Abstract
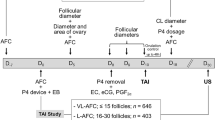
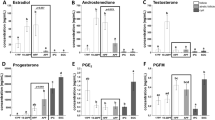
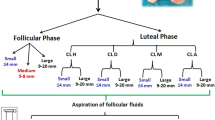
In cattle, the oviduct plays a major role in the reproductive process; however, molecular control of oviduct receptivity to the embryo is poorly understood. A model for receptivity based on size of the pre-ovulatory follicle (POF) was used to compare oviductal morphology, cellular proliferation, and candidate transcript abundance. Growth of the POF of Nelore (Bos indicus) cows was manipulated to produce two groups: a large POF-large corpus luteum (CL) group (LF-LCL; greater receptivity) and a small POF-small CL group (SF-SCL). Samples of the ampulla and isthmus ipsilateral and contralateral to CL were collected 4 days after GnRH-induced ovulation. Tissues were either embedded in paraffin for Harris-Hematoxylin and Eosin and periodic acid-Schiff staining and KI67 immunostaining, followed by morphological analyses, or stored at −80 °C for RNA extraction, cDNA synthesis, and qPCR analyses. The effects of group (LF-LCL and SF-SCL), region (ampulla and isthmus), and side (ipsilateral and contralateral) were analyzed using three-way nested ANOVA. The ipsilateral ampulla of the LF-LCL group presented more primary mucosal folds, a greater mucosal-folding grade and luminal perimeter, and more secretory cells and proliferating cells when compared with the ampulla of the SF-SCL group and with the contralateral ampulla of both groups. There were no morphological differences in the isthmus between groups and sides. Changes in transcript abundance are suggestive of LF-LCL-stimulated secretory activity. In summary, ovulation of a larger POF generates a periovulatory endocrine milieu that modulates morphological and functional features of the bovine oviduct which may support embryo survival and development.








Similar content being viewed by others
References
Abe H (1996) The mammalian oviductal epithelium: regional variations in cytological and functional aspects of the oviductal secretory cells. Histol Histopathol 11:743–768
Agduhr E (1927) Studies on the structure and development of the bursa ovarica and the tuba uterina in the mouse. Acta Zool 8:1–133
Aguilar J, Reyley M (2005) The uterine tubal fluid: secretion, composition and biological effects. Anim Reprod 2:91–105
Ahumada CJ, Salvador I, Cebrian-Serrano A, Lopera R, Silvestre MA (2013) Effect of supplementation of different growth factors in embryo culture medium with a small number of bovine embryos on in vitro embryo development and quality. Animal 7:455–462
Ashworth CJ, Sales DI, Wilmut I (1989) Evidence of an association between the survival of embryos and the periovulatory plasma progesterone concentration in the ewe. J Reprod Fertil 87:23–32
Aviles M, Gutierrez-Adan A, Coy P (2010) Oviductal secretions: will they be key factors for the future ARTs? Mol Hum Reprod 16:896–906
Ayen E, Shahrooz R, Kazemie S (2012) Histological and histomorphometrical changes of different regions of oviduct during follicular and lutealphases of estrus cycle in adult Azarbaijan buffalo. Iran J Vet Res 13:42–48
Bacha WJ, Bacha LM (2012) Color atlas of veterinary histology, 3rd edn. Wiley-Blackwell, New York
Besenfelder U, Havlicek V, Brem G (2012) Role of the oviduct in early embryo development. Reprod Domest Anim 47:156–163
Brower L, Anderson E (1969) Cytological events associated with the secretory process in the rabbit oviduct. Biol Reprod 1:130–148
Buhi WC, Bazer FW, Alvarez IM, Mirando MA (1991) In vitro synthesis of oviductal proteins associated with estrus and 17 beta-estradiol-treated ovariectomized ewes. Endocrinology 128:3086–3095
Buhi WC, Ashworth CJ, Bazer FW, Alvarez IM (1992) In vitro synthesis of oviductal secretory proteins by estrogen-treated ovariectomized gilts. J Exp Zool 262:426–435
Buhi WC, Alvarez IM, Kouba AJ (2000) Secreted proteins of the oviduct. Cells Tissues Organs 166:165–179
Chanrota M, Guoa Y, Dalina AM, Perssonb E, Bågea R, Svenssona A, Gustafssona H, Humblota P (2017) Dose related effects of LPS on endometrial epithelial cell populations from dioestrus cows. Anim Reprod Sci 177:12–24
Demetrio DG, Santos RM, Demetrio CG, Vasconcelos JL (2007) Factors affecting conception rates following artificial insemination or embryo transfer in lactating Holstein cows. J Dairy Sci 90:5073–5082
Donnez J, Casanas-Roux F, Caprasse J, Ferin J, Thomas K (1985) Cyclic changes in ciliation, cell height, and mitotic activity in human tubal epithelium during reproductive life. Fertil Steril 43:554–559
Eriksen T, Terkelsen O, Hyttel P, Greve T (1994) Ultrastructural features of secretory-cells in the bovine oviduct epithelium. Anat Embryol 190:583–590
Gandolfi F, Brevini TAL, Richardson L, Brown CR, Moor RM (1989) Characterization of proteins secreted by sheep oviduct epithelial-cells and their function in embryonic-development. Development 106:303–312
García EV, Hamdi M, Barrera AD, Sánchez-Calabuig MJ, Gutiérrez-Adán A, Rizos D (2017) Bovine embryo-oviduct interaction in vitro reveals an early cross talk mediated by BMP signaling. Reproduction 153:631–643
Gonella-Diaza A, Andrade S, Sponchiado M, Pugliesi G, Mesquita F, Van Hoeck V, Strefezzi R, Gasparin G, Coutinho L, Binelli M (2015) Size of the ovulatory follicle dictates spatial differences in the oviductal transcriptome in cattle. PLoS ONE 10:e0145321
Gray CA, Bartol FF, Tarleton BJ, Wiley AA, Johnson GA, Bazer FW, Spencer TE (2001) Developmental biology of uterine glands. Biol Reprod 65:1311–1323
Hunter RH (1998) Have the fallopian tubes a vital role in promoting fertility? Acta Obstet Gynecol Scand 77:475–486
Hunter RH (2012) Components of oviduct physiology in eutherian mammals. Biol Rev Camb Philos Soc 87:244–255
Hunter RH, Cook B, Poyser NL (1983) Regulation of oviduct function in pigs by local transfer of ovarian steroids and prostaglandins: a mechanism to influence sperm transport. Eur J Obstet Gynecol Reprod Biol 14:225–232
Ito S, Kobayashi Y, Yamamoto Y, Kimura K, Okuda K (2016) Remodeling of bovine oviductal epithelium by mitosis of secretory cells. Cell Tissue Res 366:403–410
Jordaens L, Van Hoeck V, Maillo V, Gutierrez-Adan A, Marei WFA, Vlaeminck B, Thys S, Sturmey RG, Bols PEJ, Leroy JLMR (2017) Maternal metabolic stress may affect oviduct gatekeeper function. Reproduction 153:759–773
Kenngott RA, Sinowatz F (2007) Prenatal development of the bovine oviduct. Anat Histol Embryol 36:272–283
Kolle S, Dubielzig S, Reese S, Wehrend A, Konig P, Kummer W (2009) Ciliary transport, gamete interaction, and effects of the early embryo in the oviduct: ex vivo analyses using a new digital videomicroscopic system in the cow. Biol Reprod 81:267–274
Konishi I, Fujii S, Parmley TH, Mori T (1987) Development of ciliated cells in the human fetal oviduct: an ultrastructural study. Anat Rec 219:60–68
Leese HJ (1988) The formation and function of oviduct fluid. J Reprod Fertil 82:843–856
McDaniel JW, Scalzi H, Black D (1968) Influence of ovarian hormones on histology and histochemistry of the bovine oviduct. J Dairy Sci 51:754–761
Mesquita FS, Pugliesi G, Scolari SC, Franca MR, Ramos RS, Oliveira M, Papa PC, Bressan FF, Meirelles FV, Silva LA, Nogueira GP, Membrive CM, Binelli M (2014) Manipulation of the periovulatory sex steroidal milieu affects endometrial but not luteal gene expression in early diestrus Nelore cows. Theriogenology 81:861–869
Mesquita FS, Ramos RS, Pugliesi G, Andrade SC, Van Hoeck V, Langbeen A, Oliveira ML, Gonella-Diaza AM, Gasparin G, Fukumasu H, Pulz LH, Membrive CM, Coutinho LL, Binelli M (2015) The receptive endometrial transcriptomic signature indicates an earlier shift from proliferation to metabolism at early Diestrus in the cow. Biol Reprod 93:52
Mokhtar DM (2015) Microscopic and histochemical characterization of the bovine uterine tube during the follicular and luteal phases of estrous cycle. J Microsc Ultrastruct 3:44–52
Morris D, Diskin M (2008) Effect of progesterone on embryo survival. Animal 2:1112–1119
Peres RF, Claro I Jr, Sa Filho OG, Nogueira GP, Vasconcelos JL (2009) Strategies to improve fertility in Bos indicus postpubertal heifers and non-lactating cows submitted to fixed-time artificial insemination. Theriogenology 72:681–689
Pugliesi G, Santos FB, Lopes E, Nogueira É, Maio JR, Binelli M (2016) Improved fertility in suckled beef cows ovulating large follicles or supplemented with long-acting progesterone after timed-AI. Theriogenology 85:1239–1248
Restall BJ (1966) Histological observations on the reproductive tract of the ewe. Aust J Biol Sci 19:673–686
Rizos D, Fair T, Papadopoulos S, Boland MP, Lonergan P (2002a) Developmental, qualitative, and ultrastructural differences between ovine and bovine embryos produced in vivo or in vitro. Mol Reprod Dev 62:320–327
Rizos D, Lonergan P, Boland MP, Arroyo-Garcia R, Pintado B, de la Fuente J, Gutierrez-Adan A (2002b) Analysis of differential messenger RNA expression between bovine blastocysts produced in different culture systems: implications for blastocyst quality. Biol Reprod 66:589–595
Rizos D, Ward F, Duffy P, Boland MP, Lonergan P (2002c) Consequences of bovine oocyte maturation, fertilization or early embryo development in vitro versus in vivo: implications for blastocyst yield and blastocyst quality. Mol Reprod Dev 61:234–248
Simintiras CA, Fröhlich T, Sathyapalan T, Arnold GJ, Ulbrich SE, Leese HJ, Sturmey RG (2016) Modelling oviduct fluid formation in vitro. Reproduction 153:23–33
Smit A, Hubley R, Green P (1996-2010) RepeatMasker Open-3.0. http://www.repeatmasker.org
Vasconcelos JLM, Sartori R, Oliveira HN, Guenther JG, Wiltbank MC (2001) Reduction in size of the ovulatory follicle reduces subsequent luteal size and pregnancy rate. Theriogenology 56:307–314
Wang CK, Robinson RS, Flint APF, Mann GE (2007) Quantitative analysis of changes in endometrial gland morphology during the bovine oestrous cycle and their association with progesterone levels. Reproduction 134:365–371
Wetscher F, Havlicek V, Huber T, Muller M, Brem G, Besenfelder U (2005) Effect of morphological properties of transferred embryonic stages on tubal migration implications for in vivo culture in the bovine oviduct. Theriogenology 64:41–48
Wijayagunawardane MPB, Cerbito WA, Miyamoto A, Acosta TJ, Takagi M, Miyazawa K, Sato K (1996) Oviductal progesterone concentration and its spatial distribution in cyclic and early pregnant cows. Theriogenology 46:1149–1158
Wijayagunawardane MPB, Miyamoto A, Cerbito WA, Acosta TJ, Takagi M, Sato K (1998) Local distributions of oviductal estradiol, progesterone, prostaglandins, oxytocin and endothelin-1 in the cyclic cow. Theriogenology 49:607–618
Yoshioka S, Abe H, Sakumoto OK (2013) Proliferation of luteal steroidogenic cells in cattle. PLoS ONE 8:e84186
Acknowledgements
The authors thank M. R. França, R. S. Ramos, E. Lopes, E. R. Araujo, M. L. Oliveira, and C. M. Membrive for technical assistance; and the administration of the Pirassununga campus of the University of São Paulo for assistance in animal handling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding
This research was funded by: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) PEC-PG program: AMGD grant number 15068–12-9; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq); Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP): MB grant number 2011/03226–4. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Rights and permissions
About this article
Cite this article
Gonella-Diaza, A.M., Mesquita, F.S., da Silva, K.R. et al. Sex steroids modulate morphological and functional features of the bovine oviduct. Cell Tissue Res 370, 319–333 (2017). https://doi.org/10.1007/s00441-017-2666-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-017-2666-0