Abstract
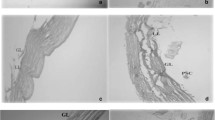
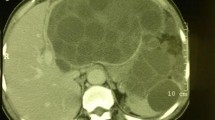
Recurrence of cystic echinococcosis as a result of treatment failure is frequently reported to cause a major problem in management of such serious parasitic infection. The deeply seated innermost germinal layer of hydatid cysts is a relatively delicate layer, yet responsible for viability maintenance of this parasitic stage. In this study, a trial was done to explore the ultrastructural changes in germinal and laminated layer of the hydatid cyst for the first time in human cases exposed to different therapeutic approaches which were done earlier to the final open surgical intervention. Four groups were included: group 1 did not receive any earlier form of treatment; group 2 was previously treated with only medical therapy; group 3 was treated with a single course of medical treatment, plus a single PAIR technique; group 4 was treated with multiple courses of medical treatment plus multiple PAIR techniques. Complete alteration of ultrastructural features of germinal and laminated layers were observed only with samples from group 4, indicating a kind of failure of the therapeutic approaches used in group, 1, 2, and 3, unless repeated in group 4 to achieve a real change regarding the fitness of the parasitic cystic lesions. Searching for more effective, safe, therapeutic method is highly recommended which may end the suffering of the affected patients.





Similar content being viewed by others
References
Allis CD, Jenuwein T (2016) The molecular hallmarks of epigenetic control. Nat Rev Genet 17:487–500. https://doi.org/10.1038/nrg.2016.59
Atmatzidis KS, Pavlidis TE, Papaziogas BT, Mirelis C, Papaziogas TB (2005) Recurrence and long-term outcome after open cystectomy with omentoplasty for hepatic hydatid disease in an endemic area. Acta Chir Belg 105:198–202
Bekhti A, Schaaps JP, Capron M, Dessaint JP, Santoro F, Capron A (1977) Treatment of hepatic hydatid disease with mebendazole: preliminary results in four cases. Br Med J 2:1047–1051
Brunetti E, Kern P, Vuitton DA, Writing Panel for the WHO-IWGE (2010) Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop 114:1–16. https://doi.org/10.1016/j.actatropica.2009.11.001
Burkitt HG, Young B, Heath JW (1993) Wheater’s functional histology–a text and colour atlas by, 3rd edition, 416 pp. Churchill Livingston, Edinburgh
Casaravilla C, Brearley C, Soulé S, Fontana C, Veiga N, Bessio MI, Ferreira F, Kremer C, Díaz A (2006) Characterization of myo-inositol hexakisphosphate deposits from larval Echinococcus granulosus. FEBS J 273:3192–3203. https://doi.org/10.1111/j.1742-4658.2006.05328.x
Corfield AP (2015) Mucins: a biologically relevant glycan barrier in mucosal protection. Biochim Biophys Acta 1850:236–252. https://doi.org/10.1016/j.bbagen.2014.05.003
Díaz A, Casaravilla C, Allen JE, Sim RB, Ferreira AM (2011a) Understanding the laminated layer of larval Echinococcus II: immunology. Trends Parasitol 27:264–273. https://doi.org/10.1016/j.pt.2011.01.008
Díaz A, Casaravilla C, Irigoín F, Lin G, Previato JO, Ferreira F (2011b) Understanding the laminated layer of larval Echinococcus I: structure. Trends Parasitol 27:204–213. https://doi.org/10.1016/j.pt.2010.12.012
Dziri C, Haouet K, Fingerhut A (2004) Treatment of Hydatid cyst of the liver: Where is the evidence? World J Surg 28:731–736
Flores-Pérez I, Fragoso Gonzalez G, Sciutto E, de Aluja AS (2003) Apoptosis induced by gamma irradiation of Taenia solium metacestodes. Parasitol Res 90:203–208. https://doi.org/10.1007/s00436-002-0819-9
Galindo M, Schadebrodt G, Galanti N (2008) Echinococcus granulosus: cellular territories and morphological regions in mature protoscoleces. Exp Parasitol 119:524–533. https://doi.org/10.1016/j.exppara.2008.04.013
Gupta S (2001) Molecular steps of death receptor and mitochondrial pathways of apoptosis. Life Sci 69:2957–2964
Hoberg EP, Sims DE, Odense PH (1995) Comparative morphology of the scolices and microtriches among five species of Tetrabothrius (Eucestoda: Tetrabothriidae). J Parasitol 81:475–481
Ingold K, Gottstein B, Hemphill A (2000) High molecular mass glycans are major structural elements associated with the laminated layer of in vitro cultivated Echinococcus multilocularis metacestodes. Int J Parasitol 30:207–214
Ingold K, Dai W, Rausch RL, Gottstein B, Hemphill A (2001) Characterization of the laminated layer of in vitro cultivated Echinococcus vogeli metacestodes. J Parasitol 87:55–64. https://doi.org/10.1645/0022-3395(2001)087[0055:COTLLO]2.0.CO;2
Irigoín F, Casaravilla C, Iborra F, Sim RB, Ferreira F, Díaz A (2004) Unique precipitation and exocytosis of a calcium salt of myo-inositol hexakisphosphate in larval Echinococcus granulosus. J Cell Biochem 93:1272–1281. https://doi.org/10.1002/jcb.20262
Irvine RF, Schell MJ (2001) Back in the water: the return of the inositol phosphates. Nat Rev Mol Cell Biol 2:327–338. https://doi.org/10.1038/35073015
Johannessen JV (ed) (1978) Cellular pathobiology: metabolic and storage diseases. McGraw-Hill, New York
Kim SH, Lee HS, Ryu DS et al (2011) Antibacterial activity of silver-nanoparticles against Staphylococcus aureus and Escherichia coli. Korean J Microbiol Biotechnol
Lacey E (1990) Mode of action of benzimidazoles. Parasitol Today 6:112–115
Manouras A, Genetzakis M, Lagoudianakis EE, Papadima A, Triantafillou C, Kekis PV, Filis K, Katergiannakis V (2007) Intact germinal layer of liver hydatid cysts removed after administration of albendazole. Neth J Med 65:112–116
Mehlhorn H, Eckert J, Thompson RC (1983) Proliferation and metastases formation of larval Echinococcus multilocularis. II. Ultrastructural investigations. Z Parasitenkd 69:749–763
Mueller PR, Dawson SL, Ferrucci JT, Nardi GL (1985) Hepatic echinococcal cyst: successful percutaneous drainage. Radiology 155:627–628. https://doi.org/10.1148/radiology.155.3.3890001
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Richards KS, Arme C, Bridges JF (1983) Echinococcus granulosus equinus: an ultrastructural study of murine tissue response to hydatid cysts. Parasitology 86:407–417. https://doi.org/10.1017/S0031182000050605
Robinson DG, Ehlers U, Herken R, Herrmann B, Mayer F, Schürmann FW (1987) Methods of preparation for electron microscopy. Springer, Berlin
Rogan MT, Richards KS (1989) Development of the tegument of Echinococcus granulosus (Cestoda) protoscoleces during cystic differentiation in vivo. Parasitol Res 75:299–306
Safioleas M, Misiakos E, Manti C, Katsikas D, Skalkeas G (1994) Diagnostic evaluation and surgical management of hydatid disease of the liver. World J Surg 18:859–865
Saimot AG, Meulemans A, Cremieux AC, Giovanangeli MD, Hay JM, Delaitre B, Coulaud JP (1983) Albendazole as a potential treatment for human hydatidosis. Lancet 2:652–656
Stettler M, Fink R, Walker M, Gottstein B, Geary TG, Rossignol JF, Hemphill A (2003) In vitro parasiticidal effect of Nitazoxanide against Echinococcus multilocularis metacestodes. Antimicrob Agents Chemother 47:467–474
Tamarozzi F, Vuitton L, Brunetti E, Vuitton DA, Koch S (2014) Non-surgical and non-chemical attempts to treat echinococcosis: do they work? Parasite 21:75. https://doi.org/10.1051/parasite/2014071
Tato P, Fernández AM, Solano S, Borgonio V, Garrido E, Sepulveda J, Molinari JL (2004) A cysteine protease from Taenia solium metacestodes induce apoptosis in human CD4+ T-cells. Parasitol Res 92:197–204. https://doi.org/10.1007/s00436-003-1008-1
Velasco-Tirado V, Romero-Alegría Á, Belhassen-García M, Alonso-Sardón M, Velasco CE, López-Bernús A, Carpio-Perez A, Jimenez López MF, Bellido JM, Muro A, Cordero-Sanchez M, Pardo-Lledias J, Muñoz-Bellvis L (2017) Recurrence of cystic echinococcosis in an endemic area: a retrospective study. BMC Infect Dis 17:455. https://doi.org/10.1186/s12879-017-2556-9
Verma VC, Gangwar M, Yashpal M, Nath G (2013) Anticestodal activity of endophytic Pestalotiopsis sp. on protoscoleces of hydatid cyst Echinococcus granulosus. Biomed Res Int 2013:308515. https://doi.org/10.1155/2013/308515
Walker M, Rossignol JF, Torgerson P, Hemphill A (2004) In vitro effects of nitazoxanide on Echinococcus granulosus protoscoleces and metacestodes. J Antimicrob Chemother 54(3):609–616
Zimmermann KC, Bonzon C, Green DR (2001) The machinery of programmed cell death. Pharmacol Ther 92:57–70
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This work was performed according to the national institutional ethical and professional guidelines for the management, follow-up, and post-operative care of hydatidosis cases. Informed written consent was supplied by individual patients.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: David Bruce Conn
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shaheen, H.A.A.A.S., El-Ahl, S.A.H.S., Raouf, A.M.A. et al. Ultrastructural changes in hydatid cyst walls obtained from human cases, exposed to different therapeutic approaches. Parasitol Res 118, 3149–3157 (2019). https://doi.org/10.1007/s00436-019-06446-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-019-06446-z