiol attenuates the effects of ethanol in osteoblasts. J Bone Miner Res 24(2):221–230. https://doi.org/10.1359/jbmr.081011
Chen JR, Lazarenko OP, Zhao H, Wankhade UD, Pedersen K, Watt J, Ronis MJJ (2020) Nox4 expression is not required for OVX-induced osteoblast senescence and bone loss in mice. JBMR plus 4(8):e10376. https://doi.org/10.1002/jbm4.10376
Dallas SL, Prideaux M, Bonewald LF (2013) The osteocyte: an endocrine cell … and more. Endocr Rev 34(5):658–690. https://doi.org/10.1210/er.2012-1026
Farr JN, Fraser DG, Wang H, Jaehn K, Ogrodnik MB, Weivoda MM, Khosla S (2016) Identification of senescent cells in the bone microenvironment. J Bone Miner Res 31(11):1920–1929. https://doi.org/10.1002/jbmr.2892
Farr JN, Khosla S (2019) Cellular senescence in bone. Bone 121:121–133. https://doi.org/10.1016/j.bone.2019.01.015
Farr JN, Xu M, Weivoda MM, Monroe DG, Fraser DG, Onken JL, Khosla S (2017) Targeting cellular senescence prevents age-related bone loss in mice. Nat Med 23(9):1072–1079. https://doi.org/10.1038/nm.4385
Fu J, Hao L, Tian Y, Liu Y, Gu Y, Wu J (2017) miR-199a-3p is involved in estrogen-mediated autophagy through the IGF-1/mTOR pathway in osteocyte-like MLO-Y4 cells. J Cell Physiol. https://doi.org/10.1002/jcp.26101
Fu J, Hao L, Tian Y, Liu Y, Gu Y, Wu J (2018) miR-199a-3p is involved in estrogen-mediated autophagy through the IGF-1/mTOR pathway in osteocyte-like MLO-Y4 cells. J Cell Physiol 233(3):2292–2303. https://doi.org/10.1002/jcp.26101
Glyn N, James W, Chunfang W, Diana J, Conor L, Carmen M-R, Thomas, v. Z. (2012) A senescent cell bystander effect: senescence-induced senescence. Aging Cell 11:345–349
Gorgoulis VG, Halazonetis TD (2010) Oncogene-induced senescence: the bright and dark side of the response. Curr Opin Cell Biol 22(6):816–827. https://doi.org/10.1016/j.ceb.2010.07.013
Hao L, Fu J, Tian Y, Wu J (2017) Systematic analysis of lncRNAs, miRNAs and mRNAs for the identification of biomarkers for osteoporosis in the mandible of ovariectomized mice. Int J Mol Med 40(3):689–702. https://doi.org/10.3892/ijmm.2017.3062
Heldring N, Pike A, Andersson S, Matthews J, Cheng G, Hartman J, Gustafsson JA (2007) Estrogen receptors: how do they signal and what are their targets. Physiol Rev 87(3):905–931. https://doi.org/10.1152/physrev.00026.2006
Jian Yuan KL, Zhang L, Cheville J, Lou Z (2010) USP10 Regulates p53 Localization and Stability by Deubiquitinating p53. Cell. https://doi.org/10.1016/j.cell.2009.12.032
Kim HN, Chang J, Iyer S, Han L, Campisi J, Manolagas SC, Almeida M (2019) Elimination of senescent osteoclast progenitors has no effect on the age-associated loss of bone mass in mice. Aging Cell e12923. https://doi.org/10.1111/acel.12923
Kirkland JL, Tchkonia T (2017) Cellular Senescence: a Translational Perspective Ebiomedicine 21:21–28. https://doi.org/10.1016/j.ebiom.2017.04.013
Ko A, Han SY, Choi CH, Cho H, Lee M-S, Kim S-Y, Song J (2018) Oncogene-induced senescence mediated by c-Myc requires USP10 dependent deubiquitination and stabilization of p14ARF. Cell Death Differ 25(6):1050–1062. https://doi.org/10.1038/s41418-018-0072-0
Kwon SK, Saindane M, Baek KH (2017) p53 stability is regulated by diverse deubiquitinating enzymes. Biochim Biophys Acta 1868(2):404–411. https://doi.org/10.1016/j.bbcan.2017.08.001
LeAnn MTL, Yixia X, Molly AH, Richard C, Mark RD, Vladimir D, Sarah LD (2017) Degeneration of the osteocyte network in the C57BL/6 mouse model of aging. Aging 9(10):2190–2208
Lee JT, Gu W (2010) The multiple levels of regulation by p53 ubiquitination. Cell Death Differ 17(1):86–92. https://doi.org/10.1038/cdd.2009.77
Liu J, Xia H, Kim M, Xu L, Li Y, Zhang L, Yuan J (2011) Beclin1 controls the levels of p53 by regulating the deubiquitination activity of USP10 and USP13. Cell 147(1):223–234. https://doi.org/10.1016/j.cell.2011.08.037
Moustapha Kassem PJM (2011) Senescence-associated intrinsic mechanisms of osteoblast dysfunctions. Aging Cell 191–197
Munoz-Espin D, Serrano M (2014) Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 15(7):482–496. https://doi.org/10.1038/nrm3823
Nandini K, Angelika B, Jun Yeob K, Gu X, Chong G, Jill B (2017) Estrogen-activated MDM2 disrupts mammary tissue architecture through a p53-independent pathway. Oncotarget 8(29):47916–47930
Panieri E, Gogvadze V, Norberg E, Venkatesh R, Orrenius S, Zhivotovsky B (2013) Reactive oxygen species generated in different compartments induce cell death, survival, or senescence. Free Radic Biol Med 57:176–187. https://doi.org/10.1016/j.freeradbiomed.2012.12.024
Patsch JM, Deutschmann J, Pietschmann P (2011) Gender aspects of osteoporosis and bone strength. Wien Med Wochenschr 161(5–6):117–123. https://doi.org/10.1007/s10354-011-0891-9
Patullo IM, Takayama L, Patullo RF, Jorgetti V, Pereira RM (2009) Influence of ovariectomy and masticatory hypofunction on mandibular bone remodeling. Oral Dis 15(8):580–586. https://doi.org/10.1111/j.1601-0825.2009.01599.x
Ren J, Wang XH, Wang GC, Wu JH (2013) 17beta estradiol regulation of connexin 43-based gap junction and mechanosensitivity through classical estrogen receptor pathway in osteocyte-like MLO-Y4 cells. Bone 53(2):587–596. https://doi.org/10.1016/j.bone.2012.12.004
Riley T, Sontag E, Chen P, Levine A (2008) Transcriptional control of human p53-regulated genes. Nat Rev Mol Cell Biol 9(5):402–412. https://doi.org/10.1038/nrm2395
Rosser J, Bonewald LF (2012) Studying osteocyte function using the cell lines MLO-Y4 and MLO-A5. Methods Mol Biol 816:67–81. https://doi.org/10.1007/978-1-61779-415-5_6
Salama R, Sadaie M, Hoare M, Narita M (2014) Cellular senescence and its effector programs. Genes Dev 28(2):99–114. https://doi.org/10.1101/gad.235184.113
Wei Y, Fu J, Wu W, Wu J (2020) Comparative profiles of DNA methylation and differential gene expression in osteocytic areas from aged and young mice. Cell Biochem Funct 38(6):721–732. https://doi.org/10.1002/cbf.3539
Wu W, Fu J, Gu Y, Wei Y, Ma P, Wu J (2020) JAK2/STAT3 regulates estrogen-related senescence of bone marrow stem cells. J Endocrinol 245(1):141–153. https://doi.org/10.1530/JOE-19-0518
Yang Y, Zheng X, Li B, Jiang S, Jiang L (2014) Increased activity of osteocyte autophagy in ovariectomized rats and its correlation with oxidative stress status and bone loss. Biochem Biophys Res Commun 451(1):86–92. https://doi.org/10.1016/j.bbrc.2014.07.069
Yang YH, Chen K, Li B, Chen JW, Zheng XF, Wang YR, Jiang LS (2013) Estradiol inhibits osteoblast apoptosis via promotion of autophagy through the ER-ERK-mTOR pathway. Apoptosis 18(11):1363–1375. https://doi.org/10.1007/s10495-013-0867-x
Funding
This work was supported by the National Natural Science Foundation of China (grant no. 81470716), the Natural Science Foundation of Shanghai (grant no.19ZR462000), and the Science and Technology Committee Foundation of Shanghai (grant no. 14411967200).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Institute of Animal Care and Use Committee of Tongji University (no. TJLAC-018–035). During the experiment and routine feeding, animals received humane care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (NIH).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wei, Y., Fu, J., Wu, W. et al. Estrogen prevents cellular senescence and bone loss through Usp10-dependent p53 degradation in osteocytes and osteoblasts: the role of estrogen in bone cell senescence. Cell Tissue Res 386, 297–308 (2021). https://doi.org/10.1007/s00441-021-03496-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-021-03496-7