Abstract
Background
Observational studies have revealed relationships between circulating vitamin D concentrations and the risk of different types of cancer, although the potential causal relationship remains controversial. This study aims to investigate the presence of a causal relationship between circulating vitamin D concentrations and the risk of different types of cancer.
Methods
Summary statistics from corresponding genome-wide association studies (GWASs) were used to investigate the causal relationship between circulating vitamin D concentrations and the risk of 14 cancers. A two-sample Mendelian randomization (MR) analysis using inverse-variance weighting (IVW) as the primary method was performed. Additionally, the results were verified using four other methods, including MR-Egger, weighted median, weighted mode, and simple mode. Multiple sensitivity analyses were conducted to ensure the robustness of our MR findings.
Results
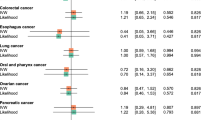
The MR analysis showed no causal relationship between circulating vitamin D concentrations and most types of cancer, except for a causal relationship with melanoma skin cancer (MSC) (odds ratio [OR]IVW = 1.003, 95% confidence intervals [CI] 1.001–1.005, P = 0.004). Conversely, reverse MR revealed a causal relationship between circulating vitamin D concentration and colorectal cancer (ORIVW = 0.398, 95% CI 0.195–0.813, P = 0.01; ORweighted median = 0.352, 95% CI 0.135–0.917, P = 0.03).
Conclusions
Our findings provide support for a causal relationship between circulating vitamin D concentration and risk of MSC. Additionally, we found a causal relationship between circulating vitamin D concentration and risk of colorectal cancer in reverse-MR analysis. This evidence indicate that vitamin D is of great significance in the prevention and treatment of MSC and the prognosis of colorectal cancer.



Similar content being viewed by others
Availability of data and materials
This study was based on publicly available summarized data [IEU OPEN GWAS PROJECT: https://gwas.mrcieu.ac.uk/ (accessed on 1 June 2023)].
Abbreviations
- MR:
-
Mendelian randomization
- GWAS:
-
Genome-wide association study
- OR:
-
Odds ratio
- SNPs:
-
Single-nucleotide polymorphisms
- CI:
-
Confidence interval
- IVW:
-
Inverse-variance-weighted
- MR-PRESSO:
-
MR pleiotropy RESidual sum and outlier
- MSC:
-
Melanoma skin cancer
- NMSC:
-
Non-melanoma skin cancer
- NSNP:
-
Number of single-nucleotide polymorphism
References
Asgari MM, Maruti SS, Kushi LH, White E (2009) A cohort study of vitamin D intake and melanoma risk. J Invest Dermatol 129(7):1675–1680. https://doi.org/10.1038/jid.2008.451
Asgari MM, Tang J, Warton ME, Chren M-M, Quesenberry CP, Bikle D, Horst RL, Orentreich N, Vogelman JH, Friedman GD (2010) Association of prediagnostic serum vitamin D levels with the development of basal cell carcinoma. J Invest Dermatol 130(5):1438–1443. https://doi.org/10.1038/jid.2009.402
Bauer SR, Hankinson SE, Bertone-Johnson ER, Ding EL (2013) Plasma vitamin D levels, menopause, and risk of breast cancer: dose-response meta-analysis of prospective studies. Medicine 92(3):123–131. https://doi.org/10.1097/MD.0b013e3182943bc2
Ben-Shoshan M, Amir S, Dang DT, Dang LH, Weisman Y, Mabjeesh NJ (2007) 1alpha,25-dihydroxyvitamin D3 (calcitriol) inhibits hypoxia-inducible factor-1/vascular endothelial growth factor pathway in human cancer cells. Mol Cancer Ther 6(4):1433–1439. https://doi.org/10.1158/1535-7163.MCT-06-0677
Bowden J, Smith GD, Burgess S (2015) Mendelian randomization with invalid instruments: effect estimation and bias detection through egger regression. Int J Epidemiol 44(2):512–525. https://doi.org/10.1093/ije/dyv080
Bowden J, Smith GD, Haycock PC, Burgess S (2016) Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 40(4):304–314. https://doi.org/10.1002/gepi.21965
Burgess S, Thompson SG (2011) Bias in causal estimates from mendelian randomization studies with weak instruments. Stat Med 30(11):1312–1323. https://doi.org/10.1002/sim.4197
Burgess S, Thompson SG (2017) Interpreting findings from mendelian randomization using the MR-Egger method. Eur J Epidemiol 32(5):377–389. https://doi.org/10.1007/s10654-017-0255-x
Burgess S, Foley CN, Zuber V (2018) Inferring causal relationships between risk factors and outcomes from genome-wide association study data. Annu Rev Genomics Hum Genet 19(August):303–327. https://doi.org/10.1146/annurev-genom-083117-021731
Burgess S, Foley CN, Allara E, Staley JR, Howson JMM (2020) A robust and efficient method for mendelian randomization with hundreds of genetic variants. Nat Commun 11(1):376. https://doi.org/10.1038/s41467-019-14156-4
Cho M, Peddi PF, Ding K, Chen L, Thomas D, Wang J, Lockhart AC, Tan B, Wang-Gillam A (2013) Vitamin D deficiency and prognostics among patients with pancreatic adenocarcinoma. J Transl Med 11(September):206. https://doi.org/10.1186/1479-5876-11-206
Chung I, Han G, Seshadri M, Gillard BM, Wei-dong Yu, Foster BA, Trump DL, Johnson CS (2009) Role of vitamin D receptor in the antiproliferative effects of calcitriol in tumor-derived endothelial cells and tumor angiogenesis in vivo. Can Res 69(3):967–975. https://doi.org/10.1158/0008-5472.CAN-08-2307
Díaz GD, Paraskeva C, Thomas MG, Binderup L, Hague A (2000) Apoptosis is induced by the active metabolite of vitamin D3 and its analogue EB1089 in colorectal adenoma and carcinoma cells: possible implications for prevention and therapy. Can Res 60(8):2304–2312
Dimitrakopoulou VI, Tsilidis KK, Haycock PC, Dimou NL, Al-Dabhani K, Martin RM, Lewis SJ et al (2017) Circulating vitamin D concentration and risk of seven cancers: mendelian randomisation study. BMJ (clinical Research Ed.) 359(October):j4761. https://doi.org/10.1136/bmj.j4761
Emdin CA, Khera AV, Kathiresan S (2017) Mendelian randomization. JAMA 318(19):1925–1926. https://doi.org/10.1001/jama.2017.17219
Feldman D, Krishnan AV, Swami S, Giovannucci E, Feldman BJ (2014) The role of vitamin D in reducing cancer risk and progression. Nat Rev Cancer 14(5):342–357. https://doi.org/10.1038/nrc3691
Feskanich D, Ma J, Fuchs CS, Kirkner GJ, Hankinson SE, Hollis BW, Giovannucci EL (2004) Plasma vitamin D metabolites and risk of colorectal cancer in women. Cancer Epidemiol Biomark Prev 13(9):1502–1508
Field S, Newton-Bishop JA (2011) Melanoma and vitamin D. Mol Oncol 5(2):197–214. https://doi.org/10.1016/j.molonc.2011.01.007
Folkersen L, Gustafsson S, Wang Q, Hansen DH, Hedman ÅK, Schork A, Page K et al (2020) Genomic and drug target evaluation of 90 cardiovascular proteins in 30,931 individuals. Nat Metab 2(10):1135–1148. https://doi.org/10.1038/s42255-020-00287-2
Gandini S, Boniol M, Haukka J, Byrnes G, Cox B, Sneyd MJ, Mullie P, Autier P (2011) Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int J Cancer 128(6):1414–1424. https://doi.org/10.1002/ijc.25439
Giammanco M, Di Majo D, La Guardia M, Aiello S, Crescimannno M, Flandina C, Tumminello FM, Leto G (2015) Vitamin D in cancer chemoprevention. Pharm Biol 53(10):1399–1434. https://doi.org/10.3109/13880209.2014.988274
Hektoen HH, Robsahm TE, Stenehjem JS, Axcrona K, Babigumira R, Mondul AM, Gislefoss RE, Andreassen BK (2021) Vitamin D and vitamin D-binding protein and risk of bladder cancer: a nested case-control study in the Norwegian Janus serum bank cohort. Cancer Med 10(12):4107–4116. https://doi.org/10.1002/cam4.3960
Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D, Laurin C et al (2018) The MR-base platform supports systematic causal inference across the human phenome. Elife 7(4):e34408. https://doi.org/10.7554/eLife.34408
Hiraki LT, Conghui Qu, Hutter CM, Baron JA, Berndt SI, Bézieau S, Brenner H et al (2013) Genetic predictors of circulating 25-hydroxyvitamin d and risk of colorectal cancer. Cancer Epidemiol Biomark Prev 22(11):2037–2046. https://doi.org/10.1158/1055-9965.EPI-13-0209
Jenab M, Bas Bueno-de-Mesquita H, Ferrari P, van Duijnhoven FJB, Norat T, Pischon T, Jansen EHJM et al (2010) Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations: a nested case-control study. BMJ (clinical Research ) 340(January):b5500. https://doi.org/10.1136/bmj.b5500
Jeon S-M, Shin E-A (2018) Exploring vitamin D metabolism and function in cancer. Exp Mol Med 50(4):1–14. https://doi.org/10.1038/s12276-018-0038-9
Liang G, Nan H, Qureshi AA, Han J (2012) Pre-diagnostic plasma 25-hydroxyvitamin D levels and risk of non-melanoma skin cancer in women. PLoS ONE 7(4):e35211. https://doi.org/10.1371/journal.pone.0035211
Liyanage UE, Law MH, Melanoma Meta-analysis Consortium, Barrett JH, Iles MM, MacGregor S (2020) Is there a causal relationship between vitamin D and melanoma risk? A mendelian randomization study. Br J Dermatol 182(1):97–103. https://doi.org/10.1111/bjd.18238
McCullough ML, Zoltick ES, Weinstein SJ, Fedirko V, Wang M, Cook NR, Heather Eliassen A et al (2019) Circulating vitamin D and colorectal cancer risk: an international pooling project of 17 cohorts. J Natl Cancer Inst 111(2):158–169. https://doi.org/10.1093/jnci/djy087
Mezawa H, Sugiura T, Watanabe M, Norizoe C, Takahashi D, Shimojima A, Tamez S, Tsutsumi Y, Yanaga K, Urashima M (2010) Serum vitamin D levels and survival of patients with colorectal cancer: post-hoc analysis of a prospective cohort study. BMC Cancer 10(July):347. https://doi.org/10.1186/1471-2407-10-347
Mondul AM, Shui IM, Kai Yu, Travis RC, Stevens VL, Campa D, Schumacher FR et al (2013) Genetic variation in the vitamin D pathway in relation to risk of prostate cancer-results from the breast and prostate cancer cohort consortium. Cancer Epidemiol Biomark Prev 22(4):688–696. https://doi.org/10.1158/1055-9965.EPI-13-0007-T
Newton-Bishop JA, Beswick S, Randerson-Moor J, Chang Y-M, Affleck P, Elliott F, Chan M et al (2009) Serum 25-hydroxyvitamin D3 levels are associated with Breslow thickness at presentation and survival from melanoma. J Clin Oncol 27(32):5439–5444. https://doi.org/10.1200/JCO.2009.22.1135
Palanca A, Ampudia-Blasco FJ, Real JT (2022) The controversial role of vitamin D in thyroid cancer prevention. Nutrients 14(13):2593. https://doi.org/10.3390/nu14132593
Pálmer HG, Larriba MJ, García JM, Ordóñez-Morán P, Peña C, Peiró S, Puig I et al (2004) The transcription factor SNAIL represses vitamin D receptor expression and responsiveness in human colon cancer. Nat Med 10(9):917–919. https://doi.org/10.1038/nm1095
Reichrath J (2007) Vitamin D and the skin: an ancient friend. Revisited Exp Dermatol 16(7):618–625. https://doi.org/10.1111/j.1600-0625.2007.00570.x
Revez JA, Lin T, Qiao Z, Xue A, Holtz Y, Zhu Z, Zeng J et al (2020) Genome-wide association study identifies 143 loci associated with 25 hydroxyvitamin D concentration. Nat Commun 11(1):1647. https://doi.org/10.1038/s41467-020-15421-7
Skrivankova VW, Richmond RC, Woolf BAR, Yarmolinsky J, Davies NM, Swanson SA, VanderWeele TJ et al (2021) Strengthening the reporting of observational studies in epidemiology using mendelian randomization: the STROBE-MR statement. JAMA 326(16):1614–1621. https://doi.org/10.1001/jama.2021.18236
Soares AM, Szejnfeld VL, Enokihara MY, Michalany N, Castro CH (2018) High serum 25-hydroxyvitamin D concentration in patients with a recent diagnosis of non-melanoma skin cancer: a case-control study. Eur J Dermatol EJD 28(5):649–653. https://doi.org/10.1684/ejd.2018.3401
Tang JY, Parimi N, Angela W, John Boscardin W, Shikany JM, Chren M-M, Cummings SR, Epstein EH, Bauer DC (2010) Inverse association between serum 25(OH) vitamin D levels and non-melanoma skin cancer in elderly men. Cancer Causes Control CCC 21(3):387–391. https://doi.org/10.1007/s10552-009-9470-4
van der Pols JC, Russell A, Bauer U, Neale RE, Kimlin MG, Green AC (2013) Vitamin D status and skin cancer risk independent of time outdoors: 11-year prospective study in an Australian community. J Investig Dermatol 133(3):637–641. https://doi.org/10.1038/jid.2012.346
Verbanck M, Chen C-Y, Neale B, Do R (2018) Detection of widespread horizontal pleiotropy in causal relationships inferred from mendelian randomization between complex traits and diseases. Nat Genet 50(5):693–698. https://doi.org/10.1038/s41588-018-0099-7
Vinceti M, Malagoli C, Fiorentini C, Longo C, Crespi CM, Albertini G, Ricci C et al (2011) Inverse association between dietary vitamin D and risk of cutaneous melanoma in a Northern Italy population. Nutr Cancer 63(4):506–513. https://doi.org/10.1080/01635581.2011.539314
Wactawski-Wende J, Kotchen JM, Anderson GL, Assaf AR, Brunner RL, O’Sullivan MJ, Margolis KL et al (2006) Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med 354(7):684–696. https://doi.org/10.1056/NEJMoa055222
Wei H, Jing H, Wei Q, Wei G, Heng Z (2018) Associations of the risk of lung cancer with serum 25-hydroxyvitamin D level and dietary vitamin d intake: a dose-response PRISMA meta-analysis. Medicine 97(37):e12282. https://doi.org/10.1097/MD.0000000000012282
Winsløw UC, Nordestgaard BG, Afzal S (2018) High plasma 25-hydroxyvitamin D and high risk of nonmelanoma skin cancer: a mendelian randomization study of 97 849 individuals. Br J Dermatol 178(6):1388–1395. https://doi.org/10.1111/bjd.16127
Wu J, Yang N, Yuan M (2021) Dietary and circulating vitamin D and risk of renal cell carcinoma: a meta-analysis of observational studies. Int Braz J Urol off J Brazil Soc Urol 47(4):733–744. https://doi.org/10.1590/S1677-5538.IBJU.2020.0417
Xin Yu, He L, Luan Z, Lv H, Yang H, Zhou Y, Zhao X et al (2017) E-Cadherin mediates the preventive effect of vitamin D3 in colitis-associated carcinogenesis. Inflamm Bowel Dis 23(9):1535–1543. https://doi.org/10.1097/MIB.0000000000001209
Zgaga L, O’Sullivan F, Cantwell MM, Murray LJ, Thota PN, Coleman HG (2016) Markers of vitamin D exposure and esophageal cancer risk: a systematic review and meta-analysis. Cancer Epidemiol Biomark Prev 25(6):877–886. https://doi.org/10.1158/1055-9965.EPI-15-1162
Zhang R, Naughton DP (2010) Vitamin D in health and disease: current perspectives. Nutr J 9(December):65. https://doi.org/10.1186/1475-2891-9-65
Acknowledgements
This work was made possible by the generous sharing of GWAS summary statistics. The authors would like to thank the participants, researchers, and staff associated with the many other studies from which we used data for this report. The authors would also like to thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
DY and CY designed the study and drafted the paper. DY, CY, PY, and JR critically revised the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have given consent to the publication of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Additional file 1:
Table S1. The characteristics of SNPs adopted as instrumental variables of circulating 25(OH)D concentration. Table S2. Mendelian randomization results from different types of cancer to circulating 25(OH)D concentration. Table S3. The characteristics of SNPs adopted as instrumental variables of reverse Mendelian randomization. Table S4. Confounding factorsassociated with different types of cancer. Additional file 2: Figure S1. Leave-one-out inverse-variance weighted mendelian randomization analyses of circulating 25(OH)D concentration on different types of cancer. Figure S2. Scatter plot of the causal effect of circulating 25(OH)D concentration on different types of cancer. Figure S3. Funnel plot of the causal effect of circulating 25(OH)D concentration on different types of cancer. Figure S4. Forest plot of the causal effect of circulating 25(OH)D concentration on different types of cancer. Figure S5. Leave-one-out inverse-variance weighted mendelian randomization analyses of different types of cancer on circulating 25(OH)D concentration. Figure S6. Scatter plot of the causal effect of different types of cancer on circulating 25(OH)D concentration. Figure S7. Funnel plot of the causal effect of different types of cancer on circulating 25(OH)D concentration. Figure S8. Forest plot of the causal effect of different types of cancer on circulating 25(OH)D concentration. Figure S9. Mendelian randomization results plot of circulating 25(OH) D concentration and melanoma skin cancer (secondary validation). (RAR 4700 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dai, Y., Chen, Y., Pu, Y. et al. Circulating vitamin D concentration and risk of 14 cancers: a bidirectional Mendelian randomization study. J Cancer Res Clin Oncol 149, 15457–15467 (2023). https://doi.org/10.1007/s00432-023-05322-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05322-9