Abstract
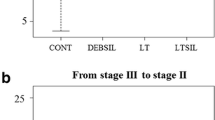
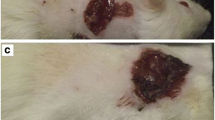
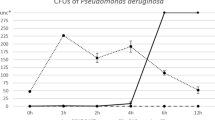
Larval therapy (LT) is the application of carrion flies (Diptera) sterile larvae on chronic or infected wounds to promote or accelerate the healing process. High cost and the development of resistance by certain groups of pathogenic bacteria to these drugs encouraged the resurgence of LT, currently used in approximately 20 countries and more recently in Brazil. This study evaluated the behavior and larval density of Cochliomyia macellaria F. (Calliphoridae), one of the most appropriate species for debridement of injuries with necrotic tissue. Tegumentar lesions were induced in Wistar rats by subcutaneously application of 0.2 ml of a 1:4 hydrochloric acid and sterile distilled water in the dorsal region. Five experimental groups were set up: (LT 5) treatment with 5 larvae/cm2; (LT 15) 15 larvae/cm2; (LT 25) 25 larvae/cm2; (DEB) mechanical debridement, and (NUL) animals that did not receive any treatment. In the LT groups, larvae used were sterilized with sodium hypochlorite (NaClO) and maintained for 12 h in the lesions. The healing process was assessed qualitatively (macroscopically and microscopically) and quantitatively (time interval to complete healing). It was observed that the immature fed only on necrotic tissue, thus C. macellaria is an excellent candidate for use in LT. There was no significant difference in healing time between experimental groups. However, it was observed that in LT 25, there was greater vascularization in tissues when compared to the other treatments. The mechanisms involved in this process are unknown, but it is evident that the larvae have an important role in modulating the host immune response. It is essential that future applications of larval therapy consider using a higher density of larvae (minimum of 25 larvae/cm2) than is currently recommended.


Similar content being viewed by others
References
Dokur M, Eroglu F, Ipek DNS, Ulutasdemir N (2015) Two different myiasis cases in southeast of Turkey: ophthalmomyiasis and cutaneous myiasis. Parasitol Res. doi:10.1007/s00436-015-4455-6
Fleischmann W, Grassberger M, Sherman R (2004) Maggot therapy: a handbook of maggot-assisted wound healing. Thieme, New York, 84p
Grella MD, Thyssen PJ (2011) Chave taxonômica interativa para espécies de dípteros califorídeos (Infraordem: Muscomorpha) do Brasil. http://keys.lucidcentral.org/keys/v3/calliphoridae_brazil. Accessed 09 January 2015
Horn KL, Cobb JRAH, Gates GA (1976) Maggot therapy for subacute mastoiditis. Arch Otolaryngol Head Neck Surg 102:377–379
Linhares AX, Thyssen PJ (2007) Miíases de importância médica: moscas e entomologia forense. In: De Carli GA (ed) Parasitologia clínica: seleção de métodos e técnicas de laboratório para o diagnóstico das parasitoses humanas, 2nd edn. Atheneu, São Paulo, pp 709–730
Marcondes CB (2006) Terapia larval: de lesões de pele causadas por diabetes e outras doenças. Editora da UFSC, Santa Catarina, 89p
Martini RK, Sherman RA (2003) Terapia de desbridamento com larvas. JBM 85:82–85
Masiero FS, Nassu MP, Soares MP, Thyssen PJ (2015) Histological patterns in healing chronic wounds using Cochliomyia macellaria (Diptera: Calliphoridae) larvae and other therapeutic measures. Parasitol Res. doi:10.1007/s00436-015-4487-y
Mehlhorn H, Al-Rasheid KA, Abdel-Ghaffar F, Klimpel S, Pohle H (2010) Life cycle and attacks of ectoparasites on ruminants during the year in Central Europe: recommendations for treatment with insecticides (e.g., Butox). Parasitol Res 107(2):425–431
Moretti TC, Thyssen PJ, Solis DR (2009) Breeding of the scuttle fly Megaselia scalaris in a fish carcass and implications for the use in forensic entomology (Diptera: phoridae). Entomol Gen 31:349–353
Pinheiro MARQ, Ferraz JB, Junior MAA, Moura AD, Costa MESM, Costa FJMD, Neto VFA, Neto RM, Gama RA (2015) Use of maggot therapy for treating a diabetic foot ulcer colonized by multidrug resistant bacteria in Brazil. Indian J Med Res 141:340–342
Santos VLCG (2000) Avanços tecnológicos no tratamento de feridas e algumas aplicações em domicílio. In: Duarte YAO, Diogo MJD (eds) Atendimento domiciliar: um enfoque gerontológico. Atheneu, São Paulo, pp 265–306
SAS Institute Incorporation (2006) S.A.S. user’s guide: statistics, version 6.12. S.A.S. Institute Inc, Cary
Sherman RA (2003) Maggot therapy for treating diabetic foot ulcers unresponsive to conventional therapy. Diabetes Care 26:446–451
Sherman RA, Pechter EA (1988) Maggot therapy a review of therapeutic applications of fly larvae in human medicine, especially for treating osteomyelitis. Med Vet Entomol 2:225–230
Sherman RA, Wyle FA, Vulpe M (1995) Maggot therapy for treating pressure ulcers in spinal cord injury patients. J Spinal Cord Med 18:71–74
Sherman RA, Hall MJR, Thomas S (2000) Medical maggots: an ancient remedy for some contemporary afflictions. Annu Rev Entomol 45:55–81
Simmons SW (1935) A bactericidal principle in excretions of surgical maggots which destroys important etiological agents of pyogenic infections. J Bacteriol 30:253–267
Soares MO, Iglesias CP, Bland JM, Cullum N, Dumville JC, Nelson EA, Torgerson DJ, Worthy G (2009) Cost effectiveness analysis of larval therapy for leg ulcers. BMJ 338:1–8
Teich S, Myers RA (1986) Maggot therapy for severe skin infection. South Med J 79:1153–1155
Telford G, Brown AP, Rich A, English JSC, Pritchard DI (2012) Wound debridement potential of glycosidases of the wound-healing maggot, Lucilia sericata. Med Vet Entomol 26:291–299
Thyssen PJ, Nassu MP, Costella AM, Costella ML (2012) Record of oral myiasis by Cochliomyia hominivorax (Diptera: Calliphoridae): case evidencing negligence in the treatment of incapable. Parasitol Res 111(2):957–959
Thyssen PJ, Nassu MP, Nitsche MJT, Leite DS (2013) Sterilization of immature blowflies (Calliphoridae) for use in larval therapy. J Med Med Sci 4:405–409
Turkmen A, Graham K, Mcgrouther DA (2010) Therapeutic applications of the larvae for wound debridement. J Plast Reconstr Aesthet Surg 63:184–188
Van der Plas MJA, Van der Does AM, Baldry M, Dogterom-Ballering HCM, Van Gulpen C, Van Dissel JT, Nibbering PH, Jukema GN (2007) Maggot excretions/secretions inhibit multiple neutrophil pro-inflammatory responses. Microbes Infect 9:507–514
Vistnes LM, Lee R, Ksander GA (1981) Proteolytic activity of blowfly larvae secretions in experimental burns. Surgery 90:835–841
Zhang Z, Wang S, Diao Y, Zhang J, Lv D (2010) Fatty acid extracts from Lucilia sericata larvae promote murine cutaneous wound healing by angiogenic activity. Lipids Health Dis 9:24
Acknowledgments
The authors would like to thank Dr. Mauro P. Soares for assistance in histopathology. Financial support grant 2012/06033-5, São Paulo Research Foundation (FAPESP).
Ethical standards
All experiments were carried out in accordance with the Committee of Ethics in the Use of Animals (CEUA/Unicamp) of October 25 2013 (protocol n° 2689-1A).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nassu, M.P., Thyssen, P.J. Evaluation of larval density Cochliomyia macellaria F. (Diptera: Calliphoridae) for therapeutic use in the recovery of tegumentar injuries. Parasitol Res 114, 3255–3260 (2015). https://doi.org/10.1007/s00436-015-4542-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4542-8