Abstract
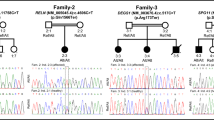
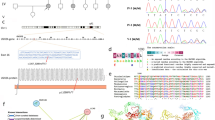
Pelizaeus-Merzbacher-like disease (PMLD) is an autosomal recessive hypomyelinating leukodystrophy, which is clinically and radiologically similar to X-linked Pelizaeus-Merzbacher disease (PMD). PMLD is characterized by early-onset nystagmus, delayed development (motor delay, speech delay and dysarthria), dystonia, hypotonia typically evolving into spasticity, ataxia, seizures, optic atrophy, and diffuse leukodystrophy on magnetic resonance imaging (MRI). We identified a 12-year-old Caucasian/Hispanic male with the classical clinical characteristics of PMLD with lack of myelination of the subcortical white matter, and absence of the splenium of corpus callosum. Exome sequencing in the trio revealed novel compound heterozygous pathogenic mutations in SNAP29 (p.Leu119AlafsX15, c.354DupG and p.0?, c.2T > C). Quantitative analysis of the patient’s blood cells through RNA sequencing identified a significant decrease in SNAP29 mRNA expression, while western blot analysis on fibroblast cells revealed a lack of protein expression compared to parental and control cells. Mutations in SNAP29 have previously been associated with cerebral dysgenesis, neuropathy, ichthyosis, and keratoderma (CEDNIK) syndrome. Typical skin features described in CEDNIK syndrome, such as generalized ichthyosis and keratoderma, were absent in our patient. Moreover, the early onset nystagmus and leukodystrophy were consistent with a PMLD diagnosis. These findings suggest that loss of SNAP29 function, which was previously associated with CEDNIK syndrome, is also associated with PMLD. Overall, our study expands the genetic spectrum of PMLD.


Similar content being viewed by others
Data deposition and access
The exome sequencing datasets have been added to the Database of Genotypes and Phenotypes (dbGaP; http://www.ncbi.nlm.nih.gov/gap) under project phs000816. Both SNAP29 variants have been reported to ClinVar (htpp://www.ncbi.nlm.nih.gov/clinvar/) under variation IDs 279932 and 279894 (NM_004782.3). The raw sequence data of the father, mother, and child were submitted to the Sequence Read Archive (http://www.ncbi.nlm.nih.gov/sra) with the following Biosample ID numbers: SAMN05687153 (father), SAMN05687449 (mother), SAMN05687373 (child) (DNA), and SAMN05687278 (child) (RNA).
References
Abdollahi MR, Morrison E, Sirey T, Molnar Z, Hayward BE, Carr IM, Sheridan E (2009) Mutation of the variant alpha-tubulin TUBA8 results in polymicrogyria with optic nerve hypoplasia. Am J Hum Genet 85(5):737–744. https://doi.org/10.1016/j.ajhg.2009.10.007
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, Sunyaev SR (2010) A method and server for predicting damaging missense mutations. Nat Methods. https://doi.org/10.1038/nmeth0410-248
Anders S, Pyl PT, Huber W (2014) HTSeq—a python framework to work with high-throughput sequencing data. Bioinformatics (Oxford, England) 31(2):166–169. https://doi.org/10.1093/bioinformatics/btu638
Ben-Salem S, Nara S, Al-Shamsi AM, Valle D, Ali BR, Al-Gazali L (2015) A new Arab family with CEDNIK syndrome suggests a possible founder effect for the c.223delG mutation. J Dermatol 42(8):821–822
Biancheri R, Rosano C, Denegri L, Lamantea E, Pinto F, Lanza F, Filocamo M (2013) Expanded spectrum of Pelizaeus-Merzbacher-like disease: literature revision and description of a novel GJC2 mutation in an unusually severe form. Eur J Hum Genet 21(1):34–39
Cingolani P, Platts A, Wang LL, Coon M, Nguyen T, Wang L, Ruden DM (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 6(2):80–92. https://doi.org/10.4161/fly.19695
Cooper GM, Goode DL, Ng SB, Sidow A, Bamshad MJ, Shendure J, Nickerson DA (2010) Single-nucleotide evolutionary constraint scores highlight disease-causing mutations. Nat Methods. https://doi.org/10.1038/nmeth0410-250
Diggle CP, Martinez-Garay I, Molnar Z, Brinkworth MH, White E, Fowler E, Sheridan E (2017) A tubulin alpha 8 mouse knockout model indicates a likely role in spermatogenesis but not in brain development. PLoS One 12(4):e0174264. https://doi.org/10.1371/journal.pone.0174264
Farwell KD, Shahmirzadi L, El-Khechen D, Powis Z, Chao EC, Tippin Davis B, Tang S (2015) Enhanced utility of family-centered diagnostic exome sequencing with inheritance model-based analysis: results from 500 unselected families with undiagnosed genetic conditions. Genet Med 17(7):578–586. https://doi.org/10.1038/gim.2014.154
Feinstein M, Markus B, Noyman I, Shalev H, Flusser H, Shelef I, Birk OS (2010) Pelizaeus-merzbacher-like disease caused by AIMP1/p43 homozygous mutation. Am J Hum Genet 87(6):820–828. https://doi.org/10.1016/j.ajhg.2010.10.016
Fuchs-Telem D, Stewart H, Rapaport D, Nousbeck J, Gat A, Gini M, Sprecher E (2011) CEDNIK syndrome results from loss-of-function mutations in SNAP29. Br J Dermatol 164(3):610–616. https://doi.org/10.1111/j.1365-2133.2010.10133.x
Hsu T, Coughlin CC, Monaghan KG, Fiala E, McKinstry RC, Paciorkowski AR, Shinawi M (2017) Phenotypic and molecular characterization of an additional patient and review of the literature. Child Neurol Open. https://doi.org/10.1177/2329048X17733214
Kircher M, Witten DM, Jain P, O’roak BJ, Cooper GM, Shendure J (2014) A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 46(3):310–315. https://doi.org/10.1038/ng.2892
Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, Exome Aggregation Consortium (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536(7616):285–291. https://doi.org/10.1038/nature19057
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25(14):1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Lossos A, Elazar N, Lerer I, Schueler-Furman O, Fellig Y, Glick B, Peles E (2015) Myelin-associated glycoprotein gene mutation causes Pelizaeus-Merzbacher disease-like disorder. Brain 138(9):2521–2536. https://doi.org/10.1093/brain/awv204
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-Seq data with DESeq2. bioRxiv. Cold Spring Harbor Labs J. https://doi.org/10.1101/002832
Madry J, Hoffman-Zacharska D, Królicki L, Jakuciński M, Friedman A (2010) PLP1 gene duplication as a cause of the classic form of Pelizaeus-Merzbacher disease—case report. Neurol Neurochir Pol 44(5):511–515. https://doi.org/10.1016/S0028-3843(14)60142-0
McDonald-McGinn D, Fahiminiya S, Revil T, Nowakowska B, Suhl J, Bailey A, Jerome-Majewska L (2013) Hemizygous mutations in SNAP29 unmask autosomal recessive conditions and contribute to atypical findings in patients with 22q11.2DS. J Med Genet 50:80–90. https://doi.org/10.1136/jmedgenet-2012-101320
McDonald-McGinn DM, Sullivan KE, Marino B, Philip N, Swillen A, Vorstman JAS, Bassett AS (2015) 22q11.2 deletion syndrome. Nat Rev Dis Primers. https://doi.org/10.1038/nrdp.2015.71
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, DePristo MA (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20(9):1297–1303. https://doi.org/10.1101/gr.107524.110
Morelli E, Mastrodonato V, Beznoussenko GV, Mironov AA, Vaccari T (2016) An essential step of kinetochore formation controlled by the SNARE protein Snap29. Embo J 35(20):1–40. https://doi.org/10.15252/embj.201693991
Rapaport D, Lugassy Y, Sprecher E, Horowitz M (2010) Loss of snap29 impairs endocytic recycling and cell motility. PLoS One. https://doi.org/10.1371/journal.pone.0009759
Schardt A, Brinkmann BG, Mitkovski M, Sereda MW, Werner HB, Nave K (2009) The SNARE protein SNAP-29 interacts with the GTPase Rab3A: implications for membrane trafficking in Myelinating Glia. J Neurosci Res 87:3465–3479. https://doi.org/10.1002/jnr.22005
Schiller SA, Seebode C, Wieser GL, Goebbels S, Möbius W, Horowitz M, Emmert S (2016) Establishment of two mouse models for CEDNIK syndrome reveals the pivotal role of SNAP29 in epidermal differentiation. J Investig Dermatol 136(3):672–679. https://doi.org/10.1016/j.jid.2015.12.020
Schwarz JM, Rödelsperger C, Schuelke M, Seelow D (2010) MutationTester evaluates disease-causing potential of sequence alterations. Nat Methods. https://doi.org/10.1038/nmeth0810-575
Sprecher E, Ishida-Yamamoto A, Mizrahi-Koren M, Rapaport D, Goldsher D, Indelman M, Mandel H (2005) A mutation in SNAP29, coding for a SNARE protein involved in intracellular trafficking, causes a novel neurocutaneous syndrome characterized by cerebral dysgenesis, neuropathy, ichthyosis, and palmoplantar keratoderma. Am J Hum Genet 77(2):242–251. https://doi.org/10.1086/432556
Su Q, Mochida S, Tian JH, Mehta R, Sheng ZH (2001) SNAP-29: general SNARE protein that inhibits SNARE disassembly and is implicated in synaptic transmission. Proc Natl Acad Sci USA 98(24):14038–14043. https://doi.org/10.1073/pnas.251532398
Villegas J, McPhaul M (2005) Establishment and culture of human skin fibroblasts. In: Frederick MA (ed) Current protocols in molecular biology, chapter 28, unit 283. Wiley, Hoboken. https://doi.org/10.1002/0471142727.mb2803s71
Widmann J, Stombaugh J, McDonald D, Chocholousova J, Gardner P, Iyer MK, Knight R (2012) RNASTAR: an RNA STructural alignment repository that provides insight into the evolution of natural and artificial RNAs. RNA (New York, NY) 18(7):1319–1327. https://doi.org/10.1261/rna.032052.111
Yamamoto T, Yamamoto-Shimojima K, Ueda Y, Imai K, Takahashi Y, Imagawa E, Miyake N, Matsumoto N (2018) Independent occurrence of de novo HSPD1 and HIP1 variants in brothers with different neurological disorders—leukodystrophy and autism. Human Genome Var 5:18. https://doi.org/10.1038/s41439-018-0020-z
Zhu X, Need AC, Petrovski S, Goldstein DB (2014) One gene, many neuropsychiatric disorders: lessons from Mendelian diseases. Nat Neurosci. https://doi.org/10.1038/nn.3713
Acknowledgements
The authors thank the family for participating in this study and all the previous members of the C4RCD research group not included in the author list.
Funding
This work was supported by the TGen Foundation funding to Center for Rare Childhood Disorders (C4RCD).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to disclose.
Informed consent
Written informed consent for publication of clinical details and clinical images was obtained from the legally authorized representative and the patient’s family. The study protocol and consent procedure was approved by the Western Institutional Review Board (WIRB; study number 20120789).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2019_2077_MOESM2_ESM.tiff
A list of variants of unknown significance (VUS) found by WES in our patient. Maximum MAF cut-off is 0.001 (ExAC, ESP, 1000G). Listed are also number of homozygotes, heterozygotes or hemizygotes found in the gnomAD database (TIFF 94 kb)
439_2019_2077_MOESM3_ESM.tif
Images of the patient at 12 years of age, showing acne in the Front chest (A), Back (B), Hand (C), and Leg (D) (TIFF 1051 kb)
439_2019_2077_MOESM4_ESM.tif
Heatmap of expression of the significantly dysregulated genes between the patient and unaffected age-matched controls (DeSeq 2; PAdj < 0.05). Expression values are plotted as Rlog (counts) (TIFF 331 kb)
Rights and permissions
About this article
Cite this article
Llaci, L., Ramsey, K., Belnap, N. et al. Compound heterozygous mutations in SNAP29 is associated with Pelizaeus-Merzbacher-like disorder (PMLD) . Hum Genet 138, 1409–1417 (2019). https://doi.org/10.1007/s00439-019-02077-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-019-02077-7