Abstract
Background
PD-1/PD-L1 inhibitors prolong survival in treatment-naïve, locally advanced, and metastatic non-small cell lung cancer (NSCLC) with positive PD-L1 expression (> 1%/ > 50%). Recent evidence has suggested that tumors with < 1% PD-L1 expression may also be predictive of PD-1/PD-L1 inhibiting agents.
Methods
Systematic review and meta-analysis were conducted of randomized controlled trials (RCTs) evaluating PD-1/PD-L1 inhibitors that have assessed tumors with < 1% PD-L1 expression (negative PD-L1 expression). PD-1/PD-L1 inhibitors–chemotherapy combinations (PC) were compared with histology-selected chemotherapy with respect to overall survival (OS) and progression-free survival (PFS).
Results
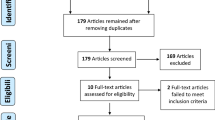
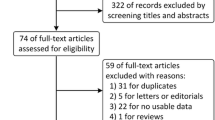
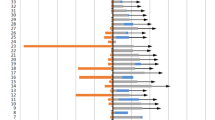
Twelve RCTs comprising 5410 participants (PD-1/PD-L1 inhibitors–chemotherapy: 3051; chemotherapy: 2359) met the inclusion criteria. Tumors with PD-L1 expression < 1% were evident in 38.9% of the pooled study population. A significant OS [hazard ratio (HR) 0.71 95% confidence interval (CI) 0.63–0.80, p < 0.00001] and PFS [HR 0.65 95% CI 0.58–0.72, p < 0.00001] benefit of PC was evident in tumors with negative PD-L1 expression. PD-1/PD-L1 inhibitors–chemotherapy combinations were more likely to achieve an objective response than chemotherapy [odds ratio, 1.86; 95% CI, 1.46–2.38, p < 0.00001]. Histologic subtypes and diagnostic assays did not modify the OS and PFS treatment effects for PC compared to chemotherapy.
Conclusion
Tumors harboring < 1% PD-L1 expression are likely to benefit from PD-1/PD-L1 inhibitor–chemotherapy regimens in advanced NSCLC.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available in the Open Science Framework repository, https://osf.io/kzqjg/ without third-party permission.
References
Adam J, Le Stang N, Rouquette I et al (2018) Multicenter harmonization study for PD-L1 IHC testing in non-small-cell lung cancer. Ann Oncol 29(4):953–958. https://doi.org/10.1093/annonc/mdy014
Awad MM, Gadgeel SM, Borghaei H et al (2021) Long-term overall survival from KEYNOTE-021 cohort g: pemetrexed and carboplatin with or without pembrolizumab as first-line therapy for advanced nonsquamous NSCLC. J Thorac Oncol 16(1):162–168. https://doi.org/10.1016/j.jtho.2020.09.015
Casarrubios M, Cruz-Bermúdez A, Nadal E et al (2021) Pretreatment tissue TCR repertoire evenness is associated with complete pathologic response in patients with NSCLC receiving neoadjuvant chemoimmunotherapy. Clin Cancer Res 27(21):5878–5890. https://doi.org/10.1158/1078-0432.CCR-21-1200
Chae YK, Pan A, Davis AA et al (2016) Biomarkers for PD-1/PD-L1 blockade therapy in non–small-cell lung cancer: is PD-L1 expression a good marker for patient selection? Clin Lung Cancer 17(5):350–361. https://doi.org/10.1016/j.cllc.2016.03.011
Chae YK, Arya A, Iams W et al (2018) Current landscape and future of dual anti-CTLA4 and PD-1/PD-L1 blockade immunotherapy in cancer; lessons learned from clinical trials with melanoma and non-small cell lung cancer (NSCLC). J Immunother Cancer 6(1):1–27
Chen J, Liu X, Zhang J et al (2022) Frontline anti-PD-1/PD-L1 versus bevacizumab in advanced non-small-cell lung cancer: a network meta-analysis. Futur Oncol 18(13):1651–1664. https://doi.org/10.2217/fon-2021-0838
Duval S, Tweedie R (2000) Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56(2):455–463. https://doi.org/10.1111/j.0006-341x.2000.00455.x
Egger M, Smith GD, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315(7109):629–634
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Ettinger DS, Wood DE, Aisner DL et al (2021) NCCN guidelines insights: non-small cell lung cancer, version 2.2021. J Natl Compr Canc Netw 19(3):254–266. https://doi.org/10.6004/jnccn.2021.0013
European Medicines Agency. Imfizi: EPAR – Public Assessment Report.; 2018.
Gandhi L, Rodríguez-Abreu D, Gadgeel S et al (2018) Pembrolizumab plus chemotherapy in metastatic non–small-cell lung cancer. N Engl J Med 378(22):2078–2092. https://doi.org/10.1056/nejmoa1801005
Grant MJ, Herbst RS, Goldberg SB (2021) Selecting the optimal immunotherapy regimen in driver-negative metastatic NSCLC. Nat Rev Clin Oncol 18(10):625–644. https://doi.org/10.1038/s41571-021-00520-1
Hellmann MD, Ciuleanu T-E, Pluzanski A et al (2018) Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med 378(22):2093–2104. https://doi.org/10.1056/NEJMoa1801946
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Hofman P (2017) PD-L1 immunohistochemistry for non-small cell lung carcinoma: which strategy should be adopted? Expert Rev Mol Diagn 17(12):1097–1108. https://doi.org/10.1080/14737159.2017.1398083
Hofman P, Heeke S, Alix-Panabières C, Pantel K (2019) Liquid biopsy in the era of immuno-oncology: is it ready for prime-time use for cancer patients? Ann Oncol 30(9):1448–1459. https://doi.org/10.1093/annonc/mdz196
Hui TP, Kandola A, Shen L et al (2019) A systematic review and meta-analysis of clinical predictors of lithium response in bipolar disorder. Acta Psychiatr Scand 140(2):94–115. https://doi.org/10.1111/acps.13062
Ilie M, Hofman V, Dietel M, Soria J-C, Hofman P (2016a) Assessment of the PD-L1 status by immunohistochemistry: challenges and perspectives for therapeutic strategies in lung cancer patients. Virchows Arch 468(5):511–525. https://doi.org/10.1007/s00428-016-1910-4
Ilie M, Long-Mira E, Bence C et al (2016b) Comparative study of the PD-L1 status between surgically resected specimens and matched biopsies of NSCLC patients reveal major discordances: a potential issue for anti-PD-L1 therapeutic strategies. Ann Oncol off J Eur Soc Med Oncol 27(1):147–153. https://doi.org/10.1093/annonc/mdv489
National Cancer Institute. SEER Cancer Stat Facts: Lung and Bronchus Cancer. National Cancer Institute. Bethesda, MD. Accessed April 11, 2021. https://seer.cancer.gov/statfacts/html/lungb.html
Jensen TJ, Goodman AM, Kato S et al (2019) Genome-wide sequencing of cell-free DNA identifies copy-number alterations that can be used for monitoring response to immunotherapy in cancer patients. Mol Cancer Ther 18(2):448–458. https://doi.org/10.1158/1535-7163.MCT-18-0535
Jones AP, Riley RD, Williamson PR, Whitehead A (2009) Meta-analysis of individual patient data versus aggregate data from longitudinal clinical trials. Clin Trials 6(1):16–27. https://doi.org/10.1177/1740774508100984
Jotte R, Cappuzzo F, Vynnychenko I et al (2020) Atezolizumab in combination with carboplatin and nab-paclitaxel in advanced squamous NSCLC (IMpower131): results from a randomized phase III trial. J Thorac Oncol 15(8):1351–1360
Kerr KM, Hirsch FR (2016) Programmed Death Ligand-1 Immunohistochemistry: Friend or Foe? Arch Pathol Lab Med 140(4):326–331. https://doi.org/10.5858/arpa.2015-0522-SA
Lambert PC, Sutton AJ, Abrams KR, Jones DR (2002) A comparison of summary patient-level covariates in meta-regression with individual patient data meta-analysis. J Clin Epidemiol 55(1):86–94. https://doi.org/10.1016/S0895-4356(01)00414-0
Lu S, Stein JE, Rimm DL et al (2019) Comparison of biomarker modalities for predicting response to PD-1/PD-L1 checkpoint blockade: a systematic review and meta-analysis. JAMA Oncol 5(8):1195–1204. https://doi.org/10.1001/jamaoncol.2019.1549
Lu S, Wang J, Yu Y et al (2021) Tislelizumab plus chemotherapy as first-line treatment for locally advanced or metastatic non-squamous NSCLC (RATIONALE 304): a randomized phase 3 trial. J Thorac Oncol 16(9):1512–1522
McLaughlin J, Han G, Schalper KA et al (2016) Quantitative assessment of the heterogeneity of PD-L1 expression in non-small-cell lung cancer. JAMA Oncol 2(1):46–54. https://doi.org/10.1001/jamaoncol.2015.3638
Mitsudomi T (2014) Molecular epidemiology of lung cancer and geographic variations with special reference to EGFR mutations. Transl Lung Cancer Res 3(4):205–211. https://doi.org/10.3978/j.issn.2218-6751.2014.08.04
Nishio M, Barlesi F, West H et al (2021) Atezolizumab plus chemotherapy for first-line treatment of non-squamous NSCLC: results from the randomized phase 3 IMpower132 trial. J Thorac Oncol 16(4):653–664
Page MJ, McKenzie JE, Bossuyt PM, The PRISMA et al (2020) statement: an updated guideline for reporting systematic reviews. BMJ 2021:372. https://doi.org/10.1136/bmj.n71
Patsopoulos NA, Evangelou E, Ioannidis JPA (2008) Sensitivity of between-study heterogeneity in meta-analysis: proposed metrics and empirical evaluation. Int J Epidemiol 37(5):1148–1157. https://doi.org/10.1093/ije/dyn065
Paz-Ares L, Luft A, Vicente D et al (2018) Pembrolizumab plus Chemotherapy for Squamous Non–Small-Cell Lung Cancer. N Engl J Med 379(21):2040–2051. https://doi.org/10.1056/NEJMoa1810865
Peng L, Liang W-H, Mu D-G et al (2021) First-line treatment options for PD-L1–negative non-small cell lung cancer: a bayesian network meta-analysis. Front Oncol 11:2371
Ready N, Hellmann MD, Awad MM et al (2019) First-line nivolumab plus ipilimumab in advanced non–small-cell lung cancer (checkmate 568): outcomes by programmed death ligand 1 and tumor mutational burden as biomarkers. J Clin Oncol 37(12):992–1000. https://doi.org/10.1200/JCO.18.01042
Ren S, Chen J, Xu X et al (2022) Camrelizumab plus carboplatin and paclitaxel as first-line treatment for advanced squamous NSCLC (CameL-Sq): a phase 3 trial. J Thorac Oncol 17(4):544–557
Rittmeyer A, Barlesi F, Waterkamp D et al (2017) Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet 389(10066):255–265. https://doi.org/10.1016/S0140-6736(16)32517-X
Roach C, Zhang N, Corigliano E et al (2016) Development of a companion diagnostic PD-L1 immunohistochemistry assay for pembrolizumab therapy in non-small-cell lung cancer. Appl Immunohistochem Mol Morphol AIMM 24(6):392–397. https://doi.org/10.1097/PAI.0000000000000408
Rodríguez-Abreu D, Powell SF, Hochmair MJ et al (2021) Pemetrexed plus platinum with or without pembrolizumab in patients with previously untreated metastatic non-squamous NSCLC: protocol-specified final analysis from KEYNOTE-189. Ann Oncol 32(7):881–895. https://doi.org/10.1016/j.annonc.2021.04.008
Sezer A, Kilickap S, Gümüş M et al (2021) Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial. Lancet 397(10274):592–604. https://doi.org/10.1016/S0140-6736(21)00228-2
Sterne JAC, Egger M (2001) Funnel plots for detecting bias in meta-analysis: guidelines on choice of axis. J Clin Epidemiol 54(10):1046–1055. https://doi.org/10.1016/S0895-4356(01)00377-8
Teramukai S, Matsuyama Y, Mizuno S, Sakamoto J (2004) Individual patient-level and study-level meta-analysis for investigating modifiers of treatment effect. Jpn J Clin Oncol 34(12):717–721. https://doi.org/10.1093/jjco/hyh138
Wang J, Lu S, Yu X et al (2021) Tislelizumab plus chemotherapy vs chemotherapy alone as first-line treatment for advanced squamous non–small-cell lung cancer: a phase 3 randomized clinical trial. JAMA Oncol 7(5):709–717. https://doi.org/10.1001/jamaoncol.2021.0366
West H, McCleod M, Hussein M et al (2019) Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 20(7):924–937. https://doi.org/10.1016/S1470-2045(19)30167-6
Xu Y, Wan B, Chen X, et al. The association of PD-L1 expression with the efficacy of anti- PD-1/PD-L1 immunotherapy and survival of non-small cell lung cancer patients: a meta-analysis of randomized controlled trials. Transl Lung Cancer Res. Published online 2019. https://tlcr.amegroups.com/article/view/31044
Yang Y, Sun J, Wang Z et al (2021) Updated overall survival data and predictive biomarkers of sintilimab plus pemetrexed and platinum as first-line treatment for locally advanced or metastatic non-squamous NSCLC in the Phase 3 ORIENT-11 study. J Thorac Oncol 16(12):2109–2120
Yu H, Chen P, Xia L et al (2021) PD-1/PD-L1 inhibitor plus chemotherapy versus bevacizumab plus chemotherapy in first-line treatment for non-squamous non-small-cell lung cancer. J Immunother Cancer 9(11):e003431. https://doi.org/10.1136/jitc-2021-003431
Zhou C, Chen G, Huang Y et al (2021a) Camrelizumab plus carboplatin and pemetrexed versus chemotherapy alone in chemotherapy-naive patients with advanced non-squamous non-small-cell lung cancer (CameL): a randomised, open-label, multicentre, phase 3 trial. Lancet Respir Med 9(3):305–314. https://doi.org/10.1016/S2213-2600(20)30365-9
Zhou C, Wu L, Fan Y et al (2021b) Sintilimab plus platinum and gemcitabine as first-line treatment for advanced or metastatic squamous NSCLC: results from a randomized, double-blind, phase 3 trial (ORIENT-12). J Thorac Oncol 16(9):1501–1511
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
DW: conceptualization, methodology, software, data curation, writing original draft preparation. PH: writing-reviewing, editing, supervision. SG: methodology, data curation, writing-reviewing, editing, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This is a systematic review and meta-analysis. The institutional ethics committee has confirmed that no ethics approval is required.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wankhede, D., Hofman, P. & Grover, S. PD-1/PD-L1 inhibitors in treatment-naïve, advanced non-small cell lung cancer patients with < 1% PD-L1 expression: a meta-analysis of randomized controlled trials. J Cancer Res Clin Oncol 149, 2179–2189 (2023). https://doi.org/10.1007/s00432-022-04286-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04286-6