Abstract
Background
Recent studies have shown that inflammatory bowel disease (IBD) is associated with bladder cancer (BC) incidence. But there is still a lack of understanding regarding its pathogenesis. Thus, this study aimed to identify potential hub genes and their important pathways and pathological mechanisms of interactions between IBD and BC using bioinformatics methods.
Methods
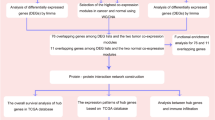
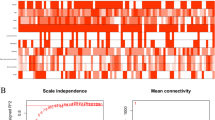
The data from Gene Expression Omnibus (GEO) and the cancer genome atlas (TCGA) were analyzed to screen common differentially expressed genes (DEGs) between IBD and BC. The “clusterProfiler” package was used to analyze GO term and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment in DEGs. After that, we conducted a weighted gene co-expression network analysis (WGCNA) on these DEGs to determine the vital modules and genes significantly related to BC. Protein–protein interaction (PPI) networks was used to identify hub genes. Further, the hub genes were used to develop a prognostic signature by Cox analysis. The validity of the ten hub DEGs was tested using three classification algorithms. Finally, we analyzed the microRNAs (miRNA)-mRNA, transcription factors (TFs)-mRNA regulatory network.
Results
Positive regulation of organelle fission, chromosomal region, tubulin binding, and cell cycle signaling pathway were the major enriched pathways for the common DEGs. PPI networks identified three hub proteins (AURKB, CDK1, and CCNA2) with high connectivity. Three machine-learning classification algorithms based on ten hub genes performed well for IBD and BC (accuracy > 0.80). The robust predictive model based on the ten hub genes could accurately classify BC cases with various clinical outcomes. Based on the gene-TFs and gene-miRNAs network construction, 9 TFs and 6 miRNAs were identified as potential critical TFs and miRNAs. There are 13 drugs that interact with the hub gene based on gene-drug interaction analysis.
Conclusions
This study explored common gene signatures and the potential pathogenesis of IBD and BC. We revealed that an unbalanced immune response, cell cycle pathway, and neutrophil infiltration might be the common pathogenesis of IBD and BC. Molecular mechanisms for the treatment of IBD and CC still require further investigation.













Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article. The data are not publicly available due to data protection reasons.
Abbreviations
- AUC:
-
Area under the curve
- AURKB:
-
Aurora kinases B
- BC:
-
Bladder cancer
- BP:
-
Biological processes
- CC:
-
Cellular component
- CD:
-
Crohn’s disease
- CDKs:
-
Cyclin‐dependent kinases
- DCA:
-
Decision curve analysis
- DEGs:
-
Differentially expressed genes
- DGIdb:
-
Drug Gene Interaction Database
- FC:
-
Foldchange
- FDR:
-
False Discovery Rate
- GEO:
-
Gene expression omnibus
- GO:
-
Gene ontology
- GS:
-
Gene significance
- HPA:
-
Human Protein Atlas
- IBD:
-
Inflammatory bowel disease
- IHC:
-
Immunohistochemistry
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- MCODE:
-
Molecular complex detection
- MDM2:
-
Mouse double minute 2
- ME:
-
Module eigengene
- MF:
-
Molecular function
- MM:
-
Module membership
- OS:
-
Overall survival
- PPI:
-
Protein–protein interaction
- ROC:
-
Receiver operating characteristic
- SVM:
-
Support vector machine
- TFCP2L1:
-
Transcription factor CP2‐like protein 1
- TFs:
-
Transcription factors
- TCGA:
-
The cancer genome atlas
- UALCAN:
-
University of ALabama at Birmingham CANcer data analysis Portal
- UC:
-
Ulcerative colitis
- WGCNA:
-
Weighted gene co-expression network analysis
References
Abedi Z, MotieGhader H, Hosseini SS, Sheikh Beig Goharrizi MA, Masoudi-Nejad A (2022) mRNA-miRNA bipartite networks reconstruction in different tissues of bladder cancer based on gene co-expression network analysis. Sci Rep 12(1):5885
Algaba A, Guerra I, Castaño A, de la Poza G, Castellano VM, López M et al (2013) Risk of cancer, with special reference to extra-intestinal malignancies, in patients with inflammatory bowel disease. World J Gastroenterol 19(48):9359–9365
Arooz T, Yam CH, Siu WY, Lau A, Li KK, Poon RY (2000) On the concentrations of cyclins and cyclin-dependent kinases in extracts of cultured human cells. Biochemistry 39(31):9494–9501
Axelrad JE, Lichtiger S, Yajnik V (2016) Inflammatory bowel disease and cancer: the role of inflammation, immunosuppression, and cancer treatment. World J Gastroenterol 22(20):4794–4801
Bancil AS, Sandall AM, Rossi M, Chassaing B, Lindsay JO, Whelan K (2021) Food additive emulsifiers and their impact on gut microbiome, permeability, and inflammation: mechanistic insights in inflammatory bowel disease. J Crohns Colitis 15(6):1068–1079
Baumgart DC, Carding SR (2007) Inflammatory bowel disease: cause and immunobiology. Lancet 369(9573):1627–1640
Belluti S, Semeghini V, Rigillo G, Ronzio M, Benati D, Torricelli F et al (2021) Alternative splicing of NF-YA promotes prostate cancer aggressiveness and represents a new molecular marker for clinical stratification of patients. J Exp Clin Cancer Res 40(1):362
Bendris N, Cheung CT, Leong HS, Lewis JD, Chambers AF, Blanchard JM et al (2014) Cyclin A2, a novel regulator of EMT. Cell Mol Life Sci 71(24):4881–4894
Biancone L, Armuzzi A, Scribano ML, Castiglione F, D’Incà R, Orlando A et al (2020) Cancer risk in inflammatory bowel disease: a 6-year prospective multicenter nested case-control IG-IBD study. Inflamm Bowel Dis 26(3):450–459
Bourrier A, Carrat F, Colombel JF, Bouvier AM, Abitbol V, Marteau P et al (2016) Excess risk of urinary tract cancers in patients receiving thiopurines for inflammatory bowel disease: a prospective observational cohort study. Aliment Pharmacol Ther 43(2):252–261
Burgess EF, Livasy C, Trufan S, Zhu J, O’Connor HF, Hartman A et al (2022) Clinical outcomes associated with expression of aurora kinase and p53 family members in muscle-invasive bladder cancer. Mol Clin Oncol 16(5):102
Chang JT (2020) Pathophysiology of Inflammatory Bowel Diseases. N Engl J Med 383(27):2652–2664
Cheng C, Varn FS, Marsit CJ (2015) E2F4 program is predictive of progression and intravesical immunotherapy efficacy in bladder cancer. Mol Cancer Res 13(9):1316–1324
Cheng J, Yang Q, Han X, Wang H, Wu K, Zhao H (2022) Yin Yang 1-stimulated long noncoding RNA bladder cancer-associated transcript 1 upregulation facilitates esophageal carcinoma progression via the microRNA-5590-3p/programmed cell death-ligand 1 pathway. Bioengineered 13(4):10244–10257
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420(6917):860–867
De Marzo AM, Platz EA, Sutcliffe S, Xu J, Grönberg H, Drake CG et al (2007) Inflammation in prostate carcinogenesis. Nat Rev Cancer 7(4):256–269
Gao Q, Fan L, Chen Y, Cai J (2022) Identification of the hub and prognostic genes in liver hepatocellular carcinoma via bioinformatics analysis. Front Mol Biosci 9:1000847
Geng Z, Geng Q (2021) Risk of urinary bladder cancer in patients with inflammatory bowel diseases: a meta-analysis. Front Surg 8:636791
González-Loyola A, Fernández-Miranda G, Trakala M, Partida D, Samejima K, Ogawa H et al (2015) Aurora B overexpression causes aneuploidy and p21Cip1 repression during tumor development. Mol Cell Biol 35(20):3566–3578
Greten FR, Grivennikov SI (2019) Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51(1):27–41
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140(6):883–899
Gully CP, Velazquez-Torres G, Shin J-H, Fuentes-Mattei E, Wang E, Carlock C et al (2012) Aurora B kinase phosphorylates and instigates degradation of p53. Proc Natl Acad Sci USA 109(24):E1513–E1522
Heo J, Noh B-J, Lee S, Lee H-Y, Kim Y, Lim J et al (2020) Phosphorylation of TFCP2L1 by CDK1 is required for stem cell pluripotency and bladder carcinogenesis. EMBO Mol Med 12(1):e10880
Heo J, Lee J, Nam YJ, Kim Y, Yun H, Lee S et al (2022) The CDK1/TFCP2L1/ID2 cascade offers a novel combination therapy strategy in a preclinical model of bladder cancer. Exp Mol Med 54(6):801–811
Hu Y, Pan J, Xin Y, Mi X, Wang J, Gao Q et al (2018) Gene expression analysis reveals novel gene signatures between young and old adults in human prefrontal cortex. Front Aging Neurosci 10:259
Jasek K, Kubatka P, Samec M, Liskova A, Smejkal K, Vybohova D et al (2019) DNA methylation status in cancer disease: modulations by plant-derived natural compounds and dietary interventions. Biomolecules 9(7):289
Jiang X, Li H, Fang Y, Xu C (2022) LncRNA PVT1 contributes to invasion and doxorubicin resistance of bladder cancer cells through promoting MDM2 expression and AURKB-mediated p53 ubiquitination. Environ Toxicol 37(6):1495–1508
Kandimalla R, van Tilborg AAG, Kompier LC, Stumpel DJPM, Stam RW, Bangma CH et al (2012) Genome-wide analysis of CpG island methylation in bladder cancer identified TBX2, TBX3, GATA2, and ZIC4 as pTa-specific prognostic markers. Eur Urol 61(6):1245–1256
Kaplan GG (2015) The global burden of IBD: from 2015 to 2025. Nat Rev Gastroenterol Hepatol 12(12):720–727
Kappelman MD, Farkas DK, Long MD, Erichsen R, Sandler RS, Sørensen HT et al (2014) Risk of cancer in patients with inflammatory bowel diseases: a nationwide population-based cohort study with 30 years of follow-up evaluation. Clin Gastroenterol Hepatol 12(2):265–273
Keller DS, Windsor A, Cohen R, Chand M (2019) Colorectal cancer in inflammatory bowel disease: review of the evidence. Tech Coloproctol 23(1):3–13
Kristensen SL, Lindhardsen J, Ahlehoff O, Erichsen R, Lamberts M, Khalid U et al (2013) Increased risk of atrial fibrillation and stroke during active stages of inflammatory bowel disease: a nationwide study. EP Europace 16(4):477–484
Laish I, Biron-Shental T, Katz H, Liberman M, Kitay-Cohen Y, Konikoff FM et al (2015) Asynchronous replication in lymphocytes from patients with inflammatory bowel disease and primary sclerosing cholangitis. Cytogenet Genome Res 145(1):35–41
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9:559
Li J, Ying Y, Xie H, Jin K, Yan H, Wang S et al (2019) Dual regulatory role of CCNA2 in modulating CDK6 and MET-mediated cell-cycle pathway and EMT progression is blocked by miR-381-3p in bladder cancer. FASEB J 33(1):1374–1388
Li Y, Sun C, Tan Y, Li L, Zhang H, Liang Y et al (2020) Transcription levels and prognostic significance of the NFI family members in human cancers. PeerJ 8:e8816
Li P, Zhang H-Y, Gao J-Z, Du W-Q, Tang D, Wang W et al (2022) Mesenchymal stem cells-derived extracellular vesicles containing miR-378a-3p inhibit the occurrence of inflammatory bowel disease by targeting GATA2. J Cell Mol Med 26(11):3133–3146
Lin J-T, Tsai K-W (2021) Circulating miRNAs act as diagnostic biomarkers for bladder cancer in urine. Int J Mol Sci 22(8):4278
Liu Y, Huang X, Guo L, Luo N (2022) LINC00649 facilitates the cellular process of bladder cancer cells via signaling axis miR-16-5p/JARID2. Urol Int 106(3):304–312
Lu Y-T, Xu T, Iqbal M, Hsieh T-C, Luo Z, Liang G et al (2022) FOXC1 binds enhancers and promotes cisplatin resistance in bladder cancer. Cancers 14(7):1717
Luo W, Wang J, Xu W, Ma C, Wan F, Huang Y et al (2021) LncRNA RP11-89 facilitates tumorigenesis and ferroptosis resistance through PROM2-activated iron export by sponging miR-129-5p in bladder cancer. Cell Death Dis 12(11):1043
Lv S, Liu L, Yang B, Zhao X (2022) Association of miR-9-5p and NFIC in the progression of gastric cancer. Hum Exp Toxicol 41:9603271221084672
Madanchi M, Zeitz J, Barthel C, Samaras P, Scharl S, Sulz MC et al (2016) Malignancies in patients with inflammatory bowel disease: a single-centre experience. Digestion 94(1):1–8
Magzoub MM, Prunello M, Brennan K, Gevaert O (2019) The impact of DNA methylation on the cancer proteome. PLoS Comput Biol 15(7):e1007245
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454(7203):436–444
Mark-Christensen A, Erichsen R, Veres K, Laurberg S, Sørensen HT (2020) Extracolonic cancer risk after total colectomy for inflammatory bowel disease: a population-based cohort study. J Crohns Colitis 14(5):630–635
Matthews HK, Bertoli C, de Bruin RAM (2022) Cell cycle control in cancer. Nat Rev Mol Cell Biol 23(1):74–88
Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI et al (2017) Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390(10114):2769–2778
Nguyen TB, Do DN, Nguyen-Thi M-L, Hoang-The H, Tran T-T, Nguyen-Thanh T (2022a) Identification of potential crucial genes and key pathways shared in Inflammatory Bowel Disease and cervical cancer by machine learning and integrated bioinformatics. Comput Biol Med 149:105996
Nguyen TB, Do DN, Nguyen TTP, Nguyen TL, Nguyen-Thanh T, Nguyen HT (2022b) Immune-related biomarkers shared by inflammatory bowel disease and liver cancer. PLoS One 17(4):e0267358
Nikonova AS, Astsaturov I, Serebriiskii IG, Dunbrack RL, Golemis EA (2013) Aurora A kinase (AURKA) in normal and pathological cell division. Cell Mol Life Sci 70(4):661–687
Pedersen N, Duricova D, Elkjaer M, Gamborg M, Munkholm P, Jess T (2010) Risk of extra-intestinal cancer in inflammatory bowel disease: meta-analysis of population-based cohort studies. Am J Gastroenterol 105(7):1480–1487
Prencipe M, Fabre A, Murphy TB, Vargyas E, O’Neill A, Bjartell A et al (2018) Role of serum response factor expression in prostate cancer biochemical recurrence. Prostate 78(10):724–730
Rogler G, Singh A, Kavanaugh A, Rubin DT (2021) Extraintestinal manifestations of inflammatory bowel disease: current concepts, treatment, and implications for disease management. Gastroenterology 161(4):1118–1132
Tian Z, Cao S, Li C, Xu M, Wei H, Yang H et al (2019) LncRNA PVT1 regulates growth, migration, and invasion of bladder cancer by miR-31/ CDK1. J Cell Physiol 234(4):4799–4811
van Kessel KEM, van der Keur KA, Dyrskjøt L, Algaba F, Welvaart NYC, Beukers W et al (2018) Molecular markers increase precision of the european association of urology non-muscle-invasive bladder cancer progression risk groups. Clin Cancer Res 24(7):1586–1593
Wang D, Liu J, Liu S, Li W (2020a) Identification of crucial genes associated with immune cell infiltration in hepatocellular carcinoma by weighted gene co-expression network analysis. Front Genet 11:342
Wang H, Shi X, Wu S (2020b) miR-550a-3/NFIC plays a driving role in esophageal squamous cell cancer cells proliferation and metastasis partly through EMT process. Mol Cell Biochem 472(1–2):115–123
Wang J, Peng X, Li R, Liu K, Zhang C, Chen X et al (2021) Evaluation of serum miR-17-92 cluster as noninvasive biomarkers for bladder cancer diagnosis. Front Oncol 11:795837
Wilson JC, Furlano RI, Jick SS, Meier CR (2015) Inflammatory bowel disease and the risk of autoimmune diseases. J Crohns Colitis 10(2):186–193
Xu G, Zhang Y, Li N, Wu Y, Zhang J, Xu R et al (2020) LBX2-AS1 up-regulated by NFIC boosts cell proliferation, migration and invasion in gastric cancer through targeting miR-491-5p/ZNF703. Cancer Cell Int 20:136
Yi L, Wang H, Li W, Ye K, Xiong W, Yu H et al (2021) The FOXM1/RNF26/p57 axis regulates the cell cycle to promote the aggressiveness of bladder cancer. Cell Death Dis 12(10):944
Zeng B, Zhao Q, Sun Z, Liu D, Chen H, Li X et al (2021) SEC23A is an independent prognostic biomarker in bladder cancer correlated with MAPK signaling. Front Genet 12:672832
Zhang C, Wang W, Lin J, Xiao J, Tian Y (2019) lncRNA CCAT1 promotes bladder cancer cell proliferation, migration and invasion. Int Braz J Urol 45(3):549–559
Zhang M, Xu Y, Yin S, Qiu F (2021) YY1-induced long non-coding RNA PSMA3 antisense RNA 1 functions as a competing endogenous RNA for microRNA 214–5p to expedite the viability and restrict the apoptosis of bladder cancer cells via regulating programmed cell death-ligand 1. Bioengineered 12(2):9150–9161
Zhao J, Han S-X, Ma J-L, Ying X, Liu P, Li J et al (2013) The role of CDK1 in apoptin-induced apoptosis in hepatocellular carcinoma cells. Oncol Rep 30(1):253–259
Zheng L, Xu H, Di Y, Chen L, Liu J, Kang L et al (2021) ELK4 promotes the development of gastric cancer by inducing M2 polarization of macrophages through regulation of the KDM5A-PJA2-KSR1 axis. J Transl Med 19(1):342
Funding
The study was funded by the National Key Research and Development Program of China (Grant Numbers: 2018YFC2002202) and the Chinese Academy of Medical Sciences (Grant Numbers: BJ-2022-237).
Author information
Authors and Affiliations
Contributions
JL: project development, data collection, data analysis, manuscript writing. PW: data collection, data analysis, manuscript editing. SL: data collection, data analysis, manuscript editing. Jianye Wang: project development, data analysis, manuscript editing. Jianlong Wang: project development, data analysis, manuscript editing. YZ: project development, data analysis, manuscript editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Informed consent statement
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Wu, P., Lai, S. et al. Identifying possible hub genes and biological mechanisms shared between bladder cancer and inflammatory bowel disease using machine learning and integrated bioinformatics. J Cancer Res Clin Oncol 149, 16885–16904 (2023). https://doi.org/10.1007/s00432-023-05266-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05266-0