Abstract
This study determines the occurrence and molecular characterisation of Monogenea from three commercially important Australian fish: Australian sardine Sardinops sagax (Jenyns), Australian anchovy Engraulis australis (White), and eastern school whiting Sillago flindersi McKay. Earlier studies have provided only morphological species identification, whereas this study combines both morphological and molecular methods. A total of 247 fish across 3 species, sourced from the New South Wales and Victorian coasts, were examined for Monogenea. A total of 187 monogenean parasites were recovered from the gills. The overall prevalence, mean intensity, and mean abundance were 34%, 2.23, and 0.78, respectively. The parasites were initially classified morphologically as three species across two families. Family Mazocraeidae was represented by Mazocraes australis Timi et al. J Parasitol 85:28–32, 1999, and family Microcotylidae by Polylabris sillaginae (Woolcock, Parasitology 28:79–91, 1936) Dillon, Hargis, and Harrises, 1983 and P. australiensis Hayward, 1996. Molecular identification of parasites was conducted through sequencing of the mitochondrial cytochrome c oxidase subunit 1 (cox1) gene. The fish hosts in the present study were also barcoded (mitochondrial cox1 gene) to confirm specific identities. There was no comparable cox1 sequence available in GenBank for the parasites found in the present study. However, the phylogenetic tree clustered the monogenean species identified in this study according to their familial groups of Mazocraeidae and Microcotylidae. The presence of M. australis on E. australis and S. sagax was confirmed in this study. Polylabris australiensis was only found on S. sagax but Si. flindersi was found to be a host for both Polylabris species. This study is the first to explore the mitochondrial cox1 genes of these three-monogenean species. These findings will serve as a foundation for future monogenean research in Australian waters and elsewhere.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Monogenea are generally ectoparasitic platyhelminths that live on the body surfaces, fins, head, gills, eyes, and oral and branchial cavities depending on the species (Whittington and Chisholm 2008). In general, these worms cause severe damage to the gills due to the invasiveness of suckers, clamps, and hooks at the site of attachment (Hutson et al. 2007; Whittington and Chisholm 2008). Serious pathology and marked pathogenicity leading to the death of fish from Australian waters have previously been reported (Deveney et al. 2001). Approximately, 3500 monogenean species have been described worldwide from marine fish species (Rohde 2005) and a total of at least 316 monogenean species have been identified from various Australian fish hosts (ABRS 2009).
The Australian sardine Sardinops sagax (Jenyns) is a small, schooling pelagic fish of the family Clupeidae (Stewart et al. 2010) which inhabits a depth range of 0–200 m (Froese and Pauly 2019). This species is distributed all along the waters of the continental shelves of Australia and New Zealand (Fletcher 1990; Hoedt and Dimmlich 1995). Sardinops sagax is a planktivorous fish, which as juveniles or young feed on zooplankton such as copepods and as mature fish on phytoplankton (Froese and Pauly 2019). This fish is extensively used as a live bait food for aquaculture fish, pet food as well as human food. Therefore, S. sagax is considered a valuable ‘target fish’ because of its growing market demand (Stewart et al. 2010).
The Australian anchovy Engraulis australis (White) is also a small pelagic schooling fish of the family Engraulidae and widely distributed throughout the temperate waters of the Australian and New Zealand coasts (Arnott and McKinnon 1985; Dimmlich and Ward 2006). This species inhabits a depth range between 31 and 70 m (Froese and Pauly 2019). Juvenile E. australis are mainly found in inlets, bays, and estuaries, whereas the adults are distributed in open coastal waters (Ward et al. 2003). It feeds primarily on small copepods and other zooplankton. Engraulis australis is an important forage fish and is preyed upon by many aquatic predators such as carnivorous fish, squid, dolphins, and seabirds (Froese and Pauly 2019). This fish is economically important because of its extensive use in the fishmeal industry and for human consumption in various parts of the world (Rowling et al. 2010).
Sardinops sagax and E. australis are generally similar in appearance (Dimmlich et al. 2004). These two species can form dense and broad schools and are targeted by Australian commercial fisheries (Hoedt and Dimmlich 1995; Savage and Hobsbawn 2015).
The eastern school whiting Sillago flindersi McKay is a small schooling fish of the family Sillaginidae and found near the seabed, preferring sandy substrates (Froese and Pauly 2019). Sillago flindersi is endemic to Australia and is distributed along subtropical and temperate coastal shelves and estuaries (Froese and Pauly 2019; Gray et al. 2014). This species inhabits a water depth less than 100 m, though the species may be found at depth of 180 m along the eastern and southern Australian coasts (Froese and Pauly 2019; Gray et al. 2014). Juvenile Si. flindersi usually occur in shallow waters. The species feeds on small invertebrates such as polychaetes, crustaceans (amphipods, decapods, mysids), and ichthyofaunas (Day 2010; Froese and Pauly 2019) within the benthic zone.
In Australia, the above-mentioned three fish species are underrepresented in research on monogenean infection (ABRS 2009 and Table 1). No research has ever been conducted on S. sagax in Australia (Table 1) and only a single study conducted by Williams (1988) on E. australis from the Swan River Estuary, Perth, Western Australia (Table 1). Williams (1988) identified a novel mazocraeid monogenean species Pseudanthocotyloides mamaevi Williams, 1988 from E. australis. Extensive studies were undertaken by Dillon et al. (1985a); Dillon et al. (1985b); Hayward (1996a, 1996b); Rohde et al. (1995); Sandars (1945); Williams (1991); Woolcock (1936); Young (1969, 1970) in Australia to identify monogenean infection on multiple sillaginid (whiting) fish species (Table 2) however, Si. flindersi received little attention. For example, Hayward (1996b) and Rohde et al. (1995) performed only two studies on Si. flindersi from the waters of Coffs Harbour, New South Wales (NSW), and Lakes Entrance, Victoria, Australia, and yielded a single monogenean species, Polylabris sillaginae (Woolcock, 1936) Dillon, Hargis, and Harrises, 1983 (Microcotylidae), and an unspecified Microcotylidae species, respectively (Table 1).
Globally, earlier monogenean researches on the three-fish species have used morphological methods only to identify the worms. Previous morphological species identification has created challenges in the accurate identification of Monogenea from fish in Australian waters Rohde (1989b); Rohde and Watson (1985a, 1985b). As a result, the names of the Monogenea genera and species have been changed, revised, and amended multiple times. Previous researchers, Hayward (1996a, 1996b); Rohde (1989b); Rohde and Watson (1985a, 1985b); Williams (1991) concluded that careful consideration should be given before naming and revising monogenean species within the families Mazocraeidae and Microcotylidae based on low morphological variations. There have been no studies in Australia and elsewhere that have used a combination of morphological and molecular methods to classify and describe monogenean species from S. sagax, E. australis, and Si. flindersi. As a result, specific identification of monogenean species from these hosts using a combined morphological and molecular tool is warranted.
The present study aimed to host record investigation of monogenean species from S. sagax, E. australis, and Si. flindersi and to characterise the species genetically based on partial mitochondrial cox1 gene to validate their taxonomic and geographic status.
Materials and methods
Fish collection
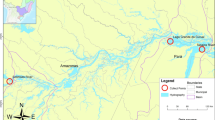
Three fish species, Australian sardine S. sagax (n = 55), Australian anchovy E. australis (n = 70), and eastern school whiting Si. flindersi (n = 122), were purchased from two retail fish markets in Australia. The fish had been caught from two localities, off the coast of NSW and Victoria, Australia. The details of the fish sampling and examination are provided in Table 3. Fish were transported on ice in an insulated box to the Parasitology Laboratory of Charles Sturt University, Wagga Wagga Campus, Australia. Fish from each batch were examined on the day of arrival at the University. The morphological identification of host fish was confirmed using the keys provided by Gommon et al. (2008).
Parasite collection
Individual fish were examined externally for the presence of monogenean and then dissected to remove the gills. The gills were placed in an individual Petri dish containing saline water (35 g of salt in 1000 ml of water). The surfaces of all gills were thoroughly examined under a stereomicroscope (Leica EZ4, China) for the presence of Monogenea. A total of 187 worms belonging to Mazocraes australis Timi, Sardella & Etchegoin, 1999 of family Mazocraeidae and Polylabris sillaginae and Polylabris australiensis Hayward, 1996 of family Microcotylidae were collected from the examined fish. All parasites were recovered from the gills using fine dissection needles with none observed on the external surface of the fish. Monogenea were washed in saline water, counted, and preserved in 70% ethanol for further morphological and molecular analyses.
The overall prevalence, mean intensity, and mean abundance were 34%, 2.23, and 0.78, respectively. The prevalence, intensity, and abundance of infection of Monogenea were highest in the fish sourced from off the coast of NSW. Among the three Monogenea identified, M. australis had the highest overall prevalence and mean intensity at 63% and 2.64, respectively from E. australis. Infection with M. australis on S. sagax was much lower and the prevalence and mean intensity at 21% and 1, respectively. The second most abundant species was P. sillaginae infecting Si. flindersi from the NSW coast, having the highest overall prevalence and mean intensity, at 55% and 1.93, respectively. Infections of P. sillaginae from Si. flindersi from the Victorian coast was much lower at prevalence and mean intensity of 3% and 1, respectively. Polylabris australiensis, although found on two host species (S. sagax and Si. flindersi), in NSW and Victoria, was the least prevalent monogenean species found in this study. Table 3 shows the infection data of Monogenea identified from the three species of fish in the present study.
Morphological examination
Mature Monogenea which were not contracted, broken, folded, or twisted were selected for morphological examination. Handling and processing of specimens were carried out according to Gussev (1973, 1985). Initial morphological analyses were conducted using a compound microscope (Upright Motorized Microscope ECLIPSE Ni-E, Nikon, Japan) fitted with a computer screen. Monogenea were initially grouped based on their key morphological traits such as body shape and size; morphology and morphometry of the sucker, haptor, male copulatory organ, and genital atrium; number and organisation of clamp; shape, size, and the number of hamuli according to Agarwal (1988); Dillon et al. (1985a); Gupta and Krishna (1988); Hayward (1996b); Mamaev (1982); Sailaja et al. (2019); Timi et al. (1999); Williams (1991); Woolcock (1936). The characteristics of systematic importance were measured directly with an eyepiece micrometre (BX‐43 Olympus Microscope, Olympus Corporation, Japan). All measurements are in micrometres and are given as the range, followed by the mean in parentheses. A dash (–) indicates that measurements could not be made. The prevalence, mean intensity, and mean abundance of the monogeneans were determined according to Bush et al. (1997).
Molecular barcoding of host and parasite
A small piece of the host’s muscle tissue and a small piece from each parasite (the same specimens that were used for morphology as described above) was transferred into separate 1.5 ml autoclaved Eppendorf tubes for molecular study. The remaining anterior and posterior regions of the parasites were processed for microscopy and morphological study. DNA was extracted using DNeasy Blood and Tissue Kits (Qiagen, Hilden, Germany), as per the manufacturer’s instructions, and modified (Shamsi et al. 2017) to be eluted in 40 µl of elution buffer. Polymerase chain reaction (PCR) amplification of the fragment of the mitochondrial cox1 gene of both hosts and parasite was carried out using the following primer sets. For fish, FishF1 (forward: 5′-TCA ACC AAC CAC AAA GAC ATT GGC AC-3′) and FishR1 (reverse: 5′-TAG ACT TCT GGG TGG CCA AAG AAT CA -3′) were used and for Monogenea, COI-ASmit1 (forward: 5′-TTT TTT GGG CAT CCT GAG GTT TAT-3′) and COI-ASmit2 (reverse: 5′-TAA AGA AAG AAC ATA ATG AAA ATG-3′) were used (Littlewood et al. 1997; Ward et al. 2005). The cycling conditions to amplify the host’s mitochondrial gene was initial 95 °C for 2 min, followed by 35 cycles of denaturation at 95 °C for 30 s, annealing at 54 °C for 30 s, extension at 72 °C for 1 min, and final extension at 72 °C for 10 min. The mitochondrial gene of Monogenea was amplified according to the protocol described in Hossen et al. (2020). An aliquot (3 µl) of each amplicon was examined on a 1.5% w/v agarose gel, stained with GelRed™, and photographed using a gel documentation system.
Representative samples from hosts and parasites were sent to the Australian Genome Research Facility (AGRF), Queensland, Australia, and were subjected to Sanger sequencing using the same primer sets as for PCR. Sequence data including chromatograms were observed initially through Sequence Scanner Software (Applied Biosystems® Genetic Analysers). The sequences were compared with the GenBank database content with BLAST and deposited in GenBank under accession numbers of hosts and parasites, respectively (Table 4). The evolutionary (pairwise) genetic distance was calculated using MEGA v. 10 (Kumar et al. 2016).
Phylogenetic analysis
The phylogenetic tree was constructed from the sequences obtained in this study for the mitochondrial cox1 region along with available GenBank sequences (Table 4). All available sequences in GenBank for the species belonging to the families Mazocraeidae and Microcotylidae were included during the construction of the phylogenetic tree. All sequences were then aligned with MUSCLE in MEGA v. 10 (Kumar et al. 2016) and manually adjusted. The phylogenetic relationships among the species were inferred using the Bayesian method (MrBayes v 3.2) (Ronquist and Huelsenbeck 2003). The General Time Reversible with Gamma distributed (GTR + G) model was used during Bayesian phylogenetic analysis. This model was proposed by jModelTest v.2.1.4 under Bayesian Information Criterion (BIC) as the best-fit model of nucleotide substitution (Darriba et al. 2012). Choricotyle australiensis Roubal, Armitage and Rohde, 1983 (sequence ID: MT783686) identified from Australian waters was used as the outgroup based on close as possible to the species identified (ingroup) in this study (Hossen et al. 2020). The sampling frequency was set at 1000 and the run length continued for 2,000,000 generations until the p value reached less than 0.01. After the Markov Chain Monte Carlo (MCMC) run, the first 30% samples were excluded through the ‘burnin’ command. The ‘sumt’ command was executed to summarise the tree. The phylogenetic tree was visualised in FigTree v 1.4.3 (Rambaut 2014).
Results
Molecular identification of fish
The preliminary identification of the host species using morphological characteristics was confirmed by the sequencing of mitochondrial cytochrome c oxidase subunit I gene (cox1). A search in GenBank for the representative sequence generated for S. sagax (sequence ID: MZ274053–54) in this study showed 100% similarity with S. sagax (sequence ID: DQ107708) identified from the Australian waters (Ward et al. 2005), thus confirming the host’s taxonomic status. The sequences obtained for Si. flindersi (sequence ID: MZ274055–56) showed 99% similarity (654/655 with no nucleotide gap) with Si. flindersi (sequence ID: EF609468) identified from the Australian waters (Ward and Holmes 2007). In the present study, the exploration of genetic data for E. australis was unsuccessful due to the poor quality of the chromatograms.
Morphological identification of Monogenea
Microscopic examination (which included morphological, morphometric, and meristic data analyses) revealed three species of Monogenea, Mazocraes australis of family Mazocraeidae, and Polylabris sillaginae and Polylabris australiensis of family Microcotylidae.
The morphology and measurements of the monogenean species recovered in this study matched with earlier descriptions given by Dillon et al. (1985a); Hayward (1996b); Timi et al. (1999); Williams (1991); Woolcock (1936) in studies of fish from Australian waters and elsewhere (Tables 5 and 6).
Mazocraeid monogenean
Mazocraes australis
Based on 15 whole-mount specimens, NSW waters (Table 5). Body elongated, fusiform, or lanceolate. Maximum width near middle and tapering to narrow anterior and posterior region at haptor. Buccal suckers separated. Oesophagus oval. The genital atrium mazocraeid-type and armed with one pair of large lateral hooks and 12–14 small median hooks. Genital hooks organised in two transverse semicircular rows. Follicular vitellaria extending from level of the genital atrium to beginning of peduncle. No vitellaria observed in peduncle and haptor. Haptor well separated from body proper by short peduncle. Haptor heart-shaped and containing eight clamps. Clamps arranged in two opposing rows of four each side. Distance between clamp rows decreased towards posterior region of haptor. Clamps similar in structure. Two anterior pairs larger than remaining posterior pairs in some specimens. Clamp description similar to Timi et al. (1999). Short terminal lappet containing one pair of hamuli and two pairs of marginals. Hamuli strong and stout with hook and moderate ridged shaft. Mature specimens containing spindle-shaped eggs.
Microcotylid Monogenea
Polylabris sillaginae
Based on 15 specimens, NSW waters (Table 6). Body elongated, lanceolate, or fusiform. Two distinct buccal suckers containing sclerotized, tooth-like papillae on rims. Maximum width observed near middle of the body and tapering to narrow posterior region containing clamps. Symmetrical haptor not well separated from body proper. Pharynx and oesophageal diverticula present. Gut bifurcating at or immediately behind the male copulatory organ. Worm bivaginated. Male copulatory organ sclerotised, short, conical, and containing inner tube and outer covering. In relaxed specimens, haptor appears much longer. Clamps organised in two nearly equal ventrolateral rows bearing up to 27–40 pairs in each row. Clamps structurally similar along rows, but differ in size depending on the location, such as anterior, middle, and posterior. Clamp’s description very similar to Dillon et al. (1985a); Woolcock (1936). Vitellaria extends up to intestinal crura.
Polylabris australiensis
Based on five specimens, NSW and Victorian waters (Table 6). Body morphology similar to P. sillaginae, with an exception in structure of the male copulatory organ. In P. australiensis, the copulatory organ comparatively larger and highly sclerotised. Continuous haptor containing 20–35 pairs of clamps observed in elongated and relaxed specimens.
Molecular characterisation of Monogenea
A total of 16 worms, representing each of the three-monogenean species, were sequenced for cox1 gene.
Mazocraeid Monogenean
Five worms (voucher numbers 38, 60, 61, 67, and 127) belonging to M. australis, from NSW S. sagax and E. australis fish were subjected to sequencing. The genetic sequence for M. australis from S. sagax (voucher number 38) was failed. The cox1 sequences produced for M. australis from E. australis were 396 bp long and identical. Mazocraes species has no cox1 sequence data deposited in GenBank that is comparable. As a result, no genetic similarity index was shown for the present specimens. The sequences obtained in this study were deposited in GenBank under the accession numbers (sequence ID: MZ273894–97).
Microcotylid Monogenea
Eight specimens (voucher numbers 41, 211, 212, 213, 483, 485, 488, and 490) belonging to P. sillaginae, recovered from NSW Si. flindersi were subjected to sequencing. The length of the cox1 sequences generated for the specimens was 396 bp and identical. Three Monogenea (voucher numbers 53, 404, and 409) belonging to P. australiensis, obtained from NSW Si. flindersi (53) and Victorian S. sagax (404 and 409) were subjected to sequencing. The length of the cox1 sequences explored for the specimens was 396 bp and identical.
A single Polylabris species, P. halichoeres Wang and Zhang, 1998 sequence (for the complete mitochondrial gene) is available in GenBank (sequence ID: JF505509) from an unpublished study (Table 4). A nucleotide BLAST search in GenBank for one of the representative sequences obtained from P. sillaginae (sequence ID: MZ273898–MZ273905) showed 84% similarity with P. halichoeres (sequence ID: JF505509). The sequences obtained for P. australiensis in this study did not reveal any similarity with the P. halichoeres. The sequences obtained for P. australiensis in this study were deposited in GenBank under the sequence ID MZ273906–08.
Phylogenetic analyses
Bayesian Phylogenetic tree grouped mazocraeid and microcotylid worms separately. Mazocraes australis clustered with closely related mazocraeid species. The sequences obtained in this study for P. sillaginae and P. australiensis clustered with closely related microcotylid species. The sequences generated in this study for P. sillaginae and P. australiensis grouped according to their species with a 100% posterior probability value. The phylogenetic relationship of Monogenea found in this study is shown in Fig. 1.
The phylogenetic relationship of Monogenea found in this study and those closely related species available in GenBank (Table 4 for details). The tree has been constructed using Bayesian method. *indicates the cox1 sequences generated in the present study. Bayesian posterior probability values more than 90% were shown on the node
Discussion
The present study provided for the first-time exploration of cox1 sequences of three monogenean species, M. australis, P. sillaginae, and P. australiensis, infecting three Australian commercial fish species. The occurrence of mazocraeid monogenean M. australis in two Australian hosts S. sagax and E. australis is new geographical and host records. The present research also identified two microcotylid Monogenea, P. sillaginae, and P. australiensis, from two hosts S. sagax and Si. flindersi; Polylabris sillaginae was found in Si. flindersi, while P. australiensis was found in both host species. In this work, the fish species S. sagax was discovered to be a novel host for the monogenean P. australiensis. A single earlier study identified P. sillaginae from Si. flindersi (Hayward 1996b); however, the worm P. australiensis was identified for the first-time infecting Si. flindersi in this study.
The highest overall prevalence, mean intensity, and mean abundance of monogenean infection were observed in E. australis compared to the remaining two hosts S. sagax and Si. flindersi examined in this study. The monogenean, M. australis had the highest overall occurrence and infection (prevalence: 63% and mean intensity: 2.64 (1–8)) and infected two species E. australis and S. sagax (Table 3). Timi et al. (1999) observed the prevalence and mean intensity of M. australis from Argentine anchovy E. anchoita Hubbs and Marini at 2.83% and 1.56 (1–9), respectively. The result of the occurrence of M. australis in the present study was appreciably higher in Australian waters than observed by Timi et al. (1999) from Argentine waters. There has been no Australian study in which to compare the findings of the present study.
The monogenean, Polylabris sillaginae recovered from the Si. flindersi was found to be the second most common species in this study, with a prevalence of 55% and a range in infected fish up to 5. Earlier Australian studies observed P. sillaginae infection in Si. flindersi however, no infection datum was provided (Hayward 1996b). The infection data of P. sillaginae found in this study agree with a previous Australian study conducted by Williams (1991) on a sillaginid host, the yellowfin whiting Si. schomburgkii Peters. The Williams (1991) study found an infection prevalence of 48% and a range in infected host of 7. The least common monogenean species found in this study was P. australiensis. Similar findings were seen for P. australiensis in two previous parasitological studies conducted in Australian waters (Hayward 1996b; Williams 1991), though hosts were different (golden-lined sillago Si. analis Whitely, and Si. schomburgkii).
Mazocraes species
The genus Mazocraes Hermann, 1782 was first proposed by Hermann in 1782 and was later revised by Mamaev (1982). To date, around 14 valid Mazocraes species have been identified globally (Table 7). The morphological features, which included morphometric and meristic data of Mazocraes monogenean found in the present study, were compared with the valid Mazocraes species. Our data showed a complete or partial match with the M. australis only, and Table 5 demonstrates the comparative measurements.
There have been three publications only, which examined Mazocraes species genetically and of the 14-valid species globally 12 were identified morphologically. For example, M. alosae Hermann, 1782 has seven sequences deposited in GenBank for the nuclear genes only, one sequence for the 18S gene (Bao et al. 2015), and six sequences for the 28S genes (Schade et al. 2016). However, Bao et al. (2015) did not provide any morphological description of M. alosae.
A single species M. bengalensis Sailaja, Shameem & Madhavi, 2019 has molecular data in GenBank (single sequence deposited for 28S gene) along with a morphological description (Sailaja et al. 2019). However, no cox1 data for any Mazocraes species is available in GenBank. A nucleotide search in GenBank for one of the four sequences generated in this study showed 79% similarity (306/389; inclusive of 12 nucleotides gap) with another mazocraeid species, Neomazocraes dorosomatis (Yamaguti, 1938) Price, 1943 (Sequence ID: JQ038229) from an unpublished study. Therefore, the present study was the first to explore the mitochondrial cox1 gene sequences of this monogenean M. australis. The phylogenetic tree separated the sequences generated in the present study from the existing mazocraeid species. The strong posterior probability value demonstrated in this study supports the taxonomic status of M. australis identification in Australian waters.
The morphological species identification is often difficult, particularly when dealing with small mazocraeid species (Rohde 1989a, b; Rohde and Watson 1985a, b). The following authors concluded that if populations of Monogenea from the same host species or genus occur in different geographical areas, they are likely to be conspecific and should not be classified as different species if they are just slightly different from one another (Rohde 1989a, b; Rohde and Watson 1985a, b). Therefore, seven previously identified Mazocraes species are now considered as ‘species inquirendae’ (Sailaja et al. 2019). Further genetic analyses are required to verify the taxonomic status of morphologically identified all Mazocraes species.
Polylabris species
Polylabris Euzet and Cauwet, 1967 species are distinguished from other microcotylids by the presence of a single male copulatory organ, which is sclerotised and typically conical (Hayward 1996b). The taxonomic status of species within the genus Polylabris is uncertain. Williams (1991) found morphological inter-species variations which had been considered as species novel. As a result, Hayward (1996b) thoroughly revised the genus ‘with the key to species Polylabris’ and recognised 17 valid species, as well as three more ‘species inquirendae’ that infected the gills of several Perciformes fish. The morphometric and meristic data of our Polylabris worms partially or completely matched with two species, P. sillaginae and P. australiensis according to the key and diagnostic features provided by Dillon et al. (1985a); Hayward (1996b); Sandars (1945); Williams (1991); Woolcock (1936); Young (1970). The comparative measurements of Polylabris specimens are provided in Table 6.
According to Hayward (1996b) and Williams (1991), the taxonomic position of P. sillaginae is complicated. Polylabris sillaginae was first identified and described from the King George whiting Sillaginodes punctatus (Cuvier) sourced from Victorian waters under the name of Microcotyle sillaginae Woolcock, 1936. This worm was later recorded from the same host in Western Australian waters and named Mi. parasillaginae Sandars, 1945. However, Williams (1991) synonymised the above-mentioned two Microcotyle Monogenea as P. sillaginae based on their general body forms. In a parasitic study, Dillon et al. (1985a) also identified P. sillaginae from the above-mentioned sillaginid host in South Australia and Western Australia. Polylabris sillaginae has also been identified from another sillaginid host, Si. schomburgkii in Western Australian waters (Williams 1991). However, five of the P. sillaginae species identified by Williams (1991) were reidentified as P. australiensis (Hayward 1996b). Williams (1991) identified P. sandarsae Williams, 1991 from the trumpeter whiting Si. maculata Quoy and Gaimard which was described as a novel species based on a few morphological variations (male copulatory organ and testes (shape, size, and number)). Williams (1991) also described Polylabris sp. 1 and Polylabris sp. 2 from the Sillaginodes punctatus and southern school whiting Si. bassensis Cuvier, respectively. The names of P. sandarsae, as well as Polylabris sp. 1 and 2, were later updated and merged into a single species P. sillaginae (Hayward 1996b). Polylabris sillaginae has previously been found in ten sillaginid hosts in Australia, New Caledonia, and the Gulf of Thailand, with Sillaginodes punctatus being the type host (Hayward 1996b). However, in this study, Si. flindersi was identified as the host for P. sillaginae. For a more comprehensive understanding of P. sillaginae, a greater sample size of sillaginid hosts should be examined. Of particular importance is clarification of the specificity of hosts, which according to the findings of the present study requires further investigation.
The general understanding is that monogeneans exhibit high host-specificity and the identification of Polylabris australiensis infecting S. sagax is an unusual finding of the present study. The name P. australiensis was first proposed by Hayward (1996b). So far, this monogenean was identified from two sillaginid fish species, Si. analis and Si. schomburgkii in Australia (Hayward 1996b). According to Hayward (1996b), earlier morphological characteristic of the species was provided by Williams (1991) under the species name ‘P. sillaginae’ identified from the Si. schomburgkii in Australia. Williams (1991) observed some morphological differences in some organs such as the size and morphology of male copulatory organs, number of testes, and clamps. Williams (1991) concluded that the morphological discrepancies within the identified P. sillaginae ‘may have been due to inaccuracies of observation and measurement’ and was ‘insufficient to separate the worm’. However, there is no further record of identification of this monogenean species in Australia or elsewhere. The present study confirmed the identification of P. australiensis from two new hosts S. sagax and Si. flindersi in Australian waters. In the present study, the occurrence of Polylabris species in S. sagax (Clupeiformes: Clupeidae) is uncommon and further research is required to elucidate the reasons for this.
Hayward (1996b) stated that the morphological plasticity of the species belongs to the genus Polylabris is high and molecular characterisation would solve the problem of accurate species identification. Polylabris species has 11 sequences deposited in GenBank. Nine sequences are available for the nuclear genes and only two sequences for the mitochondrial gene. In particular, P. halichoeres has two sequences for the complete mitochondrial gene (Li et al. 2011). Polylabris sillaginae has one sequence for the nuclear 28S gene (Catalano et al. 2010) and P. heterodus (Lebedev and Parukhin, 1969) Mollaret, Jamieson & Justine, 2000 has one sequence for the 28S (Mollaret et al. 2000). Polylabris acanthopagri Mamaev and Parukhin, 1976 has one sequence for 18S, P. bengalensis has one sequence for 18S, P. cf. mamaevi Ogawa and Egusa, 1980 has three sequences for 28S, and an unidentified species Polylabris sp. has one 18S and one 28S sequence available in GenBank as a direct submission. However, none of the Polylabris species sequences available in GenBank have a morphological description in the publication. A nucleotide search in GenBank for one of the representative Polylabris sequences produced in this study revealed 86% similarity with another microcotylid species Polylabroides guangdongensis Zhang and Yang, 2000 (Sequence ID: JQ038230) from an unpublished study. Our specimens, however, do not have the similar morphology as Polylabroides. The pairwise genetic comparison of the Polylabris sequences explored in this study showed 0–10.14% nucleotide variation, which was interpreted as an interspecific variation and confirmed as P. sillaginae and P. australiensis. The phylogenetic tree divided the cox1 sequences generated in this study into two clusters, one for P. sillaginae and another for P. australienis with a 100% posterior probability value. The tree also separated the Polylabris sequences from the existing microcotylid Monogenea sequences with a strong posterior probability value and confirmed the taxonomic status P. sillaginae and P. australienis (Fig. 1). Further genetic studies are required to confirm the taxonomic status of all Polylabris species that have been morphologically identified and described.
Conclusion
In the present study, three monogenean species M. australis, P. sillaginae, and P. australiensis were identified using morphological and molecular tools. Mazocraes australis had the highest overall prevalence, intensity, and abundance among the identified worms. A new host record was established in this study for two monogenean species M. australis and P. australiensis. The exploration of cox1 genetic sequences of these monogenean species are novel in this study. This research has highlighted that populations of Monogenea from the same host genera in different geographic areas are likely to be conspecific and should not be considered as novel species unless both morphological and genetic analyses are performed. To better understand and confirm the taxonomic status as well as geographical distribution of all Mazocraes and Polylabris species, a broader host examination is required to collect monogenean species and to identify them using combined morphological and molecular analyses.
Data availability
The sequences generated for the mitochondrial cytochrome c oxidase subunit 1 (cox1) gene in this study have been deposited in GenBank database under the accession number MZ273894–97 for Mazocraes australis, MZ273898–MZ273905 for Polylabris sillaginae, and MZ273906–08 for Polylabris australiensis, MZ274053–54 for Sardinops sagax, MZ274055–56 for Sillago flindersi.
Code availability
Not applicable.
References
ABRS (2009) Australian Biological Resources Study: Australian Faunal Directory. Available from https://biodiversity.org.au/afd/taxa/MONOGENEA (accessed on 20 April 2021)
Agarwal N (1988) A new monogenean Mazocraes mamaevi sp. nov. from a fresh water cyprinid fish, Labeo Rohita (Ham.) from Lucknow. Indian J Parasitol 12:327–329
Agrawal N, Sharma R (1989) Two new species of the genus Mazocraes Hermann, 1782 (Monogenea) from Hilsa ilisha (Ham.) from India. Helminthologia 26:187–194
Arnott GH, McKinnon AD (1985) Distribution and abundance of eggs of the anchovy, Engraulis australis antipodum Gunther, in relation to temperature and salinity in the Gippsland Lakes. Mar Freshw Res 36:433–439
Ayadi ZEM, Gey D, Justine J-L, Tazerouti F (2017) A new species of Microcotyle (monogenea: Microcotylidae) from Scorpaena notata (Teleostei: Scorpaenidae) in the Mediterranean Sea. Parasitol Int 66:37–42
Bao M, Roura A, Mota M, Nachón DJ, Antunes C, Cobo F, MacKenzie K, Pascual S (2015) Macroparasites of allis shad (Alosa alosa) and twaite shad (Alosa fallax) of the Western Iberian Peninsula Rivers: ecological, phylogenetic and zoonotic insights. Parasitol Res 114:3721–3739
Bouguerche C, Gey D, Justine J-L, Tazerouti F (2019a) Microcotyle visa n. sp. (Monogenea: Microcotylidae), a gill parasite of Pagrus caeruleostictus (Valenciennes) (Teleostei: Sparidae) off the Algerian coast. Western Mediterranean Syst Parasitol 96:131–147
Bouguerche C, Gey D, Justine J-L, Tazerouti F (2019b) Towards the resolution of the Microcotyle erythrini species complex: description of Microcotyle isyebi n. sp. (Monogenea, Microcotylidae) from Boops boops (Teleostei, Sparidae) off the Algerian coast. Parasitol Res 118:1417–1428
Bouguerche C, Tazerouti F, Gey D, Justine J-L (2019c) Redescription and molecular characterisation of Allogastrocotyle bivaginalis Nasir & Fuentes Zambrano, 1983 (Monogenea: Gastrocotylidae) from Trachurus picturatus (Bowdich) (Perciformes: Carangidae) off the Algerian coast, Mediterranean Sea. Syst Parasitol 96:681–694
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms. J Parasitol 83:575–583
Catalano SR, Hutson KS, Ratcliff RM, Whittington ID (2010) Redescriptions of two species of microcotylid monogeneans from three arripid hosts in southern Australian waters. Syst Parasitol 76:211–222
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
Day J (2010) School Whiting (Sillago flindersi): Exploration of fixed projected catches and a retrospective look at variability in recruitment estimates. Commonwealth Scientific and Industrial Research Organisation (CSIRO) Marine and Atmospheric Research, Hobart, Tasmania, 1–45 pp
Deveney M, Chisholm L, Whittington ID (2001) First published record of the pathogenic monogenean parasite Neobenedenia melleni (Capsalidae) from Australia. Dis Aquat Org 46:79–82
Dillon WA, Hargis WJ Jr, Harrises AE (1985) Monogeneans from the southern Pacific Ocean, Polyopisthocotyleids from the Australian fishes The subfamilies Polylabrinae and Microcotylinae. Trans Ser 31 Zool J 62:821–828
Dillon WA, Hargis WJ, Harrises AE (1985) Monogeneans from the southern Pacific Ocean Polyopisthocotyleids from Australian fishes The subfamily Microcotylinae. Trans Ser 32 Zool J 63:348–359
Dimmlich WF, Ward TM (2006) Ontogenetic shifts in the distribution and reproductive patterns of Australian anchovy (Engraulis australis) determined by otolith microstructure analysis. Mar Freshwat Res 57:373–381
Dimmlich WF, Breed WG, Geddes M, Ward TM (2004) Relative importance of gulf and shelf waters for spawning and recruitment of Australian anchovy, Engraulis australis, in South Australia. Fish Oceanogr 13:310–323
Fletcher WJ (1990) A synopsis of the biology and the exploitation of the Australian pilchard, Sardinops neopilchardus (Steindacher). Part 1. Biology, Fisheries Department of Western Australia. Fish Res Rep No 88:1–45
Froese R, Pauly D (2019) World Wide Web electronic publication. www.fishbase.org ( accessed on 02 February 2019)
Gommon M, Bray D, Kuiter R (eds) (2008) Fishes of Australia’s Southern Coast. New Holland Publisher, Australia
Gray CA, Barnes LM, Ochwada-Doyle FA, van der Meulen DE, Kendall BW, Robbins WD (2014) Age, growth and demographic characteristics of Sillago flindersi exploited in a multi-species trawl fishery. Fish Sci 80:915–924
Gupta V, Krishna RK (1988) On four new species of monogenetic trematodes of the family Mazocraeidae price, 1936 from fishes of India. Indian J Helminthol 40:32–43
Gussev AV (1973) Revision of the genus Protogyrodactylus (Monogenoidea) and a description of the new species P. marinus. Folia Parasitol 20:17–27
Gussev AV (1985) Parasitic metazoans: Class Monogenea. In: Bauer ON (ed) Key to the parasites of freshwater fish fauna of the USSR, Nauka, Leningrad 2:1–424 (In Russian)
Hayward CJ (1996a) Revision of diplectanid monogeneans (Monopisthocotylea, Diplectanidae) in sillaginid fishes, with a description of a new species of Monoplectanum. Zool Scr 25:203–213
Hayward CJ (1996b) Revision of the monogenean genus Polylabris (Microcotylidae). Invertebr Syst 10:995–1039
Hoedt FE, Dimmlich WF (1995) Egg and larval abundance and spawning localities of the anchovy (Engraulis australis) and pilchard (Sardinops neopilchardus) near Phillip Island, Victoria. Mar Freshwat Res 46:735–743
Hossen MS, Barton DP, Zhu X, Wassens S, Shamsi S (2020) Re-description and molecular characterisation of Choricotyle australiensis Roubal, Armitage & Rohde, 1983 (Monogenea: Diclidophoridae) infecting Chrysophrys auratus (Forster) (Perciformes: Sparidae). Syst Parasitol 97:815–825
Hossen MS, Barton DP, Wassens S, Shamsi S (2022) Molecular characterisation of the Monogenea parasitise in blue mackerel Scomber australasicus (Perciformes: Scombridae) in Australian waters. Int J Parasitol Parasites Wildl 19:115–127
Hutson KS, Ernst I, Whittington ID (2007) Risk assessment for metazoan parasites of yellowtail kingfish Seriola lalandi (Perciformes: Carangidae) in South Australian sea-cage aquaculture. Aquaculture 271:85–99
Jovelin R, Justine J-L (2001) Phylogenetic relationships within the polyopisthocotylean monogeneans (Platyhelminthes) inferred from partial 28S rDNA sequences. Int J Parasitol 31:393–401
Kumar R, Agarwal GP (1981) On a new monogenetic trematode Mazocraes chauhani n. sp. from the gill filaments of a clupeid fish Gudusia chapra (Ham.). Riv Parassitol 42:467–473
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Li M, Shi S-F, Brown CL, Yang T-B (2011) Phylogeographical pattern of Mazocraeoides gonialosae (Monogenea, Mazocraeidae) on the dotted gizzard shad, Konosirus punctatus, along the coast of China. Int J Parasitol 41:1263–1272
Littlewood DTJ, Rohde K, Clough KA (1997) Parasite speciation within or between host species?—Phylogenetic evidence from site-specific polystome monogeneans. Int J Parasitol 27:1289–1297
Mamaev YL (1982) Notes on the systematics of mazocraeid monogeneans with a redescription of some poorly studied taxa. Helminthologia 19:25–39
Mollaret I, Jamieson BGM, Justine JL (2000) Phylogeny of the Monopisthocotylea and Polyopisthocotylea (Platyhelminthes) inferred from 28S rDNA sequences. Int J Parasitol 30:171–185
Oliva ME, Sepulveda FA, González MT (2014) Parapedocotyle prolatili gen. n. et sp. n., a representative of a new subfamily of the Diclidophoridae (Monogenea), a gill parasite of Prolatilus jugularis (Teleostei: Pinguipedidae) from Chile. Folia Parasitol 61:543–548
Rambaut A (2014) FigTree v1.4.2, a graphical viewer of phylogenetic trees. http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 31 January 2019)
Reed C, MacKenzie K, Van der Lingen CD (2012) Parasites of South African sardines, Sardinops sagax, and an assessment of their potential as biological tags. Bull Eur Assoc Fish Pathol 32:41–48
Rohde K (1989a) Gill monogenea of Rastrelliger spp. (Scombridae). Syst Parasitol 14:79–91
Rohde K (1989b) Kuhnia sprostonae Price, 1961 and K. scombercolias Nasir & Fuentes Zambrano, 1983 (Monogenea: Mazocraeidae) and their microhabitats on the gills of Scomber australasicus (Teleostei: Scombridae), and the geographical distribution of seven species of gill Monogenea of Scomber spp. Syst Parasitol 14:93–100
Rohde K (2005) Marine parasitology. Csiro publishing, Collingwood, Victoria, Australia
Rohde K, Watson N (1985a) Morphology and geographical variation of Pseudokuhnia minor n.g., n. comb. (Monogenea: Polyopisthocotylea). Int J Parasitol 15:557–567
Rohde K, Watson N (1985b) Morphology, microhabitats and geographical variation of Kuhnia spp. (Monogenea: Polyopisthocotylea). Int J Parasitol 15:569–586
Rohde K, Hayward C, Heap M (1995) Aspects of the ecology of metazoan ectoparasites of marine fishes. Int J Parasitol 25:945–970
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Rowling K, Hegarty A, Ives M (eds) (2010) Australian Anchovy. In: Status of fisheries resources in NSW 2008/09. Industry & Investment NSW, Cronulla, pp 5–6
Sailaja B, Shameem U, Madhavi R (2019) Two new species of Mazocraes Hermann (Monogenea: Mazocraeidae) from clupeoid fishes off Visakhapatnam, Bay of Bengal. J Parasit Dis 43:313–318
Sandars DF (1945) Five new microcotylids from fish from Western Australian waters. J R Soc West Aust 29:107–135
Savage J, Hobsbawn P (2015) Australian fisheries and aquaculture statistics 2014, Fisheries Research and Development Corporation project 2014/245. ABARES,Canberra, December. CC BY 3.0. Canberra, Australia
Schade FM, Raupach MJ, Wegner KM (2016) Seasonal variation in parasite infection patterns of marine fish species from the Northern Wadden Sea in relation to interannual temperature fluctuations. J Sea Res 113:73–84
Shamsi S, Briand MJ, Justine JL (2017) Occurrence of Anisakis (Nematoda: Anisakidae) larvae in unusual hosts in Southern hemisphere. Parasitol Int 66:837–840
Stewart J, Ballinger G, Ferrell D (2010) Review of the biology and fishery for Australian sardines (Sardinops sagax) in New South Wales-2010, Industry & Investment NSW – Fish. Res Rep Ser No 26:1–61
Timi JT, Sardella NH, Etchegoin JA (1999) Mazocraeid monogeneans parasitic on engraulid fishes in the southwest Atlantic. J Parasitol 85:28–32
Víllora-Montero M, Pérez-del-Olmo A, Georgieva S, Raga JA, Montero FE (2020) Considerations on the taxonomy and morphology of Microcotyle spp.: redescription of M. erythrini van Beneden & Hesse, 1863 (sensu stricto) (Monogenea: Microcotylidae) and the description of a new species from Dentex dentex (L.) (Teleostei: Sparidae). Parasit Vectors 13:1–23
Ward RD, Holmes BH (2007) An analysis of nucleotide and amino acid variability in the barcode region of cytochrome c oxidase I (cox1) in fishes. Mol Ecol Notes 7:899–907
Ward TM, Staunton-Smith J, Hoyle S, Halliday IA (2003) Spawning patterns of four species of predominantly temperate pelagic fishes in the sub-tropical waters of southern Queensland. Estuar Coast Shelf Sci 56:1125–1140
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PD (2005) DNA barcoding Australia’s fish species. Philos Trans r Soc b: Biol Sci 360:1847–1857
Whittington ID, Chisholm L (2008) Diseases caused by Monogenea. In: Eiras JC, Segner H, Wahlii T, Kapoor BG (eds) Fish Diseases. Science Publishers Ltd, Manchester, NH, pp 683–816
Williams A (1988) Three new species of monogeneans of the family Mazocraeidae from clupeiform fishes in the Swan River Estuary, Western Australia. Syst Parasitol 12:93–104
Williams A (1991) Monogeneans of the families Microcotylidae Taschenberg, 1879 and Heteraxinidae Price, 1962 from Western Australia, including the description of Polylabris sandarsae n. sp. (Microcotylidae). Syst Parasitol 18:17–43
Woolcock V (1936) Monogenetic trematodes from some Australian fishes. Parasitology 28:79–91
Yan S, Wang M, Yang C-P, Zhi T-T, Brown CL, Yang T-B (2016) Comparative phylogeography of two monogenean species (Mazocraeidae) on the host of chub mackerel, Scomber japonicus, along the coast of China. Parasitol 143:594–605
Young PC (1969) Some monogenoideans of the family Diplectanidae Bychowsky, 1957 from Australian teleost fishes. J Helminthol 43:223–254
Young PC (1970) The species of Monogenoidea recorded from Australian fishes and notes on the zoogeography. Anales Inst Biol Univ Nac Autón México, Zool 41:163–176
Zhang J, Wu X, Xie M, Xu X, Li A (2011) The mitochondrial genome of Polylabris halichoeres (Monogenea: Microcotylidae). Mitochondrial DNA 22:3–5
Acknowledgements
Md. Shafaet Hossen is grateful for the PhD scholarship given by the Australian Research Training Program Scholarship through Charles Sturt University.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
MSH contributed to the development of initial concept and hypothesis, the design and implementation of all experimental works, the interpretation of data, the writing of the first draft, and funding acquisition. DPB contributed to the monitoring and assisting with experimental activity, such as observation and endorsement of microscopic species identification, article review and editing. SW and SS contributed to the overall supervision, review, and editing of the text, including an English check. All authors read and approved the final version.
Corresponding author
Ethics declarations
Ethics approval
All fish used in this work were bought from the commercial fish markets and fish were dead. Therefore, no ethical statement is needed regarding use of animals for scientific purposes.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Host-Parasite Interactions in the time of Global Change.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hossen, M., Barton, D.P., Wassens, S. et al. Molecular (cox1), geographical, and host record investigation of monogeneans Mazocraes australis (Mazocraeidae), Polylabris sillaginae, and P. australiensis (Microcotylidae). Parasitol Res 121, 3427–3442 (2022). https://doi.org/10.1007/s00436-022-07664-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07664-8