Abstract
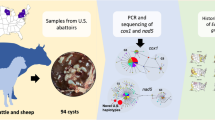
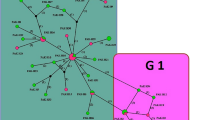
Cystic echinococcosis (CE) is a disease that can be transmitted from animals to humans, caused by the metacestode of Echinococcus granulosus. The disease has significant health and economic impacts worldwide, particularly in endemic areas. The study aimed to evaluate the prevalence of hydatid cysts in ruminants (cattle and sheep) (n = 2060) from the Setif Province of Algeria using microscopy. The results showed that hydatid cysts were detected in 9.6% (198/2060) of ruminants, with a higher prevalence in cattle (16.8%; 56/333) compared to sheep (8.2%; 142/1727). Molecular techniques were used to analyze a subset of animals consisting of 30 sheep and 4 cattle. Specifically, a fragment of the mitochondrial cytochrome c oxidase subunit 1 (mt-CO1) gene was sequenced and compared to sequences from seven humans from the same region. The results indicated that all isolates were identified as E. granulosus sensu stricto. Haplotype analysis identified 19 E. granulosus s.s. haplotypes arranged like a star, with the dominant haplotype (Hap04) at the center. Hap04 has been assigned a total of 17 positives, including positives from sheep, cattle, and two humans. This study is noteworthy for being the first to use a molecular approach to human and ruminant echinococcosis in Setif, a significant breeding region in Algeria.


Similar content being viewed by others
Data availability
No data was used for the research described in the article.
References
Alvarez Rojas CA, Romig T, Lightowlers MW (2014) Echinococcus granulosus sensu lato genotypes infecting humans – review of current knowledge. Int J Parasitol 44(1):9–18. https://doi.org/10.1016/j.ijpara.2013.08.008
Bardonnet K, Benchikh-Elfegoun MC, Bart JM et al (2003) Cystic echinococcosis in Algeria: cattle act as reservoirs of a sheep strain and may contribute to human contamination. Vet Parasitol 116(1):35–44. https://doi.org/10.1016/S0304-4017(03)00255-3
Bir A, Yakhlef H, Madani T (2014) Diversité des exploitations agricoles laitières en zone semi-aride de Sétif (Algérie). Livestock Res Rural Develop 26(2):26. http://www.lrrd.org/lrrd26/2/bir26026.htm
Boufana B, Lahmar S, Rebaï W et al (2014) Genetic variability and haplotypes of Echinococcus isolates from Tunisia. Trans R Soc Trop Med Hygiene 108(11):706–714. https://doi.org/10.1093/trstmh/tru138
Cai H, Guan Y, Ma X et al (2017) Epidemiology of Echinococcosis among schoolchildren in Golog Tibetan Autonomous Prefecture, Qinghai China. Am J Trop Med Hyg 96(3):674–9. https://doi.org/10.4269/ajtmh.16-0479
Casulli A, Interisano M, Sreter T et al (2012) Genetic variability of Echinococcus granulosus sensu stricto in Europe inferred by mitochondrial DNA sequences. Inf Gene Evol 12(2):377–383. https://doi.org/10.1016/j.meegid.2011.12.014
Daryani A, Sharif M, Amouei A, Nasrolahei M (2009) Fertility and viability rates of hydatid cysts in slaughtered animals in the Mazandaran Province, Northern Iran. Trop Anim Health Prod 41:1701–1705. https://doi.org/10.1007/s11250-009-9368-x
El Fegoun MCB, Kohil K, Benguesmia M et al (2020) Cystic Echinococcosis in Algeria: the role of cattle as reservoirs in the dynamics of transmission of Echinococcus granulosus to humans via dogs. Bull Soc Pathol Exot 113(3):130–135. https://doi.org/10.3166/bspe-2020-0130
Issa AR, Arif SH, Mohammed AA et al (2022) Insights into human cystic echinococcosis in the Kurdistan Region, Iraq: characteristics and molecular identification of cysts. Pathogens 11(4):408. https://doi.org/10.3390/pathogens11040408
Joanny G, Cappai MG, Nonnis F et al (2021a) Human cystic echinococcosis in Lebanon: a retrospective study and molecular epidemiology. Acta Parasitol 67(1):186–195. https://doi.org/10.1007/s11686-021-00453-w
Joanny G, Mehmood N, Dessi G et al (2021b) Cystic echinococcosis in sheep and goats of Lebanon. Parasitology 148:871–878. https://doi.org/10.1017/S0031182021000494
Kesik HK, Celik F, Simsek S et al (2021) Molecular characterization and haplotype analyses of lung hydatid cyst isolates of cattle and first report of Echinococcus canadensis (G6/G7) in cattle isolates in Turkey. Acta Parasitol 66(4):1538–1547. https://doi.org/10.1007/s11686-021-00432-1
Khalf MS, AlTaie LH, AlFaham MA (2014) The incidence of hydatid cyst in human in Baghdad governorate. IOSR J Pharm Biol Sci (IOSR-JPBS), 9(3):11–14
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Laatamna AE, Ebi D, Brahimi K et al (2019) Frequency and genetic diversity of Echinococcus granulosus sensu stricto in sheep and cattle from the steppe region of Djelfa. Algeria Parasitol Res 118(1):89–96. https://doi.org/10.1007/s00436-018-6118-x
Leigh JW, Bryant D (2015) POPART: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116
Maillard S, Benchikh-Elfegoun MC, Knapp J et al (2007) Taxonomic position and geographical distribution of the common sheep G1 and camel G6 strains of Echinococcus granulosus in three African countries. Parasitol Res 100:495–503. https://doi.org/10.1007/s00436-006-0286-9
Mehmood N, Arshad M, Ahmed H et al (2020) Comprehensive account on prevalence and characteristics of hydatid cysts in livestock from Pakistan. Korean J Parasitol 58(2):121–127. https://doi.org/10.3347/kjp.2020.58.2.121
Moussa D, Senouci K, Midoun N et al (2021) Genetic diversity of Echinococcus granulosus sensu stricto infecting humans in western Algeria. Parasitol Res 120(9):3195–3202. https://doi.org/10.1007/s00436-021-07223-7
Nakao M, Sako Y, Yokoyama N et al (2000) Mitochondrial genetic code in cestodes. Mol Biochem Parasitol 111:415–424
Oudni-M’rad M, Cabaret J, M’Rad S et al (2016) Genetic relationship between the Echinococcus granulosus sensu stricto cysts located in lung and liver of hosts. Infect Genet Evol 44:356–360. https://doi.org/10.1016/j.meegid.2016.07.024
Piccoli L, Bazzocchi C, Brunetti E et al (2013) Molecular characterization of Echinococcus granulosus in south-eastern Romania: evidence of G1–G3 and G6–G10 complexes in humans. Clin Microbiol Inf 19(6):578–582. https://doi.org/10.1111/j.1469-0691.2012.03993.x
Potapov V, Ong JL (2017) Examining sources of error in PCR by single-molecule sequencing. PLoS ONE 12(1):e0169774. https://doi.org/10.1371/journal.pone.0181128
Romig T, Ebi D, Wassermann M et al (2015) Taxonomy and molecular epidemiology of Echinococcus granulosus sensu lato. Vet Parasitol 213(3–4):76–84. https://doi.org/10.1016/j.vetpar.2015.07.035
Romig T, Deplazes P, Jenkins D et al (2017) Ecology and life cycle patterns of Echinococcus species. Adv Parasitol 95:213–314. https://doi.org/10.1016/bs.apar.2016.11.002
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC et al (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Samari H, Laurimäe T, Reghaissia N et al (2022) Molecular characterization of Echinococcus granulosus sensu lato genotypes in dromedary camels from extreme Sahara of Algeria based on analysis of nad2 and nad5 genetic markers. Acta Trop 234:106616. https://doi.org/10.1016/j.actatropica.2022.106616
Stoore C, Andrade C, Hidalgo C et al (2018) Echinococcus granulosus hydatid cyst location is modified by Fasciola hepatica infection in cattle. Parasit Vectors 11(1):542. https://doi.org/10.1186/s13071-018-3128-6
Tashani OA, Zhang LH, Boufana B et al (2002) Epidemiology and strain characteristics of Echinococcus granulosus in the Benghazi area of eastern Libya. Ann Trop Med Parasitol 96:369–381. https://doi.org/10.1179/000349802125000952
Thompson RA, McManus DP (2002) Towards a taxonomic revision of the genus Echinococcus. Trends Parasitol 18(10):452–457. https://doi.org/10.1016/S1471-4922(02)02358-9
World Health Organization (2015) Investing to overcome the global impact of neglected tropical diseases: third WHO report on neglected diseases 2015. Geneva, Switzerland: World Health Organization
Zait H, Kouidri M, Grenouillet FE et al (2016) Molecular characterization of Echinococcus granulosus sensu stricto and Echinococcus canadensis in humans and livestock from Algeria. Parasitol Res 115(6):2423–2431. https://doi.org/10.1007/s00436-016-4994-5
Acknowledgements
The authors thank the responsible veterinarian at the Setif slaughterhouse and the University Hospital Center of Setif (UHC) for their assistance and participation during the sampling of the material.
Funding
This work was financially supported by Firat University Scientific Research Project Management Unit (Project no: VF.22.25) in Elazig, Turkey.
Author information
Authors and Affiliations
Contributions
Amina Kheninef: investigation, methodology, data analyses, writing-original draft. Figen Celik: investigation, methodology, data analyses. Lynda Aissaoui: methodology, writing-review and editing. Sami Simsek: funding acquisition, conceptualization, supervision, writing-review and editing.
Corresponding author
Ethics declarations
Ethical approval
N/A.
Consent to participate
Informed consent was obtained from the slaughterhouse staff prior to sampling the hydatid cyst samples.
Consent for publication
All authors read and consent to the publication of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kheninef, A., Celik, F., Aissaoui, L. et al. Molecular characterization and haplotypes of hydatid cyst isolates collected from humans and ruminants in Setif Province (northeast of Algeria) based on mitochondrial cytochrome C oxidase subunit 1 (mt-CO1) gene sequences. Parasitol Res 123, 159 (2024). https://doi.org/10.1007/s00436-024-08176-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-024-08176-3