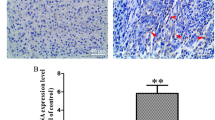
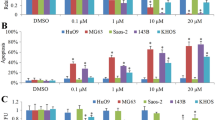
Abstract
Purpose
Although undergoing conventional chemotherapy significantly improves the prognosis of Osteosarcoma, chemoresistance and failure of therapy is still a significant challenge. Furthermore, conventional chemotherapy, like doxorubicin, would upregulate the expression of programmed death-ligand 1 (PD-L1) which caused an immunosuppressive microenvironment and unsatisfied treatment result in Osteosarcoma. Thus, it is urgent to explore a strategy to overcome this disadvantage.
Methods
Human Osteosarcoma cell line MG63 and mouse Osteosarcoma cell line K7 were included in this study. Subcutaneous tumor model was used by injection of K7 cells in BALB/C mice to test the effect of doxorubicin and sorafenib on tumor growth. PD-L1 expression was tested in vitro (flow cytometry, western blot and PCR) and in vivo (flow cytometry and immunohistochemistry). Proportion of immune cells (CD4, CD8, Tregs, and cytotoxic T lymphocytes) in vivo was analyzed with flow cytometry.
Results
Combination of sorafenib and doxorubicin inhibited tumor growth significantly in vivo. Doxorubicin increased PD-L1 expression in vitro and in vivo, while sorafenib inhibited doxorubicin-induced PD-L1 upregulation in vitro and in vivo. Proportion of interferon-γ-secreting CD8 + T lymphocytes in tumor tissue was increased significantly when sorafenib was combined with doxorubicin, while proportion of CD4, CD8, and Tregs was not significantly changed. Extracellular signal-regulated kinases (ERK) pathway could be one of the key mechanisms by which doxorubicin induced upregulation of PD-L1 in Osteosarcoma cells.
Conclusion
Combination of sorafenib and conventional chemotherapeutic reagents is a potent strategy to improve treatment effectiveness by modulating tumor microenvironment in Osteosarcoma through increasing proportion of cytotoxic T lymphocytes.






Similar content being viewed by others
Availability of data and materials
The data generated in this study are available upon request from the corresponding author.
References
Alizadeh D, Trad M, Nt H, Cb L, Janikashvili N, Bonnotte B, Katsanis E, Larmonier N (2014) Doxorubicin eliminates myeloid-derived suppressor cells and enhances the efficacy of adoptive T-cell transfer in breast cancer. Can Res 74(1):104–118. https://doi.org/10.1158/0008-5472.can-13-1545
Bielack S, Carrle D, Casali PG, Working GEG (2009) Osteosarcoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 20:137–139. https://doi.org/10.1093/annonc/mdp154
Biermann JS, Adkins DR, Agulnik M, Benjamin RS, Brigman B, Butrynski JE, Cheong D, Chow W, Curry WT, Frassica DA, Frassica FJ, Hande KR, Hornicek FJ, Jones RL, Mayerson J, Mcgarry SV, Mcgrath B, Morris CD, O’donnell RJ, Randall RL, Santana VM, Satcher RL, Siegel HJ, Von Mehren M, Bergman MA, Sundar H (2013) Bone cancer clinical practice guidelines in oncology. J Natl Comphr Cancer Netw 11(6):688–723
Chen DS, Mellman I (2013) Oncology meets immunology: the cancer-immunity cycle. Immunity 39(1):1–10. https://doi.org/10.1016/j.immuni.2013.07.012
Chen Y, Ma DG, Molyneux S, Mckee T, Waterhouse P, Jm P, Khokha R (2015) RANKL blockade prevents and treats aggressive osteosarcomas. Sci Transl Med 7(317):317ra197. https://doi.org/10.1126/scitranslmed.aad0295
Chou AJ, Gorlick R (2006) Chemotherapy resistance in osteosarcoma: current challenges and future directions. Expert Rev Anticancer Ther 6(7):1075–1085. https://doi.org/10.1586/14737140.6.7.1075
Coventon J (2017) A review of the mechanism of action and clinical applications of sorafenib in advanced osteosarcoma. J Bone Oncol 8:4–7. https://doi.org/10.1016/j.jbo.2017.07.001
Ding Zc LuX, Yu M, Lemos H, Huang L, Chandler P, Liu K, Walters M, Krasinski A, Mack M, Br B, Al M, Munn Dh, Zhou G (2014) Immunosuppressive myeloid cells induced by chemotherapy attenuate antitumor CD4+ T-cell responses through the PD-1-PD-L1 axis. Can Res 74(13):3441–3453. https://doi.org/10.1158/0008-5472.can-13-3596
Francisco Lm Salinas Vh, Ke B, Vk V, Gj F, Vk K, Sharpe Ah (2009) PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med 206(13):3015–3029
Ghebeh H, Lehe C, Barhoush E, Al-Romaih K, Tulbah A, Al-Alwan M, Hendrayani SF, Manogaran P, Alaiya A, Al-Tweigeri T, Aboussekhra A, Dermime S (2010) Doxorubicin downregulates cell surface B7–H1 expression and upregulates its nuclear expression in breast cancer cells: role of B7–H1 as an anti-apoptotic molecule. Breast Cancer Res 12(4):R48. https://doi.org/10.1186/bcr2605
Grignani G, Palmerini E, Dileo P, Asaftei SD, D’ambrosio L, Pignochino Y, Mercuri M, Picci P, Fagioli F, Casali PG, Ferrari S, Aglietta M (2012) A phase II trial of sorafenib in relapsed and unresectable high-grade osteosarcoma after failure of standard multimodal therapy: an Italian Sarcoma Group study. Ann Oncol 23(2):508–516. https://doi.org/10.1093/annonc/mdr151
Hannesdóttir L, Tymoszuk P, Parajuli N, Wasmer Mh, Philipp S, Daschil N, Datta S, Jb K, Tripp Ch, Stoitzner P, Müller-Holzner E, Gj W, Sexl V, Villunger A, Doppler W (2013) Lapatinib and doxorubicin enhance the Stat1-dependent antitumor immune response. Eur J Immunol 43(10):2718–2729. https://doi.org/10.1002/eji.201242505
Hu C, Chen X, Wen J, Gong L, Liu Z, Wang J, Liang J, Hu F, Zhou Q, Wei L, Shen Y, Zhang W (2017) Antitumor effect of focal adhesion kinase inhibitor PF562271 against human osteosarcoma in vitro and in vivo. Cancer Sci 108(7):1347–1356. https://doi.org/10.1111/cas.13256
Jaffe N (2014) Historical perspective on the introduction and use of chemotherapy for the treatment of osteosarcoma. Curr Adv Osteosarcoma 804:1–30. https://doi.org/10.1007/978-3-319-04843-7_1
Jayson GC, Kerbel R, Ellis LM, Harris AL (2016) Antiangiogenic therapy in oncology: current status and future directions. Lancet 388(10043):518–529
Jeon DG, Song WS (2010) How can survival be improved in localized osteosarcoma? Expert Rev Anticancer Ther 10(8):1313–1325. https://doi.org/10.1586/era.10.79
Kalathil SG, Hutson HA, Barbi J, Iyer R, Thanavala Y (2019) Augmentation of IFN-γ+ CD8+ T cell responses correlates with survival of HCC patients on sorafenib therapy. JCI Insight. https://doi.org/10.1172/jci.insight.130116
Lastwika KJ, Wilson W 3rd, Li QK, Norris J, Xu H, Ghazarian SR, Kitagawa H, Kawabata S, Taube JM, Yao S, Liu LN, Gills JJ, Dennis PA (2016) Control of PD-L1 expression by oncogenic activation of the AKT-mTOR pathway in non-small cell lung cancer. Cancer Res 76(2):227–238. https://doi.org/10.1158/0008-5472.CAN-14-3362
Liu J, Liu Y, Meng L, Liu K, Ji B (2017) Targeting the PD-L1/DNMT1 axis in acquired resistance to sorafenib in human hepatocellular carcinoma. Oncol Rep 38(2):899–907. https://doi.org/10.3892/or.2017.5722
Liu Y, Leng A, Li L, Yang B, Shen S, Chen H, Zhu E, Xu Q, Ma X, Shi P, Liu Y, Liu T, Li L, Li K, Zhang D, Xiao J (2022) AMTB, a TRPM8 antagonist, suppresses growth and metastasis of osteosarcoma through repressing the TGFβ signaling pathway. Cell Death Dis 13(3):288. https://doi.org/10.1038/s41419-022-04744-6
Luetke A, Meyers PA, Lewis I, Juergens H (2014) Osteosarcoma treatment—where do we stand? A state of the art review. Cancer Treat Rev 40(4):523–532. https://doi.org/10.1016/j.ctrv.2013.11.006
Ma Q, Zhang Y, Liu T, Jiang K, Wen Y, Fan Q, Qiu X (2017) Hypoxia promotes chemotherapy resistance by down-regulating SKA1 gene expression in human osteosarcoma. Cancer Biol Ther 18(3):177–185. https://doi.org/10.1080/15384047.2017.1294285
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454(7203):436–444. https://doi.org/10.1038/nature07205
Martin-Broto J, Redondo A, Valverde C, Ma V, Mora J, Garcia Del Muro X, Gutierrez A, Tous C, Carnero A, Marcilla D, Carranza A, Sancho P, Martinez-Trufero J, Diaz-Beveridge R, Cruz J, Encinas V, Taron M, Ds M, Luna P, Hindi N, Lopez-Pousa A (2017) Gemcitabine plus sirolimus for relapsed and progressing osteosarcoma patients after standard chemotherapy: a multicenter, single-arm phase II trial of Spanish Group for Research on Sarcoma (GEIS). Ann Oncol 28(12):2994–2999. https://doi.org/10.1093/annonc/mdx536
Meltzer PS, Helman LJ (2021) New horizons in the treatment of osteosarcoma. N Engl J Med 385(22):2066–2076. https://doi.org/10.1056/NEJMra2103423
Moore EC, Cash HA, Caruso AM, Uppaluri R, Hodge JW, Van Waes C, Allen CT (2016) Enhanced tumor control with combination mTOR and PD-L1 inhibition in syngeneic oral cavity cancers. Cancer Immunol Res 4(7):611–620. https://doi.org/10.1158/2326-6066.CIR-15-0252
Muraro E, Furlan C, Avanzo M, Martorelli D, Comaro E, Rizzo A, Fae DA, Berretta M, Militello L, Del Conte A, Spazzapan S, Dolcetti R, Trovo M (2017) Local high-dose radiotherapy induces systemic immunomodulating effects of potential therapeutic relevance in oligometastatic breast cancer. Front Immunol 8:1476. https://doi.org/10.3389/fimmu.2017.01476
Palmerini E, Agostinelli C, Picci P, Pileri S, Marafioti T, Lollini PL, Scotlandi K, Longhi A, Benassi MS, Ferrari S (2017) Tumoral immune-infiltrate (IF), PD-L1 expression and role of CD8/TIA-1 lymphocytes in localized osteosarcoma patients treated within protocol ISG-OS1. Oncotarget 8(67):111836–111846. https://doi.org/10.18632/oncotarget.22912
Park JA, Cheung NV (2020) GD2 or HER2 targeting T cell engaging bispecific antibodies to treat osteosarcoma. J Hematol Oncol 13(1):172. https://doi.org/10.1186/s13045-020-01012-y
Peng J, Hamanishi J, Matsumura N, Abiko K, Murat K, Baba T, Yamaguchi K, Horikawa N, Hosoe Y, Sk M, Konishi I, Mandai M (2015) Chemotherapy induces programmed cell death-ligand 1 overexpression via the nuclear factor-κB to foster an immunosuppressive tumor microenvironment in ovarian cancer. Can Res 75(23):5034–5045. https://doi.org/10.1158/0008-5472.can-14-3098
Qin X, Liu C, Zhou Y, Wang G (2010) Cisplatin induces programmed death-1-ligand 1(PD-L1) over-expression in hepatoma H22 cells via Erk /MAPK signaling pathway. Cell Mol Biol 56:OL1366-1372
Sundara YT, Kostine M, Cleven AH, Bovee JV, Schilham MW, Cleton-Jansen AM (2017) Increased PD-L1 and T-cell infiltration in the presence of HLA class I expression in metastatic high-grade osteosarcoma: a rationale for T-cell-based immunotherapy. Cancer Immunol Immunother 66(1):119–128. https://doi.org/10.1007/s00262-016-1925-3
Takemoto A, Takagi S, Ukaji T, Gyobu N, Kakino M, Takami M, Kobayashi A, Lebel M, Kawaguchi T, Sugawara M, Tsuji-Takayama K, Ichihara K, Funauchi Y, Ae K, Matsumoto S, Sugiura Y, Takeuchi K, Noda T, Katayama R, Fujita N (2022) Targeting podoplanin for the treatment of osteosarcoma. Clin Cancer Res. https://doi.org/10.1158/1078-0432.ccr-21-4509
Wang J, Hu C, Wang J, Shen Y, Bao Q, He F, Wang H, Gong L, Liu Z, Hu F, Liang J, Zhou Q, Wei L, Wen J, Zhang W (2019) Checkpoint blockade in combination with doxorubicin augments tumor cell apoptosis in osteosarcoma. J Immunother 42(9):321–330. https://doi.org/10.1097/CJI.0000000000000281
Wilhelm SM, Carter C, Tang L, Wilkie D, Mcnabola A, Rong H, Chen C, Zhang X, Vincent P, Mchugh M, Cao Y, Shujath J, Gawlak S, Eveleigh D, Rowley B, Liu L, Adnane L, Lynch M, Auclair D, Taylor I, Gedrich R, Voznesensky A, Riedl B, Post LE, Bollag G, Trail PA (2004) BAY 43–9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64(19):7099–7109. https://doi.org/10.1158/0008-5472.CAN-04-1443
Yang M, Liu P, Wang K, Glorieux C, Hu Y, Wen S, Jiang W, Huang P (2017) Chemotherapy induces tumor immune evasion by upregulation of programmed cell death ligand 1 expression in bone marrow stromal cells. Mol Oncol 11(4):358–372. https://doi.org/10.1002/1878-0261.12032
Yang YT, Vilma YG (2022) Sorafenib and Doxorubicin Show Synergistic Effects in Human and Canine Osteosarcoma Cell Lines. Int J Mol Sci 23(16): 9345
Zappasodi R, Sm P, Gc G, Bongarzone I, Magni M, Cabras Ad, Mp C, Carlo-Stella C, Am G, Di Nicola M (2010) Improved clinical outcome in indolent B-cell lymphoma patients vaccinated with autologous tumor cells experiencing immunogenic death. Can Res 70(22):9062–9072. https://doi.org/10.1158/0008-5472.can-10-1825
Zhang P, Su DM, Liang M, Fu J (2008) Chemopreventive agents induce programmed death-1-ligand 1 (PD-L1) surface expression in breast cancer cells and promote PD-L1-mediated T cell apoptosis. Mol Immunol 45(5):1470–1476. https://doi.org/10.1016/j.molimm.2007.08.013
Zhang J, Hu J, Chan HF, Skibba M, Liang G, Chen M (2016) iRGD decorated lipid-polymer hybrid nanoparticles for targeted co-delivery of doxorubicin and sorafenib to enhance anti-hepatocellular carcinoma efficacy. Nanomedicine 12(5):1303–1311. https://doi.org/10.1016/j.nano.2016.01.017
Zheng B, Ren T, Huang Y, Guo W (2018) Apatinib inhibits migration and invasion as well as PD-L1 expression in osteosarcoma by targeting STAT3. Biochem Biophys Res Commun 495(2):1695–1701. https://doi.org/10.1016/j.bbrc.2017.12.032
Acknowledgements
Thanks to Dr Zhengdong Cai for generously supporting of mouse Osteosarcoma cell line K7. This work was supported by the National Nature Science Foundation of China (No.81702661).
Funding
This work was supported by the National Nature Science Foundation of China (No. 81702661).
Author information
Authors and Affiliations
Contributions
JXW and QYB conceived and designed the experiment. JZW, FQH and PY performed the experiments and wrote the manuscript draft. JXW, JW and ZCL edited the paper. All the authors have read and approved the final manuscript and agree to be accountable for all aspects of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. The protocol was approved by the animal ethical committee of Ruijin Hospital, affiliated with Shanghai Jiaotong University School of Medicine.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2022_4458_MOESM1_ESM.tif
Supplementary file1 Supplemental Fig. 1 Western blotting analysis of PD-L1, phosphorylation level of ERK, mTOR, and Stat3 pathways was implemented in 143b and U2 cell lines. (TIF 1827 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Hu, F., Yu, P. et al. Sorafenib inhibits doxorubicin-induced PD-L1 upregulation to improve immunosuppressive microenvironment in Osteosarcoma. J Cancer Res Clin Oncol 149, 5127–5138 (2023). https://doi.org/10.1007/s00432-022-04458-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04458-4