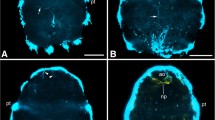
Abstract
Acoel statocysts are fluid-filled chambers formed by two parietal cells underlying basal lamina as a capsule and contain a movable statolith cell called a lithocyte. The statocyst is needed for geotaxis; however, the mechanism of the gravity receptor system has not been elucidated. We focused on the geotactic ability of the acoel Praesagittifera naikaiensis, acquired during the development of the statocyst and its nervous system after hatching, and examined the three-dimensional relationship between the statocyst and its nervous system. Acoel geotactic ability was acquired between 0 and 7 days after hatching. No major changes in neural structures, namely a commissural brain, nerve cords, and commissures, were observed between juveniles and adults. The statocyst-associated commissure (stc), a commissural brain component, was circular and was located ventral to the statocyst but was not observed in neural connections to the capsule’s lumen. Fine structures of the statocyst revealed that the statolith developed after hatching. We hypothesized that geotactic ability needs the following conditions: (1) a sufficient concentration of calcium salt in the statolith; (2) stc must work as afferent neurons; and (3) a ventral polar cell, which is present outside the capsule, must be a sensory cell stimulated by the lithocyte.








Similar content being viewed by others
References
Achatz JG, Martínez P (2012) The nervous system of Isodiametra pulchra (Acoela) with a discussion on the neuroanatomy of the Xenacoelomorpha and its evolutionary implications. Front Zool 9:27. https://doi.org/10.1186/1742-9994-9-27
Achatz JG, Hooge M, Wallberg A, Jondelius U, Tyler S (2010) Systematic revision of acoels with 9 + 0 sperm ultrastructure (Convolutida) and the influence of sexual conflict on morphology. J Zool Syst Evol Res 48:9–32. https://doi.org/10.1111/j.1439-0469.2009.00555.x
Arboleda E, Hartenstein V, Martínez P, Reichert H, Sen S, Sprecher S, Bailly X (2018) An emerging system to study photosymbiosis, brain regeneration, chronobiology, and behavior: the marine Acoel Symsagittifera roscoffensis. BioEssays 40:1800107. https://doi.org/10.1002/bies.201800107
Bailly X, Laguerre L, Correc G et al (2014) The chimerical and multifaceted marine acoel Symsagittifera roscoffensis: from photosymbiosis to brain regeneration. Front Microbiol 5:498. https://doi.org/10.3389/fmicb.2014.00498
Bery A, Martínez P (2011) Acetylcholinesterase activity in the developing and regenerating nervous system of the acoel Symsagittifera roscoffensis. Acta Zool 92:383–392. https://doi.org/10.1111/j.1463-6395.2010.00472.x
Bery A, Cardona A, Martínez P, Hartenstein V (2010) Structure of the central nervous system of a juvenile acoel, Symsagittifera roscoffensis. Dev Genes Evol 220:61–76. https://doi.org/10.1007/s00427-010-0328-2
Bezares-Calderón LA, Berger J, Jékely G (2020) Diversity of cilia-based mechanosensory systems and their functions in marine animal behaviour. Phil Trans R Soc B 375:20190376. https://doi.org/10.1098/rstb.2019.0376
Brusca RC, Moore W, Shuster SM (2016) Invertebrates, 3rd edn. Sinauer Associates, Massachusetts
Budelmann BU (1988) Morphological diversity of equilibrium receptor systems in aquatic invertebrates. In: Atema J, Fay RR, Popper AN, Tavolga WN (eds) Sensory biology of aquatic animals. Springer, New York, pp 757–782
Cannon JT, Vellutini BC, Smith J, Ronquist F, Jondelius U, Hejnol A (2016) Xenacoelomorpha is the sister group to Nephrozoa. Nature 530:89–93. https://doi.org/10.1038/nature16520
Clapham DE (2003) TRP channels as cellular sensors. Nature 426:517–524. https://doi.org/10.1038/nature02196
Dittmann IL, Zauchner T, Nevard LM, Telford MJ, Egger B (2018) SALMFamide2 and serotonin immunoreactivity in the nervous system of some acoels (Xenacoelomorpha). J Morphol 279:589–597. https://doi.org/10.1002/jmor.20794
Ehlers U (1991) Comparative morphology of statocysts in the Plathelminthes and the Xenoturbellida. In: Tyler S (ed) Turbellarian biology. Springer, Dordrecht, pp 263–271
Ehlers U, Sopott-Ehlers B (1990) Organization of statocysts in the Otoplanidae (Plathelminthes): an ultrastructural analysis with implications for the phylogeny of the Proseriata. Zoomorphology 109:309–318. https://doi.org/10.1007/BF00803571
Ferrero E (1973) A fine structural analysis of the statocyst in Turbellaria Acoela. Zool Scr 2:5–16. https://doi.org/10.1111/j.1463-6409.1973.tb00793.x
Ferrero EA, Bedini C (1991) Ultrastructural aspects of nervous-system and statocyst morphogenesis during embryonic development of Convoluta psammophila (Turbellaria, Acoela). Hydrobiol 227:131–137. https://doi.org/10.1007/BF00027592
Gamble FW, Keeble F (1904) The bionomics of Convoluta roscoffensis, with special reference to its green cells. Proc R Soc Lond 72:93–98. https://doi.org/10.1098/rspl.1903.0022
Gavilán B, Perea-Atienza E, Martínez P (2016) Xenacoelomorpha: a case of independent nervous system centralization? Phil Trans R Soc B 371:20150039. https://doi.org/10.1098/rstb.2015.0039
Gay S, Martinez-Hernandez A, Rhodes RK, Miller EJ (1981) The collagenous exocytoskeleton of smooth muscle cells. Collagen Relat Res 1:377–384. https://doi.org/10.1016/S0174-173X(81)80014-3
Gschwentner R, Baric S, Rieger R (2002) New model for the formation and function of sagittocysts: Symsagittifera corsicae n. sp. (Acoela). Invertebr Biol 121:95–103. https://doi.org/10.1111/j.1744-7410.2002.tb00050.x
Horridge GA (1969) Statocysts of medusae and evolution of stereocilia. Tissue Cell 1:341–353. https://doi.org/10.1016/S0040-8166(69)80029-7
Inoue T, Yamashita T, Agata K (2014) Thermosensory signaling by TRPM is processed by brain serotonergic neurons to produce planarian thermotaxis. J Neurosc 34:15701–15714. https://doi.org/10.1523/JNEUROSCI.5379-13.2014
Jacob MP (2003) Extracellular matrix remodeling and matrix metalloproteinases in the vascular wall during aging and in pathological conditions. Biomed Pharmacother 57:195–202. https://doi.org/10.1016/S0753-3322(03)00065-9
Jondelius U, Wallberg A, Hooge M, Raikova OI (2011) How the worm got its pharynx: phylogeny, classification and Bayesian assessment of character evolution in Acoela. Syst Biol 60:845–871. https://doi.org/10.1093/sysbio/syr073
Keeble F (1912) Plant-animals: a study in symbiosis. Cambridge University Press, London
Marlétaz F, Peijnenburg KT, Goto T et al (2019) A new spiralian phylogeny places the enigmatic arrow worms among gnathiferans. Curr Biol 29:312–318. https://doi.org/10.1016/j.cub.2018.11.042
Moran MM, Xu H, Clapham DE (2004) TRP ion channels in the nervous system. Curr Opin Neurobiol 14:362–369. https://doi.org/10.1016/j.conb.2004.05.003
Peng G, Shi X, Kadowaki T (2015) Evolution of TRP channels inferred by their classification in diverse animal species. Mol Phylogenet Evol 84:145–157. https://doi.org/10.1016/j.ympev.2014.06.016
Philippe H, Brinkmann H, Copley R et al (2011) Acoelomorph flatworms are deuterostomes related to Xenoturbella. Nature 470:255–258. https://doi.org/10.1038/nature09676
Philippe H, Poustka AJ, Chiodin M et al (2019) Mitigating anticipated effects of systematic errors supports sister-group relationship between Xenacoelomorpha and Ambulacraria. Curr Biol 29:1818–1826. https://doi.org/10.1016/j.cub.2019.04.009
Raikova OI, Reuter M, Kotikova EA, Gustafsson MK (1998) A commissural brain! The pattern of 5-HT immunoreactivity in Acoela (Plathelminthes). Zoomorphology 118:69–77. https://doi.org/10.1007/s004350050058
Reichmuth C, Becker S, Benz M et al (1995) The sap47 gene of Drosophila melanogaster codes for a novel conserved neuronal protein associated with synaptic terminals. Mol Brain Res 32:45–54. https://doi.org/10.1016/0169-328X(95)00058-Z
Sarfatti G, Bedini C (1965) The symbiont alga of the flatworm Convoluta psammophila Bekl. observed at the electron microscope. Caryologia 18:207–223. https://doi.org/10.1080/00087114.1965.10796166
Semmler H, Bailly X, Wanninger A (2008) Myogenesis in the basal bilaterian Symsagittifera roscoffensis (Acoela). Front Zool 5:14. https://doi.org/10.1186/1742-9994-5-14
Semmler H, Chiodin M, Bailly X, Martínez P, Wanninger A (2010) Steps towards a centralized nervous system in basal bilaterians: insights from neurogenesis of the acoel Symsagittifera roscoffensis. Dev Growth Differ 52:701–713. https://doi.org/10.1111/j.1440-169X.2010.01207.x
Sprecher SG, Bernardo-Garcia FJ, van Giesen L et al (2015) Functional brain regeneration in the acoel worm Symsagittifera roscoffensis. Biol Open 4:1688–1695. https://doi.org/10.1242/bio.014266
Tamm SL (1982) Ctenophora. In: Shelton GAB (ed) Electrical conduction and behaviour in ‘simple’ invertebrates. Oxford University Press, pp 266–358
Tamm SL (2014) Formation of the statolith in the ctenophore Mnemiopsis leidyi. Biol Bull 227:7–18. https://doi.org/10.1086/BBLv227n1p7
Venkatachalam K, Montell C (2007) TRP channels. Annu Rev Biochem 76:387–417. https://doi.org/10.1146/annurev.biochem.75.103004.142819
Yamasu T (1991) Fine structure and function of ocelli and sagittocysts of acoel flatworms. Hydrobiol 227:273–282. https://doi.org/10.1007/BF00027612
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sakagami, T., Watanabe, K., Ikeda, R. et al. Structural analysis of the statocyst and nervous system of Praesagittifera naikaiensis, an acoel flatworm, during development after hatching. Zoomorphology 140, 183–192 (2021). https://doi.org/10.1007/s00435-021-00521-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-021-00521-9