Abstract
Ticks are blood-sucking arthropods that can transmit pathogens to their host. As insular ecosystems can enhance tick-host interactions, this study aimed to understand tick diversity, pathogen presence, and their respective associations in the Azores and Madeira archipelagos. Unfed or partially engorged ticks (n = 120) were collected from 58 cats and dogs in the Azores (n = 41 specimens) and Madeira (n = 79 specimens) from November 2018 to March 2019. Vector identification was based on morphology and molecular criteria. For pathogen sequencing, 18S gene fragment for Babesia/Hepatozoon and gltA for Rickettsia were performed. Sequence data was explored using BLAST and BLAST and phylogenetic inference tools. In the Azores, Ixodes hexagonus, I. ventalloi, and Rhipicephalus sanguineus (n = 6; 14.6%, n = 6; 14.6%, and n = 29; 70.7% respectively) were found and in Madeira I. ricinus and R. sanguineus (n = 78, 98.7%; and n = 1, 1.3%; respectively) were identified. Tick COI markers confirmed species highlighting confirmation of R. sanguineus s.s. and genotype A of I. ventalloi. In the Azores Islands, the detected Rickettsia massiliae was linked to R. sanguineus (dogs and cats) and I. hexagonus (dogs), and in Madeira Island, R. monacensis (dogs) and Hepatozoon silvestris (cats) were found associated with I. ricinus. Further, I. ventalloi presence in the Azores expands west its known range, and Hepatozoon silvestris in Madeira may suggest that I. ricinus could have a role as a potential vector. Finally, as R. massiliae and R. monacensis presence underlines public health risks, surveillance by health authorities is crucial as pathogen-tick interactions may drive disease spread, therefore monitoring remains pivotal for disease prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ticks are obligatory hematophagous arthropods that can potentially parasitize terrestrial vertebrates and act as vectors of pathogens such as bacteria, viruses, protozoa, and helminths (Ramos et al. 2013; Sonenshine and Roe 2014; Estrada-Peña et al. 2017; de la Fuente et al. 2017). In addition to the direct impact caused by pathologies, ticks can also have an indirect negative economic impact on countries’ agro-industry, due to the reduction in meat and milk production as well as body injuries induced by blood spoliation and secondary infections in livestock (Jongejan and Uilenberg 2004; Eskezia 2016).
Ticks are considered among the most important pathogen vectors worldwide, surpassed only by mosquitoes (de la Fuente 2008). About 10% of known tick species are considered to have medical and veterinary importance (de la Fuente et al. 2017). This fact, associated with diseases they can cause, makes ticks the most common pathogen agent vectors in Europe (Jahfari et al. 2016; de la Fuente et al. 2017).
Azores and Madeira Atlantic archipelagos are part of Macaronesia (which also includes the Canary Islands and Cape Verde), an important biogeographical region due to its rich biodiversity, included in the Natura 2000 network. Previous studies based only on tick morphology have identified six different species in Madeira Island (de Almeida 1997): Ixodes ricinus (Linnaeus, 1758), Haemaphysalis punctata Canestrini and Fanzago, 1878, Rhipicephalus bursa Canestrini and Fanzago, 1878, R. sanguineus (Latreille, 1806), R. (Boophilus) annulatus (Say, 1821), and Hyalomma lusitanicum Koch, 1844. In the Azores Islands, ten species are known: I. frontalis (Panzer, 1798), I. hexagonus Leach, 1815, Dermacentor marginatus (Sulzer, 1776), H. lusitanicum, H. marginatum Koch, 1844, H. punctata, R. sanguineus, R. bursa, R. turanicus Pomerantzev, 1940, and R. (B.) annulatus (Dias 1992; Borges et al. 2010; Literak et al. 2015).
Data on tick-borne diseases or pathogens in these regions are scarce and dispersed and mainly related to clinical reports on human disease. Nonetheless, in Madeira Island Borrelia burgdorferi s.l (B. afzelii, B. burgdorferi s.s. and B. garinii) (Matuschka et al. 1998), Anaplasma phagocytophilum (Santos et al. 2004; Carvalho et al. 2008), Rickettsia monacensis, R. helvetica, and B. lusitaniae have been detected in I. ricinus (Carvalho et al. 2008; de Sousa et al. 2012). In the Azores, R. massiliae was isolated from R. sanguineus collected in one dog (Foley and Reeves 2014); B. turdi was detected in I. frontalis from birds (Literak et al. 2015); Phlebovirus was isolated from I. hexagonus and R. sanguineus (Pimentel et al. 2019); Hepatozoon sp. was observed in reptiles (Rund et al. 2019); and Theileria equi was detected in imported horses (Baptista et al. 2013).
In this exploratory study, our main objectives are to provide a precise update on the diversity of ixodids and their pathogens in pets (dogs and cats) from the Madeira and Azores Islands. Specifically, we aim to:
-
(a)
Identify the different species of ixodids present in these Atlantic islands
-
(b)
Investigate potential links between molecularly identified pathogens and their respective putative vectors. These findings will allow health authorities to better pinpoint their efforts to monitor, minimize, and control potential outbreaks caused by these vectors and pathogens
Material and methods
Tick collection and morphological identification
Ticks included in this study were collected from 36 dogs and 22 cats that attended veterinary clinics located in urban areas for routine visits or other health issues. A total of 120 ixodids (n = 41 in the Azores Islands and n = 79 in Madeira Island) were sampled during the years 2018 and 2019. Ticks were collected with tick removal tweezers during external exams in the veterinary clinic, following international recommendations and rules for handling animals, and carried out with the permission of local authorities. These specimens became part of a tick collection housed at the University of Lisbon, the “Faculdade de Ciências and Instituto Superior de Agronomia Tick Collection Database” (FCISA). Specimens were stored in tubes (1 tube/host) with 70% ethanol, and kept at room temperature, until laboratory processing not longer than two weeks.
Ixodids were examined and photographed using a Leitz Laborlux K light microscope and a Leica M165C stereomicroscope coupled to a Leica DFC420 digital microscope camera with the Leica Application Suite (LAS) live measurement software (Microsystems 2009). Identification was based on external criteria of their morphology according to several authors (Heylen et al. 2014; Coimbra-Dores et al. 2016; Estrada-Peña et al. 2017; Hornok et al. 2017, 2021; Nava et al. 2018).
Mean values of ticks found in hosts were compared between islands using Wilcoxon signed rank tests implemented in R v4.3.1. The respective script can be found in in this GitLab repository.
Molecular analyses
Vectors
Fed ticks (unengorged or partially engorged) tick specimens were washed twice in 70% ethanol to remove external contaminants and left to dry at room temperature. Subsequently, they were crushed and placed overnight in a Proteinase K solution (20mg/mL – NZYTech) at 55 °C.
Genomic DNA was extracted using a commercial kit (E.Z.N.A. Tissue DNA Isolation Kit, Omega Bio-tek) following the manufacturer’s guidelines. After extraction, DNA samples were quantified using a NanoDrop 1000 spectrophotometer (Thermo Fisher). All aliquots were stored at 4 °C until processing (not more than 1 week).
To validate their morphological identification, eighteen specimens (adults of R. sanguineus, I. ventalloi Gil Collado, 1936, and I. ricinus and nymphs of I. hexagonus) were amplified by conventional polymerase chain reaction (PCR) for the mitochondrial Cytochrome C Oxidase subunit I (COI) fragment using the following primers: LCO1490, 5′-GGTCAACAAATCATAAAGATATTGG-3′; HCO2198, 5′-TAAACTTCAGGGTGACCAAAAATCA-3′ (Folmer et al. 1994). Each reaction was performed with a total volume of 25 μL consisting of 12.5μL of Dreamtaq Green PCR Master Mix (2 ×) (Thermo Scientific), 0.4 μM of each primer and 3 μL of extracted DNA. The PCR protocol consisted of an initial denaturation step at 95 °C for 5 min, 35 cycles of 95 °C for 20 s, 45 °C for 45 s, and 72 °C for 45 s followed by a final extension step at 72 °C for 7 min.
Pathogens
For pathogen screening, all 120 ticks were tested by PCR amplification. Babesia/Hepatozoon 18S gene was amplified using 18SR/5′CCAGCAGCCGCGGTAATTC3′/18S F/5′CTTTCGCAGTAGTTYGTCTTTAACAAATCT3′ primers (Tabar et al. 2008). Rickettsia gltA gene was amplified using gltAF/5′CCTATGGCTATTATGCTTGC3′/gltAR/5′ATTGCAAAAAGTACAGTCAACA3′ primers (Roux et al. 1997). PCR amplifications were conducted in a final volume of 20 μL, comprising 10 μL of PerfeCta SYBR Green Supermix, Rox (Quanta bio), 0.3 μM of each primer, and 2.8 μL of template DNA.
Regarding the PCR conditions, for Rickettsia screening, an initial denaturation step at 95 °C for 5 min was followed by 45 cycles of a three-step amplification: 95 °C for 15 s, 51.6 °C for 30 s, and 65 °C for 45 s. For Babesia/Hepatozoon species, an initial denaturation step at 95 °C for 5 min was followed by 35 cycles of a three-step amplification: 95 °C for 15 s, 64 °C for 30 s, and 65 °C for 45 s.
To validate amplification efficiency, all PCR products with fragments with approximately 400 qb were verified by 2% agarose gel electrophoresis and purified with the commercial kit SureClean Plus (Bioline). Twenty-five successfully amplified samples (23 from Azores and 11 from Madeira) were outsourced for sequencing (STABVIDA, Portugal). Newly generated sequences were submitted to NCBI, and accession numbers can be consulted in Online Resource Tables 1 and 2.
Genetic data analyses
Sequence trace files were quality-controlled and exported as FASTA sequences using SeqTrace v0.9.1 (Stucky 2012). Obtained sequences were split into three separate datasets (Online Resource Tables 3, 4, and 5), one containing the 18S sequences of Hepatozoon samples, the other containing Rickettsia’s gltA sequences, and the third containing COI sequences of vector samples. Each obtained sequence was then queried against NCBI’s “nt” database using BLASTN v2.14.1 + (Altschul 1997) with default parameters to identify agent sequences and to confirm the identification of vector sequences.
For maximum reproducibility, analysis downstream of this point was fully automated using Snakemake (Mölder et al. 2021), resorting to a Docker image for process containerization. The workflow, scripts, and container build script can be found in this GitLab repository.
Each original dataset was complemented with sequences from NCBI (The respective accession numbers and references can be consulted in Online Resource Tables 3–5, one table per dataset). To infer phylogenies, sequences were aligned using mafft v7.520 (Katoh and Toh 2008) with “–auto” parameters and trimmed to the length of the shortest original sequence at the 5′ and 3′ ends. The best model for each alignment was selected using ModelTest-NG v0.1.7 (Darriba et al. 2020). The model was then used in RAxML-NG v1.1.0 (Kozlov et al. 2019) to infer a maximum likelihood (ML) best tree supported by 1000 Felsenstein bootstrap replicates (using a seed value of 12358). MrBayes v3.2.6 (Ronquist et al. 2012) was used to infer Bayesian trees, using default priors and 1,000,000 MCMC iterations. Inferred phylogenetic trees were plotted with a Python script using the Toytree library v2.0.5 (Eaton 2020).
Results
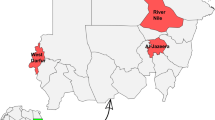
Morphological analyses of the sampled individuals revealed four different species of ticks: three in the Azores: I. hexagonus, I. ventalloi, and R. sanguineus (n = 6, 14.6%; n = 6, 14.6%; and n = 29, 70.7%; respectively), and two in Madeira: I. ricinus, and R. sanguineus (n = 78, 98.7%; and n = 1, 1.3%; respectively) (Fig. 1) (Table 1).
a Ixodes ventalloi ventral basis capituli with a horn shape oriented inwards and ventral face of the first article with a distinct spine; b Rhipicephalus sanguineus male ventral face with adanal plate, accessory adanal plate, and spiracular area with an ending inferior to half of the adjacent festoon width; c broad U-shape of female genitalia aperture with sclerites distant from each other
The great majority of tick specimens were adults, except for the six specimens of I. hexagonus, which were nymphs (n = 6, 5.0%). Of the adult specimens, 70.8% (n = 85) were female and 24.2% (n = 29) male. Concerning tick hosts, 81 (67.5%) specimens were collected from dogs and 39 (32.5%) from cats. Despite the limited sample size, parasitical load showed higher values for Azorean hosts (M = 4.13, SD = 3.64 ticks per dog and M = 2.00, SD = 0.82 ticks per cat) than for Madeira (M = 1.68, SD = 1.49 ticks per dog and M = 1.72, SD = 0.59 tocks per cat). Wilcoxon signed-rank tests revealed differences in the mean number of ticks per dog between the Azores and Madeira Islands (p = 0.00979), but not in cats (p = 0.477). Specimens of I. ricinus and R. sanguineus were found in both hosts, I. hexagonus specimens were only found in dogs, whereas I. ventalloi specimens were collected exclusively in cats.
The performed BLAST search of the pathogens’ sequences against NCBI’s “nt” database revealed that all sequences matched with similar sequences from species they were morphologically identified as. In Madeira Island, I. ricinus sequences did so with 100% and 99.85% identity (MG432681.1), and the R. sanguineus sequences corresponded with 100% identity (MF426013.1). In the Azorean samples, the I. hexagonus sequences matched with 99.71% identity (MG432679.1), the I. ventalloi sequences had 99.23% identity (LC508372.1), and R. sanguineus showed 99.85% identity (MF426019.1).
The obtained COI ML best tree shows that each species forms a well-supported monophyletic clade, except for I. ricinus, whose monophyletic clade has the lowest support of all groups (bootstrap = 61, posterior probability = 80), but is included in a monophyletic, well-supported clade with I. inopinatus. It is worth noting that Azorean I. ventalloi samples are closer to those of “Genogroup A” rather than “Genogroup B” samples. These monospecies clades contain both samples from this study and pre-identified samples from NCBI’s “nt” database, thus confirming morphological species identification results (Fig. 2).
Maximum likelihood (ML)-based tick phylogram inferred from cytochrome oxidase I gene fragment. Samples’ labels of individuals sequenced for this work start with the species name, followed by the sample ID(s). Labels of sequences obtained from NCBI start with the respective accession number, followed by the species name
In Madeira Island, R. monacensis was successfully amplified and sequenced from five I. ricinus samples (four females and one male). Hepatozoon silvestris sequences were obtained from two specimens of I. ricinus (one female and one male) (Fig. 4), both of which were also positive for R. monacensis.
In the Azorean archipelago, 23 ticks (R. sanguineus, n = 17; I. hexagonus, n = 6) were tested for pathogens. Eight ticks (34.78%) tested positive for Rickettsia massilliae in both R. sanguineus (n = 7/17, 41.18%) and I. hexagonus (n = 1/6, 16.67%). Most of these were found in Terceira Island (n = 6/17, 35.29%) R. sanguineus, but some detections occurred in S. Miguel (n = 1/17, 5.88% in R. sanguineus, and n = 1/6, 16.67% in I. hexagonus).
In Madeira Island, from the 11 tested ticks (R. sanguineus, n = 1; I. ricinus, n = 10), pathogen agents were only found in I. ricinus. Hepatozoon sylvestris (n = 2/11, 18.18%) was found only in Ribeira Brava (n = 2/10, 20.0%). Rikettsia monocensis (n = 6/10, 60.0%) was found in Ribeira Brava (n = 2/10, 20.00%), Sta Cruz (n = 2/10, 20.00%) Calheta (n = 1/10, 10.00%), and Machico (n = 1/10, 10.00%).
In the Azores archipelago, only R. massiliae was successfully sequenced (one haplotype), but sequences were obtained from both R. sanguineus (three female and four male specimens) and I. hexagonus (six nymph) vectors (Fig. 3).
Maximum likelihood (ML) phylogram of Rickettsia (gltA gene fragment sequences). Labels of samples sequenced for this work start with the word “Putative,” followed by the assumed species name, and the vector’s species and sample ID. Sequences obtained from NCBI start with the respective accession number and otherwise follow the same scheme as the original samples’. Each agent species is represented in a different color. Nodes are labelled with Bootstrap support (BS) from ML, followed by posterior probability (PP) from Bayesian inference as BS/PP. A “-” indicates either BS < 40 or PP < 0.40. If both BS and PP are below this threshold, the node is unlabeled. Candidatus Rickettsia mendelli was used as an outgroup for tree rooting
A BLAST search querying sequences from this study revealed that Rickettsia haplotypes (n = 3) from Madeira Island have a 100% identity with ten different Rickettsia sequences: the more frequent haplotype with four R. monacensis and one Rickettsia sp. sequences found in the “nt” database (GenBank)(MH618388.1, KU586332.1, KU961539.1, LN794217.1, AF141906.1, and LC060719.1) and the less frequent haplotype with four Rickettsia sp. sequences (LC060716.1, OP125492.1, OP125489.1, and AF140706.1). Likewise, the single haplotype found in the Azores was matched with 100% identity to three R. massiliae sequences (KU498299.1, CP003319.1, and KT032119.1) and one Rickettsia sp. sequence (U59720.1) from the “nt” database. Phylogenetic analyses of Rickettsia sequences revealed that samples from both species found in our study form well-supported monophyletic clades with sequences of their respective species from other studies, confirming what the BLAST search had already shown (Fig. 3).
An NCBI BLAST search revealed that obtained Hepatozoon sequences had a 99.47% identity with seven H. silvestris sequences (MH078194.1, MF614155.1, KY649445.1, KX757032.1, KX757031.1, OQ816783.1, and OQ207707.1), which led us to identify it as such. Phylogenetic analyses with other close Hepatozoon species (H. canis and H. felis) confirm this assessment (Fig. 4).
Maximum likelihood (ML) phylogram of Hepatozoon (18S gene fragment sequences). Labels of samples sequenced for this work start with the word “Putative,” followed by the assumed species name, and the vector’s species and sample ID. Sequences obtained from NCBI start with the respective accession number and otherwise follow the same scheme as the original samples’. Each agent species is represented in a different color. Nodes are labelled with bootstrap support (BS) from ML, followed by posterior probability (PP) from Bayesian inference as BS/PP. A “-” indicates whether BS < 40 or PP < 0.40. If both BS and PP are below this threshold, the node is unlabeled. Babesia sp. UN1 was used as an outgroup for tree rooting
Discussion
Although this work is of relatively small scale, and in no way a systematic study on the host, vector, and agent tick system in the Macaronesia, which is urgently needed, various important observations were made.
The presence of I. ventalloi (six specimens) in three of 11 analyzed cats, from Faial and S. Miguel Islands, confirms this vector’s presence in these islands. According to existing literature (Petney et al. 2017; Estrada-Peña et al. 2018; Santos and Santos-Silva 2019), this is the westernmost I. ventalloi that has ever been reported.
Phylogenetic analyses of I. ventalloi COI sequences confirmed its inclusion in “Genogroup A” (Latrofa et al. 2017), similar to specimens from mainland Portugal (Santos et al. 2018).
The Apicomplexa H. silvestris, previously found in wild and domestic cats in Central Europe (Hodžić et al. 2017; Hodžić and Alić 2023), was found in male and female Ixodes ricinus specimens in Madeira Island. This corroborates the results of (Duplan et al. 2018) which reported H. silvestris in fed I. ricinus in Wales (UK). These findings are insufficient to establish I. ricinus as biological vector (Hodžić and Alić 2023), but it adds more weight to that hypothesis, especially considering that males of this species exhibit blood-feeding behavior (Gray 1987). The fact that Hepatozoon spp. is known to cause severe, or even lethal pathologies in cats (Kegler et al. 2018; Simonato et al. 2022), coupled with the suspicion of its transmission by I. ricinus (Uiterwijk et al. 2023) highlight the importance of implementing systematic monitoring of this vector. Despite these molecular detections, none of the sampled hosts was reported to display any tick-borne disease symptoms by the veterinary clinics.
We also found evidence of two different R. monacensis haplotypes associated with I. ricinus, a known vector for various agents, from dogs in Madeira Island. It is worth noting that the endemic lizard species, Teira dugesii, is a known host for this tick species and its pathogens (de Sousa et al. 2012). As such, it is likely to play an important role in the dynamics of vector-borne diseases (Carvalho et al. 2008; de Sousa et al. 2012; Matuschka et al. 1998; Santos et al. 2004), not only as a vector, but also as a reservoir. This is a possible explanation for the relatively high prevalence (and diversity) of R. monacensis in Madeira hosts. A stable population of these agents that can infect pets has a high potential to become a threat to human health.
This study further identified the association of R. massiliae with R. sanguineus and I. hexagonus in the Azores Archipelago, albeit on different islands. Rickettsia massiliae’s association with R. sanguineus is known (Parola et al. 2013), but the agent’s presence in I. hexagonus, in new information, even though this species is a known competent vector of multiple pathogen agents (Jahfari et al. 2017). This unexpected association can either be attributed to the tick feeding on infected animals or to an adaptation of the SFG bacteria to a new vector. Here too, a larger, systematic study is required to fully understand this finding.
On another note, sequences from ticks identified as R. sanguineus, another tick species known as a competent pathogen vector (Solano-Gallego et al. 2008; Baneth et al. 2015; Snellgrove et al. 2020), form a monophyletic clade with R. sanguineus samples from NCBI (Coimbra-Dores et al. 2018). This places our samples in the same temperate lineage that can be found across Europe, North Africa, North, and South America (Burlini et al. 2010; Coimbra-Dores et al. 2018; Nava et al. 2018) and in the Canary Islands (Chitimia-Dobler et al. 2019). As it stands, this information rejects the hypothesis that the species found in the Azores and Madeira could be R. linnaei (Audouin, 1826) (Šlapeta et al. 2022), which can be found in tropical regions such as the Cape Verde Archipelago (Coimbra-Dores et al. 2018). Making this distinction is important, since these two species were only recently “upgraded” from lineages (Nava et al. 2018).
Even though our study suffers from the abovementioned limitations that prevent it from being considered representative of a global assessment in these insular ecosystems, it functions as a first insight into the characterization of insular vectors and agents. Addressing these limitations on a larger scale, a systematic approach may lead to finding further interesting associations with wider public health impact. Such monitoring is critical for developing effective disease prevention and mitigation measures and safeguarding human and animal health, especially considering the current fast-paced climate change scenario coupled with large-scale movements of people and animals.
Conclusions
Here, we present the first observation of I. ventalloi (Genogroup A) in the Azores, which corresponds to its westernmost distribution range.
We also challenge assumptions by detecting two previously unknown associations: R. massiliae with I. hexagonus (Azores), and H. silvestris to I. ricinus (Madeira). Although the associations are confirmed, these ticks’ roles as vectors for the pathogens warrant further research.
Despite the limitations in sample size, sequencing, and short time period, this study highlights the importance of monitoring vectors and pathogens in a globalized, changing world. Our findings unveil more of the complex interactions between all these factors that require comprehensive strategies in evolving disease landscapes.
Data availability
All sequences are deposited in GeneBank, and the accession numbers are available in Online Resource Tables 1 and 2. The code used for data analyses can be found in this GitLab repository (https://gitlab.com/StuntsPT/ticks-and-agents). It is also mirrored in Zenodo, under https://doi.org/10.5281/zenodo.8319715.
References
Altschul S (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Baneth G, Harrus S, Gal A, Aroch I (2015) Canine vector-borne co-infections: Ehrlichia canis and Hepatozoon canis in the same host monocytes. Vet Parasitol 208:30–34. https://doi.org/10.1016/j.vetpar.2014.12.013
Baptista C, Lopes MS, Tavares AC et al (2013) Diagnosis of Theileria equi infections in horses in the Azores using cELISA and nested PCR. Ticks Tick-Borne Dis 4:242–245. https://doi.org/10.1016/j.ttbdis.2012.11.008
Borges P, Costa A, Cunha R et al (2010) Listagem dos organismos terrestres e marinhos dos Açores, 1st edn. Principia, Cascais
Burlini L, Teixeira KRS, Szabó MPJ, Famadas KM (2010) Molecular dissimilarities of Rhipicephalus sanguineus (Acari: Ixodidae) in Brazil and its relation with samples throughout the world: is there a geographical pattern? Exp Appl Acarol 50:361–374. https://doi.org/10.1007/s10493-009-9321-8
Carvalho ILD, Milhano N, Santos AS et al (2008) Detection of Borrelia lusitaniae, Rickettsia sp. IRS3, Rickettsia monacensis, and Anaplasma phagocytophilum in Ixodes ricinus Collected in Madeira Island. Portugal Vector-Borne Zoonotic Dis 8:575–580. https://doi.org/10.1089/vbz.2007.0245
Chitimia-Dobler L, Kurzrock L, Molčányi T et al (2019) Genetic analysis of Rhipicephalus sanguineus sensu lato ticks, parasites of dogs in the Canary Islands, Cyprus, and Croatia, based on mitochondrial 16S rRNA gene sequences. Parasitol Res 118:1067–1071. https://doi.org/10.1007/s00436-019-06214-z
Coimbra-Dores MJ, Nunes T, Dias D, Rosa F (2016) Rhipicephalus sanguineus (Acari: Ixodidae) species complex: morphometric and ultrastructural analyses. Exp Appl Acarol 70:455–468. https://doi.org/10.1007/s10493-016-0095-5
Coimbra-Dores MJ, Maia-Silva M, Marques W et al (2018) Phylogenetic insights on Mediterranean and Afrotropical Rhipicephalus species (Acari: Ixodida) based on mitochondrial DNA. Exp Appl Acarol 75:107–128. https://doi.org/10.1007/s10493-018-0254-y
Darriba D, Posada D, Kozlov AM et al (2020) ModelTest-NG: a new and scalable tool for the selection of DNA and protein evolutionary models. Mol Biol Evol 37:291–294. https://doi.org/10.1093/molbev/msz189
de Almeida VCT (1997) Ixodídeos da ilha da Madeira, contribuição ao seu estudo. Dissertation, Universidade de Évora
de la Fuente J, Antunes S, Bonnet S, et al. (2017) Tick-pathogen interactions and vector competence: identification of molecular drivers for tick-borne diseases. Front Cell Infect Microbiol 7. https://doi.org/10.3389/fcimb.2017.00114
de la Fuente JDL (2008) Overview: ticks as vectors of pathogens that cause disease in humans and animals. Front Biosci 6938. https://doi.org/10.2741/3200
de Sousa R, de Carvalho IL, Santos AS et al (2012) Role of the lizard Teira dugesii as a potential host for Ixodes ricinus tick-borne pathogens. Appl Environ Microbiol 78:3767–3769. https://doi.org/10.1128/AEM.07945-11
Dias JAT (1992) Subsídios para o conhecimento da fauna ixodideológica dos Açores. O Médico Veterinário 7:17–23
Duplan F, Davies S, Filler S et al (2018) Anaplasma phagocytophilum, Bartonella spp., haemoplasma species and Hepatozoon spp. in ticks infesting cats: a large-scale survey. Parasites Vectors 11:201. https://doi.org/10.1186/s13071-018-2789-5
Eaton DAR (2020) Toytree: a minimalist tree visualization and manipulation library for Python. Methods Ecol Evol 11:187–191. https://doi.org/10.1111/2041-210X.13313
Eskezia BG (2016) Review on the impact of ticks on livestock health and productivity. J Biol, Agric Healthcare 6:1
Estrada-Peña A, Mihalca AD, Petney TN (eds) (2017) Ticks of Europe and North Africa: a guide to species identification, 1st ed. 2017. Springer International Publishing: Imprint: Springer, Cham
Estrada-Peña A, Venzal JM, Nava S (2018) Redescription, molecular features, and neotype deposition of Rhipicephalus pusillus Gil Collado and Ixodes ventalloi Gil Collado (Acari, Ixodidae). Zootaxa 4442. https://doi.org/10.11646/zootaxa.4442.2.4
Foley EH, Reeves WK (2014) Rickettsia massiliae (Latreille) from the Azores 1. J Agric Urban Entomol 30:25–27. https://doi.org/10.3954/JAUE14-04.1
Folmer O, Black M, Hoeh W et al (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299
Gray JS (1987) Mating and behavioural diapause in Ixodes ricinus L. Exp Appl Acarol 3:61–71. https://doi.org/10.1007/BF01200414
Heylen D, de Coninck E, Jansen F, Madder M (2014) Differential diagnosis of three common Ixodes spp. ticks infesting songbirds of Western Europe: Ixodes arboricola, I. frontalis and I. ricinus. Ticks Tick-Borne Dis 5:693–700. https://doi.org/10.1016/j.ttbdis.2014.05.006
Hodžić A, Alić A (2023) Hepatozoon silvestris: an emerging feline vector-borne pathogen in Europe? Trends Parasitol 39:163–166. https://doi.org/10.1016/j.pt.2022.12.001
Hodžić A, Alić A, Prašović S et al (2017) Hepatozoon silvestris sp. nov.: morphological and molecular characterization of a new species of Hepatozoon (Adeleorina: Hepatozoidae) from the European wild cat ( Felis silvestris silvestris ). Parasitology 144:650–661. https://doi.org/10.1017/S0031182016002316
Hornok S, Sándor AD, Beck R et al (2017) Contributions to the phylogeny of Ixodes (Pholeoixodes) canisuga, I. (Ph.) kaiseri, I. (Ph.) hexagonus and a simple pictorial key for the identification of their females. Parasites Vectors 10:545. https://doi.org/10.1186/s13071-017-2424-x
Hornok S, Meyer-Kayser E, Kontschán J et al (2021) Morphology of Pholeoixodes species associated with carnivores in the western Palearctic: pictorial key based on molecularly identified Ixodes (Ph.) canisuga, I. (Ph.) hexagonus and I. (Ph.) kaiseri males, nymphs and larvae. Ticks Tick-borne Dis 12:101715. https://doi.org/10.1016/j.ttbdis.2021.101715
Jahfari S, Hofhuis A, Fonville M et al (2016) Molecular detection of tick-borne pathogens in humans with tick bites and erythema migrans, in the Netherlands. PLoS Negl Trop Dis 10:e0005042. https://doi.org/10.1371/journal.pntd.0005042
Jahfari S, Ruyts SC, Frazer-Mendelewska E et al (2017) Melting pot of tick-borne zoonoses: the European hedgehog contributes to the maintenance of various tick-borne diseases in natural cycles urban and suburban areas. Parasites Vectors 10:134. https://doi.org/10.1186/s13071-017-2065-0
Jongejan F, Uilenberg G (2004) The global importance of ticks. Parasitology 129:S3–S14. https://doi.org/10.1017/S0031182004005967
Katoh K, Toh H (2008) Recent developments in the MAFFT multiple sequence alignment program. Brief Bioinform 9:286–298. https://doi.org/10.1093/bib/bbn013
Kegler K, Nufer U, Alic A, Posthaus H, Olias P, Basso W (2018) Fatal infection with emerging apicomplexan parasite Hepatozoon silvestris in a domestic cat. Parasit Vectors 11:428. https://doi.org/10.1186/s13071-018-2992-4
Kozlov AM, Darriba D, Flouri T et al (2019) RAxML-NG: a fast, scalable and user-friendly tool for maximum likelihood phylogenetic inference. Bioinformatics 35:4453–4455. https://doi.org/10.1093/bioinformatics/btz305
Latrofa MS, Giannelli A, Persichetti MF et al (2017) Ixodes ventalloi: morphological and molecular support for species integrity. Parasitol Res 116:251–258. https://doi.org/10.1007/s00436-016-5286-9
Literak I, Norte AC, Núncio MS et al (2015) Ticks on passerines from the Archipelago of the Azores as hosts of borreliae and rickettsiae. Ticks Tick-Borne Dis 6:607–610. https://doi.org/10.1016/j.ttbdis.2015.05.003
Matuschka F-R, Klug B, Schinkel TW et al (1998) Diversity of European lyme disease spirochetes at the southern margin of their range. Appl Environ Microbiol 64:1980–1982. https://doi.org/10.1128/AEM.64.5.1980-1982.1998
Mölder F, Jablonski KP, Letcher B et al (2021) Sustainable data analysis with Snakemake. F1000res 10:33. https://doi.org/10.12688/f1000research.29032.1
Nava S, Beati L, Venzal JM et al (2018) Rhipicephalus sanguineus (Latreille, 1806): neotype designation, morphological re-description of all parasitic stages and molecular characterization. Ticks Tick-Borne Dis 9:1573–1585. https://doi.org/10.1016/j.ttbdis.2018.08.001
Parola P, Paddock CD, Socolovschi C et al (2013) Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev 26:657–702. https://doi.org/10.1128/CMR.00032-13
Petney TN, Otranto D, Dantas-Torres F, Pfäffle MP (2017) Ixodes ventalloi Gil Collado, 1936 (Figs. 64–66). In: Estrada-Peña A, Mihalca AD, Petney TN (eds) Ticks of Europe and North Africa: A Guide to Species Identification. Springer International Publishing, Cham, pp 183–187
Pimentel V, Afonso R, Nunes M et al (2019) Geographic dispersal and genetic diversity of tick-borne phleboviruses (Phenuiviridae, Phlebovirus) as revealed by the analysis of L segment sequences. Ticks and Tick-Borne Diseases 10:942–948. https://doi.org/10.1016/j.ttbdis.2019.05.001
Ramos RAN, Giannelli A, Brianti E et al (2013) Tick vectors of Cercopithifilaria bainae in dogs: Rhipicephalus sanguineus sensu lato versus Ixodes ricinus. Parasitol Res 112:3013–3017. https://doi.org/10.1007/s00436-013-3474-4
Ronquist F, Teslenko M, Van Der Mark P et al (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Roux V, Rydkina E, Eremeeva M, Raoult D (1997) Citrate synthase gene comparison, a new tool for phylogenetic analysis, and its application for the Rickettsiae. Int J Syst Bacteriol 47:252–261. https://doi.org/10.1099/00207713-47-2-252
Rund D, Neves V, CIBIO Research center in biodiversity and genetic resources, CIBIO–Azores, Azores University, Portugal, et al (2019) Molecular survey of Hepatozoon infection of Teira dugesii in the Azores. Anim Biodiv Conserv 42:19–29. https://doi.org/10.32800/abc.2019.42.0019
Santos AS, Santos-Silva MM, Almeida VC et al (2004) Detection of Anaplasma phagocytophilum DNA in Ixodes Ticks (Acari: Ixodidae) from Madeira Island and Setúbal District, Mainland Portugal. Emerg Infect Dis 10:1643–1648. https://doi.org/10.3201/eid1009.040276
Santos AS, de Bruin A, Veloso AR et al (2018) Detection of Anaplasma phagocytophilum, Candidatus Neoehrlichia sp., Coxiella burnetii and Rickettsia spp. in questing ticks from a recreational park. Portugal Ticks and Tick-Borne Diseases 9:1555–1564. https://doi.org/10.1016/j.ttbdis.2018.07.010
Santos AS, Santos-Silva MM (2019) Ixodes ventalloi Gil Collado, 1936: a vector role to be explored. In: Savić S (ed) Vectors and Vector-Borne Zoonotic Diseases. IntechOpen
Simonato G, Franco V, Salvatore G, Zini E et al (2022) First autochthonous clinical case of Hepatozoon silvestris in a domestic cat in Italy with unusual presentation. Parasit Vectors 15:440. https://doi.org/10.1186/s13071-022-05534-x
Šlapeta J, Halliday B, Chandra S et al (2022) Rhipicephalus linnaei (Audouin, 1826) recognised as the “tropical lineage” of the brown dog tick Rhipicephalus sanguineus sensu lato: neotype designation, redescription, and establishment of morphological and molecular reference. Ticks and Tick-Borne Diseases 13:102024. https://doi.org/10.1016/j.ttbdis.2022.102024
Snellgrove AN, Krapiunaya I, Ford SL et al (2020) Vector competence of Rhipicephalus sanguineus sensu stricto for Anaplasma platys. Ticks and Tick-Borne Diseases 11:101517. https://doi.org/10.1016/j.ttbdis.2020.101517
Solano-Gallego L, Trotta M, Carli E et al (2008) Babesia canis canis and Babesia canis vogeli clinicopathological findings and DNA detection by means of PCR-RFLP in blood from Italian dogs suspected of tick-borne disease. Vet Parasitol 157:211–221. https://doi.org/10.1016/j.vetpar.2008.07.024
Sonenshine DE, Roe RM (eds) (2014) Biology of ticks, 2nd edn. Oxford University Press, New York
Stucky BJ (2012) SeqTrace: a graphical tool for rapidly processing DNA sequencing chromatograms. J Biomol Tech 23:90–93. https://doi.org/10.7171/jbt.12-2303-004
Tabar M-D, Altet L, Francino O et al (2008) Vector-borne infections in cats: molecular study in Barcelona area (Spain). Vet Parasitol 151:332–336. https://doi.org/10.1016/j.vetpar.2007.10.019
Uiterwijk M, Vojta L, Šprem N et al (2023) Diversity of Hepatozoon species in wild mammals and ticks in Europe. Parasit Vectors 16:27. https://doi.org/10.1186/s13071-022-05626-8
Acknowledgements
We are grateful to the colleagues who, under MSD Animal Health Portugal coordination, collected all the tick specimens and completed the respective forms. We are also thankful to volunteer students for lab-work assistance that helped us improve our work. Thanks also are due to FCT/MCTES for the financial support to CESAM (UIDP/50017/2020+UIDB/50017/2020) and cE3c (UIDB/00329/2020) through national funds.
Funding
Open access funding provided by FCT|FCCN (b-on). FCT/MCTES for the financial support to CESAM (UIDP/50017/2020 + UIDB/50017/2020) and cE3c (UIDB/00329/2020) through national funds.
Author information
Authors and Affiliations
Contributions
Rosa, F: conceptualization, investigation, methodology, formal analysis, writing—original draft and review; Silva, C: investigation, methodology, formal analysis, and review; Rodrigues, R: methodology and review; Esteves-Vieira, M: methodology and review; Barbosa, I: sample collection and review; Rosa, S: methodology; Dias, D: conceptualization, investigation, methodology, formal analysis, writing, original draft, review, project administration, and funding acquisition; Pina-Martins, F: investigation, methodology, formal analysis, writing—review and editing. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Section Editor: Leonhard Schnittger
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rosa, F., Silva, C., Rodrigues, R. et al. Island hitchhikers: pathogen agents of Madeira and Azores ticks. Parasitol Res 123, 261 (2024). https://doi.org/10.1007/s00436-024-08278-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-024-08278-y