Abstract
Purpose
To explore the optimal timing of locoregional therapy in patients with colorectal cancer (CRC) recurrence after radical resection and initially unresectable liver metastases but no other metastases and whether maintenance therapy should be performed after achieving no evidence of disease (NED).
Methods
This study was jointly carried out in six medical institutions in China to collect the clinical data of patients with primary CRC from January 2015 to December 2021. Research participants were identified according to the inclusion criteria of this study for statistical analysis of the clinical characteristics and recurrence time.
Results
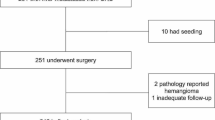
625 patients CRC with metachronous initially unresectable liver metastases but no other metastases were enrolled. Multivariate analysis showed that the number of metastases in the liver and the time from the start of first-line chemotherapy to locoregional therapy significantly affected the progression-free survival (PFS, P < 0.05) following the first-line treatment, and continued maintenance therapy reduced the risk of tumor progression in the patients (P < 0.05). Furthermore, stratified analysis showed that the median PFS of patients with 3–5 metastases in the liver was maximized when the time from the start of first-line chemotherapy to locoregional therapy was 3–4 months. Patients with > 6 metastases in the liver should extend the duration between the start of first-line chemotherapy and locoregional therapy to more than four months. Similarly, with the significant increase in the number of metastases in the liver, subsequent maintenance therapy significantly extended the PFS of the patients.
Conclusions
The overall therapeutic plan in patients with CRC recurrence after radical resection and initially unresectable liver metastases but no other metastases should consider the individual patients' situations, especially the number of metastases in the liver at initial recurrence.


Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Chibaudel B, Maindrault-Goebel F, Lledo G, Mineur L, André T, Bennamoun M, Mabro M, Artru P, Carola E, Flesch M (2009) Can chemotherapy be discontinued in unresectable metastatic colorectal cancer? The GERCOR OPTIMOX2 study. J Clin Oncol 27(34):5727–5733
Guler I, Askan G, Klostergaard J, Sahin IH (2019) Precision medicine for metastatic colorectal cancer: an evolving era. Expert Rev Gastroenterol Hepatol 13(10):919–931
Margonis GA, Buettner S, Andreatos N, Wagner D, Sasaki K, Barbon C, Beer A, Kamphues C, Løes IM, He J (2019) Prognostic factors change over time after hepatectomy for colorectal liver metastases: a multi-institutional, international analysis of 1099 patients. Ann Surg 269(6):1129–1137
Mikhail S, Bekaii-Saab T (2015) Maintenance therapy for colorectal cancer: which regimen and which patients? Drugs 75(16):1833–1842
Owen JW, Fowler KJ, Doyle MB, Saad NE, Linehan DC, Chapman WC (2016) Colorectal liver metastases: disappearing lesions in the era of eovist hepatobiliary magnetic resonance imaging. HPB 18(3):296–303
Petrelli F, Trevisan F, Cabiddu M, Sgroi G, Bruschieri L, Rausa E, Ghidini M, Turati L (2020) Total neoadjuvant therapy in rectal cancer: a systematic review and meta-analysis of treatment outcomes. Ann Surg 271(3):440–448
Siegel RL, Miller KD, Goding Sauer A, Fedewa SA, Butterly LF, Anderson JC, Cercek A, Smith RA, Jemal A (2020) (2020) Colorectal cancer statistics. CA Cancer J Clin 70(3):145–164
Simkens LH, Van Tinteren H, May A, Ten Tije AJ, Creemers G-JM, Loosveld OJ, De Jongh FE, Erdkamp FL, Erjavec Z, Van Der Torren AM (2015) Maintenance treatment with capecitabine and bevacizumab in metastatic colorectal cancer (CAIRO3): a phase 3 randomised controlled trial of the Dutch colorectal cancer group. Lancet 385(9980):1843–1852
Sonbol MB, Mountjoy LJ, Firwana B, Liu AJ, Almader-Douglas D, Mody K, Hubbard J, Borad M, Ahn DH, Murad MH (2020) The role of maintenance strategies in metastatic colorectal cancer: a systematic review and network meta-analysis of randomized clinical trials. JAMA Oncol 6(3):e194489–e194489
Song Z-Y, Yang D, Liu Y, Cheng Y (2022) The influence of radiological “disappearing lesions” on the efficacy and prognosis of patients with colorectal liver metastases undergoing conversion therapy. Gastroenterol Res Pract 2022:2200598
Tani K, Shindoh J, Akamatsu N, Arita J, Kaneko J, Sakamoto Y, Hasegawa K, Kokudo N (2018) Management of disappearing lesions after chemotherapy for colorectal liver metastases: relation between detectability and residual tumors. J Surg Oncol 117(2):191–197
Von Minckwitz G, Schwedler K, Schmidt M, Barinoff J, Mundhenke C, Cufer T, Maartense E, De Jongh FE, Baumann KH, Bischoff J (2011) Trastuzumab beyond progression: overall survival analysis of the GBG 26/BIG 3–05 phase III study in HER2-positive breast cancer. Eur J Cancer 47(15):2273–2281
Xu RH, Shen L, Li J, Xu JM, Bi F, Ba Y, Bai L, Shu YQ, Liu TS, Li YH (2016) Expert consensus on maintenance treatment for metastatic colorectal cancer in China. Cancer Commun 35(1):1–6
Xu Z, Xie H, Zhou L, Chen X, Zheng S (2019) The combination strategy of transarterial chemoembolization and radiofrequency ablation or microwave ablation against hepatocellular carcinoma. Anal Cell Pathol 2019:8619096
Yoshita H, Hosokawa A, Ueda A, Ando T, Kajiura S, Kato H, Kawabe H, Tomizawa G, Horikawa N, Yabuhita K (2014) Predictive value of optimal morphologic response to first-line chemotherapy in patients with colorectal liver metastases. Digestion 89(1):43–48
Zendel A, Lahat E, Dreznik Y, Zakai BB, Eshkenazy R, Ariche A (2014) “Vanishing liver metastases”—a real challenge for liver surgeons. Hepatobiliary Surg Nutr 3(5):295–302
Zizzo M, Galeone C, Braglia L, Ugoletti L, Siciliani A, Nachira D, Margaritora S, Pedrazzoli C, Paci M, Lococo F (2020) Long-term outcomes after surgical resection for synchronous or metachronous hepatic and pulmonary colorectal cancer metastases. Digestion 101(2):144–155
Funding
This work was supported by Beijing Xisike Clinical Oncology Research Foundation (No: Y-HR2018-321).
Author information
Authors and Affiliations
Contributions
LB took part in conception and design. YJQ, YDJ, YY, FXS, and PG took part in collection and assembly of clinical data. SSY and TTZ participated in data analysis and involved in manuscript writing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Ethical approval
This retrospective study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of The People's Liberation Army General Hospital (2/23/2017/No.S2017-009-01).
Consent to participate
Not applicable.
Consent for publishing
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ye, S., Bai, L., Qi, Y. et al. Retrospective analysis of colorectal cancer patients with metachronous initially unresectable liver metastases (and no other) achieving no evidence of disease after first-line comprehensive therapy: a multicenter real-world study. J Cancer Res Clin Oncol 149, 8403–8413 (2023). https://doi.org/10.1007/s00432-023-04768-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04768-1