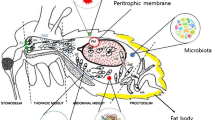
Abstract
Leishmaniasis transmission cycles are maintained and sustained in nature by the complex crosstalk of the Leishmania parasite, sandfly vector, and the mammalian hosts (human, as well as zoonotic reservoirs). Regardless of the vast research on human host-parasite interaction, there persists a substantial knowledge gap on the parasite’s development and modulation in the vector component. This review focuses on some of the intriguing aspects of the Leishmania-sandfly interface, beginning with the uptake of the intracellular amastigotes from an infected host to the development of the parasite within the sandfly’s alimentary canal, followed by the transmission of infective metacyclic stages to another potential host. Upon ingestion of the parasite, the sandfly hosts an intricate repertoire of immune barriers, either to evade the parasite or to ensure its homeostatic coexistence with the vector gut microbiome. Sandfly salivary polypeptides and Leishmania exosomes are co-egested with the parasite inoculum during the infected vector bite. This has been attributed to the modulation of the parasite infection and subsequent clinical manifestation in the host. While human host–based studies strive to develop effective therapeutics, a greater understanding of the vector-parasite-microbiome and human host interactions could help us to identify the targets and to develop strategies for effectively preventing the transmission of leishmaniasis.


Similar content being viewed by others
Data availability
All data included in the manuscript are available.
References
Alvar J, Yactayo S, Bern C (2006) Leishmaniasis and poverty. Trends Parasitol 22:552–557. https://doi.org/10.1016/j.pt.2006.09.004
Alvar J, Vélez ID, Bern C et al (2012) Leishmaniasis worldwide and global estimates of its incidence. PLoS One 7:e35671. https://doi.org/10.1371/journal.pone.0035671
Bahar A, Ren D (2013) Antimicrobial peptides. Pharmaceuticals 6:1543–1575. https://doi.org/10.3390/ph6121543
Bates PA, Depaquit J, Galati EA et al (2015) Recent advances in phlebotomine sand fly research related to leishmaniasis control. Parasites Vectors 8:131. https://doi.org/10.1186/s13071-015-0712-x
Beard CB, Cordon-Rosales C, Durvasula RV (2002) Bacterial symbionts of the Triatominae and their potential use in control of Chagas disease transmission. Annu Rev Entomol 47:123–141. https://doi.org/10.1146/annurev.ento.47.091201.145144
Boulanger N, Lowenberger C, Volf P et al (2004) Characterization of a defensin from the sand fly Phlebotomus duboscqi induced by challenge with bacteria or the protozoan parasite Leishmania major. Infect Immun 72:7140–7146. https://doi.org/10.1128/IAI.72.12.7140-7146.2004
Buchon N, Silverman N, Cherry S (2014) Immunity in Drosophila melanogaster from microbial recognition to whole-organism physiology. Nat Rev Immunol 14:796–810. https://doi.org/10.1038/nri3763
Campolina TB, Villegas LEM, Monteiro CC et al (2020) Tripartite interactions: Leishmania, microbiota and Lutzomyia longipalpis. PLoS Negl Trop Dis 14:e0008666. https://doi.org/10.1371/journal.pntd.0008666
Cecílio P, Cordeiro-da-Silva A, Oliveira F (2022) Sand flies: basic information on the vectors of leishmaniasis and their interactions with Leishmania parasites. Commun Biol 5:305. https://doi.org/10.1038/s42003-022-03240-z
Choe E (2002) Amphetamine increases phosphorylation of extracellular signal-regulated kinase and transcription factors in the rat striatum via group I metabotropic glutamate receptors. Neuropsychopharmacol. https://doi.org/10.1016/S0893-133X(02)00341-X
Claborn D (2010) The biology and control of leishmaniasis vectors. J Global Infect Dis 2:127. https://doi.org/10.4103/0974-777X.62866
Coutinho-Abreu IV, Sharma NK, Robles-Murguia M et al (2010a) Targeting the midgut secreted PpChit1 reduces Leishmania major development in its natural vector, the sand fly Phlebotomus papatasi. PLoS Negl Trop Dis 4:e901. https://doi.org/10.1371/journal.pntd.0000901
Coutinho-Abreu IV, Zhu KY, Ramalho-Ortigao M (2010b) Transgenesis and paratransgenesis to control insect-borne diseases: current status and future challenges. Parasitol Int 59:1–8. https://doi.org/10.1016/j.parint.2009.10.002
Coutinho-Abreu IV, Serafim TD, Meneses C et al (2020) Leishmania infection induces a limited differential gene expression in the sand fly midgut. BMC Genomics 21:608. https://doi.org/10.1186/s12864-020-07025-8
Da Costa-Latgé SG, Bates P, Dillon R, Genta FA (2021) Characterization of glycoside hydrolase families 13 and 31 reveals expansion and diversification of α-amylase genes in the phlebotomine Lutzomyia longipalpis and modulation of sandfly glycosidase activities by Leishmania infection. Front Physiol 12:635633. https://doi.org/10.3389/fphys.2021.635633
Da Silva GD, Iturbe-Ormaetxe I, Martins-da-Silva A et al (2019) Wolbachia introduction into Lutzomyia longipalpis (Diptera: Psychodidae) cell lines and its effects on immune-related gene expression and interaction with Leishmania infantum. Parasites Vectors 12:33. https://doi.org/10.1186/s13071-018-3227-4
De Gregorio E (2002) The Toll and Imd pathways are the major regulators of the immune response in Drosophila. EMBO J 21:2568–2579. https://doi.org/10.1093/emboj/21.11.2568
Díaz-Achirica P, Ubach J, Guinea A et al (1998) The plasma membrane of Leishmania donovani promastigotes is the main target for CA (1–8) M (1–18), a synthetic cecropin A–melittin hybrid peptide. Biochem J 330:453–460. https://doi.org/10.1042/bj3300453
Diaz-Albiter H, Mitford R, Genta FA et al (2011) Reactive oxygen species scavenging by catalase is important for female Lutzomyia longipalpis fecundity and mortality. PLoS One 6:e17486. https://doi.org/10.1371/journal.pone.0017486
Diaz-Albiter H, SantAnna MRV, Genta FA, Dillon RJ (2012) Reactive oxygen species-mediated immunity against Leishmania mexicana and Serratia marcescens in the phlebotomine sand fly Lutzomyia longipalpis. J Bio Chem 287:23995–24003. https://doi.org/10.1074/jbc.M112.376095
Di-Blasi T, Telleria EL, Marques C et al (2019) Lutzomyia longipalpis TGF-β has a role in Leishmania infantum chagasi survival in the vector. Front Cell Infect Microbiol 9:71. https://doi.org/10.3389/fcimb.2019.00071
Dillon RJ, Lane P (1993) Bloodmeal digestion in the midgut of Phlebotomus papatasi and Phlebotomus langeroni. Med Vet Entomol 7:225–232. https://doi.org/10.1111/j.1365-2915.1993.tb00681.x
Dillon RJ, Ivens AC, Churcher C et al (2006) Analysis of ESTs from Lutzomyia longipalpis sand flies and their contribution toward understanding the insect–parasite relationship. Genomics 88:831–840. https://doi.org/10.1016/j.ygeno.2006.06.011
Dostálová A, Volf P (2012) Leishmania development in sand flies: parasite-vector interactions overview. Parasites Vectors 5:276. https://doi.org/10.1186/1756-3305-5-276
Dostálová A, Votýpka J, Favreau AJ et al (2011) The midgut transcriptome of Phlebotomus (Larroussius) perniciosus, a vector of Leishmania infantum: comparison of sugar fed and blood fed sand flies. BMC Genomics 12:223. https://doi.org/10.1186/1471-2164-12-223
EI-Dirany R, Shahrour H, Dirany Z et al (2021) Activity of anti-microbial peptides (Amps) against Leishmania and other parasites: an overview. Biomolecules 11:984. https://doi.org/10.3390/biom11070984
Forestier C-L, Gao Q, Boons G-J (2015) Leishmania lipophosphoglycan: how to establish structure-activity relationships for this highly complex and multifunctional glycoconjugate? Front Cell Infect Microbiol 4. https://doi.org/10.3389/fcimb.2014.00193
Garver LS, Dong Y, Dimopoulos G (2009) Caspar controls resistance to Plasmodium falciparum in diverse anopheline species. PLoS Pathog 5:e1000335. https://doi.org/10.1371/journal.ppat.1000335
Giraud E, Lestinova T, Derrick T et al (2018) Leishmania proteophosphoglycans regurgitated from infected sand flies accelerate dermal wound repair and exacerbate leishmaniasis via insulin-like growth factor 1-dependent signaling. PLoS Pathog 14:e1006794. https://doi.org/10.1371/journal.ppat.1006794
Gossage SM, Rogers ME, Bates PA (2003) Two separate growth phases during the development of Leishmania in sand flies: implications for understanding the life cycle. Int J Parasitol 33:1027–1034. https://doi.org/10.1016/S0020-7519(03)00142-5
Han ZS, Enslen H, Hu X et al (1998) A conserved p38 mitogen-activated protein kinase pathway regulates Drosophila immunity gene expression. Mol Cell Biol 18:3527–3539. https://doi.org/10.1128/MCB.18.6.3527
Hao Z, Kasumba I, Lehane MJ et al (2001) Tsetse immune responses and trypanosome transmission: Implications for the development of tsetse-based strategies to reduce trypanosomiasis. Proc Natl Acad Sci USA 98:12648–12653. https://doi.org/10.1073/pnas.221363798
Hegedus D, Erlandson M, Gillott C et al (2009) New insights into peritrophic matrix synthesis, architecture, and function. Annu Rev Entomol 54:285–302. https://doi.org/10.1146/annurev.ento.54.110807.090559
Hillesland H, Read A, Subhadra B et al (2008) Identification of aerobic gut bacteria from the Kala Azar vector, Phlebotomus argentipes: a platform for potential para-transgenic manipulation of sand flies. Am J Trop Med Hyg 79:881–886
Holzmuller P, Sereno D, Cavaleyra M et al (2002) Nitric oxide-mediated proteasome-dependent oligonucleosomal DNA fragmentation in Leishmania amazonensis amastigotes. Infect Immun 70:3727–3735. https://doi.org/10.1128/IAI.70.7.3727-3735.2002
Hu Y, Aksoy S (2005) An antimicrobial peptide with trypanocidal activity characterized from Glossina morsitans. Insect Biochem Mol Biol 35:105–115. https://doi.org/10.1016/j.ibmb.2004.10.007
Hurwitz I, Hillesland H, Fieck A et al (2011) The paratransgenic sand fly: a platform for control of Leishmania transmission. Parasites Vectors 4:82. https://doi.org/10.1186/1756-3305-4-82
Ilg T, Stierhof Y-D, Craik D et al (1996) Purification and structural characterization of a filamentous, mucin-like proteophosphoglycan secreted by Leishmania parasites. J Biol Chem 271:21583–21596. https://doi.org/10.1074/jbc.271.35.21583
Ilgoutz SC, McConville MJ (2001) Function and assembly of the Leishmania surface coat. Int J Parasitol 31:899–908. https://doi.org/10.1016/S0020-7519(01)00197-7
Jochim RC, Teixeira CR, Laughinghouse A et al (2008) The midgut transcriptome of Lutzomyia longipalpis: comparative analysis of cDNA libraries from sugar-fed, blood-fed, post-digested and Leishmania infantum chagasi-infected sand flies. BMC Genomics 9:15. https://doi.org/10.1186/1471-2164-9-15
Jones CM, Welburn SC (2021) Leishmaniasis beyond East Africa. Front Vet Sci 8:618766. https://doi.org/10.3389/fvets.2021.618766
Kamhawi S, Ramalho-Ortigao M, Pham VM et al (2004) A role for insect galectins in parasite survival. Cell 119:329–341. https://doi.org/10.1016/j.cell.2004.10.009
Karimian F, Koosha M, Choubdar N, Oshaghi MA (2022) Comparative analysis of the gut microbiota of sand fly vectors of zoonotic visceral leishmaniasis (ZVL) in Iran; host-environment interplay shapes diversity. PLoS Negl Trop Dis 16:e0010609. https://doi.org/10.1371/journal.pntd.0010609
Kelly PH, Bahr SM, Serafim TD et al (2017) The gut microbiome of the vector Lutzomyia longipalpis is essential for survival of Leishmania infantum. mBio 8:e01121-16. https://doi.org/10.1128/mBio.01121-16
Kim G-H, Han J-K (2005) JNK and ROK alpha function in the noncanonical Wnt/RhoA signaling pathway to regulate Xenopus convergent extension movements. Dev Dyn 232:958–968. https://doi.org/10.1002/dvdy.20262
Kim M, Lee JH, Lee SY et al (2006) Caspar, a suppressor of antibacterial immunity in Drosophila. Proc Natl Acad Sci USA 103:16358–16363. https://doi.org/10.1073/pnas.0603238103
Kimblin N, Peters N, Debrabant A et al (2008) Quantification of the infectious dose of Leishmania major transmitted to the skin by single sand flies. Proc Natl Acad Sci USA 105:10125–10130. https://doi.org/10.1073/pnas.0802331105
Kleino A, Silverman N (2014) The Drosophila IMD pathway in the activation of the humoral immune response. Dev Comp Immunol 42:25–35. https://doi.org/10.1016/j.dci.2013.05.014
Kraeva N, Horáková E, Kostygov AY et al (2017) Catalase in Leishmaniinae: with me or against me? Infect Genet Evol 50:121–127. https://doi.org/10.1016/j.meegid.2016.06.054
Kulkarni MM, McMaster WR, Kamysz E et al (2006) The major surface-metalloprotease of the parasitic protozoan, Leishmania, protects against antimicrobial peptide-induced apoptotic killing. Mol Microbiol 62:1484–1497. https://doi.org/10.1111/j.1365-2958.2006.05459.x
Kykalová B, Tichá L, Volf P, Loza Telleria E (2021) Phlebotomus papatasi antimicrobial peptides in larvae and females and a gut-specific defensin upregulated by Leishmania major infection. Microorganisms 9:2307. https://doi.org/10.3390/microorganisms9112307
Lehane MJ (1997) Peritrophic matrix structure and function. Annu Rev Entomol 42:525–550. https://doi.org/10.1146/annurev.ento.42.1.525
Lemaitre B, Nicolas E, Michaut L et al (1996) The dorsoventral regulatory gene cassette spätzle/Toll/cactus controls the potent antifungal response in drosophila adults. Cell 86:973–983. https://doi.org/10.1016/S0092-8674(00)80172-5
Li YR, Trush M (2016) Defining ROS in biology and medicine. ROS 1 https://doi.org/10.20455/ros.2016.803
Louradour I, Monteiro CC, Inbar E et al (2017) The midgut microbiota plays an essential role in sand fly vector competence for Leishmania major. Cell Microbiol 19:e12755. https://doi.org/10.1111/cmi.12755
Louradour I, Ghosh K, Inbar E, Sacks DL (2019) CRISPR/Cas9 mutagenesis in Phlebotomus papatasi: the immune deficiency pathway impacts vector competence for Leishmania major. mBio 10:e01941-19. https://doi.org/10.1128/mBio.01941-19
Macleod ET, Darby AC, Maudlin I, Welburn SC (2007) Factors affecting trypanosome maturation in tsetse flies. PLoS One 2:e239. https://doi.org/10.1371/journal.pone.0000239
Maia C, Seblova V, Sadlova J et al (2011) Experimental transmission of Leishmania infantum by two major vectors: a comparison between a viscerotropic and a dermotropic strain. PLoS Negl Trop Dis 5:e1181. https://doi.org/10.1371/journal.pntd.0001181
Maroli M, Feliciangeli MD, Bichaud L et al (2013) Phlebotomine sandflies and the spreading of leishmaniases and other diseases of public health concern. Med Vet Entomol 27:123–147. https://doi.org/10.1111/j.1365-2915.2012.01034.x
Matetovici I, Van Den Abbeele J (2018) Thioester-containing proteins in the tsetse fly (Glossina) and their response to trypanosome infection: thioester-containing proteins in the tsetse fly. Insect Mol Biol 27:414–428. https://doi.org/10.1111/imb.12382
Merida-de-Barros DA, Chaves SP, Belmiro CLR, Wanderley JLM (2018) Leishmaniasis and glycosaminoglycans: a future therapeutic strategy? Parasites Vectors 11:536. https://doi.org/10.1186/s13071-018-2953-y
Mogensen TH (2009) Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev 22:240–273. https://doi.org/10.1128/CMR.00046-08
Moraes CS, Seabra SH, Castro DP et al (2008) Leishmania chagasi interactions with Serratia marcescens: ultrastructural studies, lysis and carbohydrate effects. Exp Parasitol 118:561–568. https://doi.org/10.1016/j.exppara.2007.11.015
Morin-Poulard I, Vincent A, Crozatier M (2013) The Drosophila JAK-STAT pathway in blood cell formation and immunity. JAK-STAT 2:e25700. https://doi.org/10.4161/jkst.25700
Mukhopadhyay D, Dalton JE, Kaye PM, Chatterjee M (2014) Post kala-azar dermal leishmaniasis: an unresolved mystery. Trends Parasitol 30:65–74. https://doi.org/10.1016/j.pt.2013.12.004
Myskova J, Svobodova M, Beverley SM, Volf P (2007) A lipophosphoglycan-independent development of Leishmania in permissive sand flies. Microbes Infect 9:317–324. https://doi.org/10.1016/j.micinf.2006.12.010
Myskova J, Dostálová A, Penickova L et al (2016) Characterization of a midgut mucin-like glycoconjugate of Lutzomyia longipalpis with a potential role in Leishmania attachment. Parasites Vectors 9:413. https://doi.org/10.1186/s13071-016-1695-y
Nimmo DD, Ham PJ, Ward RD, Maingon R (1997) The sandfly Lutzomyia longipalpis shows specific humoral responses to bacterial challenge. Med Vet Entomol 11:324–328. https://doi.org/10.1111/j.1365-2915.1997.tb00417.x
Okwor I, Uzonna J (2016) Social and economic burden of human leishmaniasis. Am J Trop Med Hyg 94:489–493. https://doi.org/10.4269/ajtmh.15-0408
Oliveira F, Jochim RC, Valenzuela JG, Kamhawi S (2009) Sand flies, Leishmania, and transcriptome-borne solutions. Parasitol Int 58:1–5. https://doi.org/10.1016/j.parint.2008.07.004
Pal S, Dolai S, Yadav RK, Adak S (2010) Ascorbate peroxidase from Leishmania major controls the virulence of infective stage of promastigotes by regulating oxidative stress. PLoS One 5:e11271. https://doi.org/10.1371/journal.pone.0011271
Pimenta PFP, Modi GB, Pereira ST et al (1997) A novel role for the peritrophic matrix in protecting Leishmania from the hydrolytic activities of the sand fly midgut. Parasitol 115:359–369. https://doi.org/10.1017/S0031182097001510
Plewes KA, Barr SD, Gedamu L (2003) Iron superoxide dismutases targeted to the glycosomes of Leishmania chagasi are important for survival. Infect Immun 71:5910–5920. https://doi.org/10.1128/IAI.71.10.5910-5920.2003
Pruzinova K, Votypka J, Volf P (2013) The effect of avian blood on Leishmania development in Phlebotomus duboscqi. Parasites Vectors 6:254. https://doi.org/10.1186/1756-3305-6-254
Pruzinova K, Sadlova J, Seblova V et al (2015) Comparison of bloodmeal digestion and the peritrophic matrix in four sand fly species differing in susceptibility to Leishmania donovani. PLoS One 10:e0128203. https://doi.org/10.1371/journal.pone.0128203
Pruzinova K, Sadlova J, Myskova J et al (2018) Leishmania mortality in sand fly blood meal is not species-specific and does not result from direct effect of proteinases. Parasites Vectors 11:37. https://doi.org/10.1186/s13071-018-2613-2
Ramalho-Ortigão J, Temporal P, Oliveira SD et al (2001) Characterization of constitutive and putative differentially expressed mRNAs by means of expressed sequence tags, differential display reverse transcriptase-PCR and randomly amplified polymorphic DNA-PCR from the sand fly vector Lutzomyia longipalpis. Mem Inst Oswaldo Cruz 96:105–111. https://doi.org/10.1590/S0074-02762001000100012
Ramalho-Ortigão M, Jochim RC, Anderson JM et al (2007) Exploring the midgut transcriptome of Phlebotomus papatasi: comparative analysis of expression profiles of sugar-fed, blood-fed and Leishmania major-infected sandflies. BMC Genomics 8:300. https://doi.org/10.1186/1471-2164-8-300
Ramalho-Ortigao M (2010) Sandfly-Leishmania interactions: long relationships are not necessarily easy. TOPARAJ 4:195–204. https://doi.org/10.2174/1874421401004010195
Ranasinghe S, Wickremasinghe R, Munasinghe A et al (2013) Cross-sectional study to assess risk factors for leishmaniasis in an endemic region in Sri Lanka. Am J Trop Med Hyg 89:742–749. https://doi.org/10.4269/ajtmh.12-0640
Rogers ME, Chance ML, Bates PA (2002) The role of promastigote secretory gel in the origin and transmission of the infective stage of Leishmania mexicana by the sandfly Lutzomyia longipalpis. Parasitology 124:495–507. https://doi.org/10.1017/S0031182002001439
Rogers ME, Hajmová M, Joshi MB et al (2008) Leishmania chitinase facilitates colonization of sand fly vectors and enhances transmission to mice. Cell Microbiol 10:1363–1372. https://doi.org/10.1111/j.1462-5822.2008.01132.x
Rogers ME (2012) The role of Leishmania proteophosphoglycans in sand fly transmission and infection of the mammalian host. Front Microbio 3. https://doi.org/10.3389/fmicb.2012.00223
Roque ALR, Jansen AM (2014) Wild and synanthropic reservoirs of Leishmania species in the Americas. Int J Parasitol: Parasites and Wildlife 3:251–262. https://doi.org/10.1016/j.ijppaw.2014.08.004
Sádlová J, Volf P (2009) Peritrophic matrix of Phlebotomus duboscqi and its kinetics during Leishmania major development. Cell Tissue Res 337:313–325. https://doi.org/10.1007/s00441-009-0802-1
Sant’Anna MR, Diaz-Albiter H, Mubaraki M et al (2009) Inhibition of trypsin expression in Lutzomyia longipalpis using RNAi enhances the survival of Leishmania. Parasites Vectors 2:62. https://doi.org/10.1186/1756-3305-2-62
Sant’Anna MR, Diaz-Albiter H, Aguiar-Martins K et al (2014) Colonisation resistance in the sand fly gut: Leishmania protects Lutzomyia longipalpis from bacterial infection. Parasites Vectors 7:329. https://doi.org/10.1186/1756-3305-7-329
Schlein Y, Jacobson RL (1998) Resistance of Phlebotomus papatasi to infection with Leishmania donovani is modulated by components of the infective bloodmeal. Parasitol 117:467–473. https://doi.org/10.1017/S0031182098003321
Schlein Y, Borut S, Greenblatt CL (1987) Development of sandfly forms of Leishmania major in sucrose solutions. J Parasitol 73:797. https://doi.org/10.2307/3282417
Secundino N, Kimblin N, Peters NC et al (2010) Proteophosphoglycan confers resistance of Leishmania major to midgut digestive enzymes induced by blood feeding in vector sand flies: proteophosphoglycan protects L. major. Cell Microbiol 12:906–918. https://doi.org/10.1111/j.1462-5822.2010.01439.x
Sheehan G, Farrell G, Kavanagh K (2020) Immune priming: the secret weapon of the insect world. Virulence 11:238–246. https://doi.org/10.1080/21505594.2020.1731137
Stączek S, Cytryńska M, Zdybicka-Barabas A (2023) Unraveling the role of antimicrobial peptides in insects. IJMS 24:5753. https://doi.org/10.3390/ijms24065753
Sunter J, Gull K (2017) Shape, form, function and Leishmania pathogenicity: from textbook descriptions to biological understanding. Open Biol 7:170165. https://doi.org/10.1098/rsob.170165
Surachetpong W, Singh N, Cheung KW, Luckhart S (2009) MAPK ERK signaling regulates the TGF-β1-dependent mosquito response to Plasmodium falciparum. PLoS Pathog 5:e1000366. https://doi.org/10.1371/journal.ppat.1000366
Suresh R, Mosser DM (2013) Pattern recognition receptors in innate immunity, host defense, and immunopathology. Adv Physiol Educ 37:284–291. https://doi.org/10.1152/advan.00058.2013
Svárovská A, Ant TH, Seblová V et al (2010) Leishmania major glycosylation mutants require phosphoglycans (Lpg2) but not lipophosphoglycan (Lpg1) for survival in permissive sand fly vectors. PLoS Negl Trop Dis 4:e580. https://doi.org/10.1371/journal.pntd.0000580
Telleria EL, Araújo APOD, Secundino NF et al (2010) Trypsin-like serine proteases in Lutzomyia longipalpis – expression, activity and possible modulation by Leishmania infantum chagasi. PLoS One 5:e10697. https://doi.org/10.1371/journal.pone.0010697
Telleria EL, Sant’Anna MRV, Ortigão-Farias JR et al (2012) Caspar-like gene depletion reduces Leishmania infection in sand fly host Lutzomyia longipalpis. J Bio Chem 287:12985–12993. https://doi.org/10.1074/jbc.M111.331561
Telleria EL, Sant’Anna MRV, Alkurbi MO et al (2013) Bacterial feeding, Leishmania infection and distinct infection routes induce differential defensin expression in Lutzomyia longipalpis. Parasites Vectors 6:12. https://doi.org/10.1186/1756-3305-6-12
Telleria EL, Martins-da-Silva A, Tempone AJ, Traub-Csekö YM (2018) Leishmania, microbiota and sand fly immunity. Parasitol 145:1336–1353. https://doi.org/10.1017/S0031182018001014
Telleria EL, Azevedo-Brito DA, Kykalová B et al (2021a) Leishmania infantum infection modulates the JAK-STAT pathway in Lutzomyia longipalpis ll5 embryonic cells and adult females, and affects parasite growth in the sand fly. Front Trop Dis 2:747820. https://doi.org/10.3389/fitd.2021.747820
Telleria EL, Tinoco-Nunes B, Leštinová T et al (2021b) Lutzomyia longipalpis antimicrobial peptides: differential expression during development and potential involvement in vector interaction with microbiota and Leishmania. Microorganisms 9:1271. https://doi.org/10.3390/microorganisms9061271
Tinoco-Nunes B, Telleria EL, Da Silva-Neves M et al (2016) The sandfly Lutzomyia longipalpis LL5 embryonic cell line has active Toll and Imd pathways and shows immune responses to bacteria, yeast and Leishmania. Parasites Vectors 9:222. https://doi.org/10.1186/s13071-016-1507-4
Turco SJ, Descoteaux A (1992) The lipophosphoglycan of Leishmania parasites. Annu Rev Microbiol 46:65–92. https://doi.org/10.1146/annurev.mi.46.100192.000433
Vaidyanathan R (2005) Isolation of a myoinhibitory peptide from Leishmania major (Kinetoplastida: Trypanosomatidae) and its function in the vector sand fly Phlebotomus papatasi (Diptera: Psychodidae). J Med Entomol 42:142–152. https://doi.org/10.1093/jmedent/42.2.142
Valanne S, Wang J-H, Rämet M (2011) The Drosophila toll signaling pathway. J Immunol 186:649–656. https://doi.org/10.4049/jimmunol.1002302
Verma S, Mandal A, Ansari MdY et al (2018) Leishmania donovani inhibitor of serine peptidases 2 mediated inhibition of lectin pathway and upregulation of c5ar signaling promote parasite survival inside host. Front Immunol 9:63. https://doi.org/10.3389/fimmu.2018.00063
Volf P, Myskova J (2007) Sand flies and Leishmania: specific versus permissive vectors. Trends Parasitol 23:91–92. https://doi.org/10.1016/j.pt.2006.12.010
Volf P, Hajmova M, Sadlova J, Votypka J (2004) Blocked stomodeal valve of the insect vector: similar mechanism of transmission in two trypanosomatid models. Int J Parasitol 34:1221–1227. https://doi.org/10.1016/j.ijpara.2004.07.010
Wang J, Wu Y, Yang G, Aksoy S (2009) Interactions between mutualist Wigglesworthia and tsetse peptidoglycan recognition protein (PGRP-LB) influence trypanosome transmission. Proc Natl Acad Sci USA 106:12133–12138. https://doi.org/10.1073/pnas.0901226106
Welchman DP, Aksoy S, Jiggins F, Lemaitre B (2009) Insect immunity: from pattern recognition to symbiont-mediated host defense. Cell Host Microbe 6:107–114. https://doi.org/10.1016/j.chom.2009.07.008
Wilson R, Bates MD, Dostalova A et al (2010) Stage-specific adhesion of Leishmania promastigotes to sand fly midguts assessed using an improved comparative binding assay. PLoS Negl Trop Dis 4:e816. https://doi.org/10.1371/journal.pntd.0000816
World Health Organization (WHO 2023). https://www.who.int/news-room/fact-sheets/detail/leishmaniasis. Accessed 6 June 2023
Yanase R, Moreira-Leite F, Rea E et al (2023) Formation and three-dimensional architecture of Leishmania adhesion in the sand fly vector. eLife 12:e84552. https://doi.org/10.7554/eLife.84552
Yao C, Donelson JE, Wilson ME (2003) The major surface protease (MSP or GP63) of Leishmania sp. Biosynthesis, regulation of expression, and function. Mol Biochem Parasitol 132:1–16. https://doi.org/10.1016/S0166-6851(03)00211-1
Acknowledgements
We would like to extend our gratitude to all the reviewers for their valuable comments and suggestions, which helped us to improve the quality of the manuscript. We also acknowledge ICMR-Vector Control Research Centre for providing infrastructural facilities to execute this review work. AT acknowledges the Senior Research Fellowship (SRF) support by the Indian Council of Medical Research (ICMR).
Funding
This study was partially supported by the Indian Council of Medical Research (ICMR), Government of India (no. Fellowship/129/2022-ECD-II).
Author information
Authors and Affiliations
Contributions
AT searched the literature and drafted the manuscript. NPK corrected and critically reviewed the manuscript. AK critically reviewed the manuscript. PS designed the study, analyzed the data, and corrected the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate and consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Otranto Domenico
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tom, A., Kumar, N.P., Kumar, A. et al. Interactions between Leishmania parasite and sandfly: a review. Parasitol Res 123, 6 (2024). https://doi.org/10.1007/s00436-023-08043-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-023-08043-7